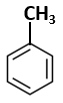
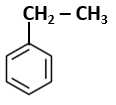
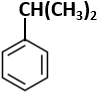
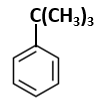
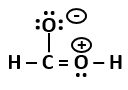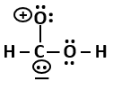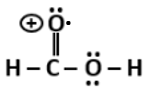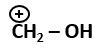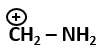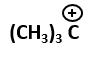Botany - Section A
1. At the root tip, some of the epidermal cells form very fine and delicate, thread-like structures called root hairs in the region of:
| 1. |
Root cap |
2. |
Meristematic activity |
| 3. |
Elongation |
4. |
Maturation |
2. A plasmid has genes for resistance against ampicillin and tetracycline. The tetracycline resistant gene is insertionally inactivated by inserting a foreign DNA within the gene. The bacterial cells are transformed. The recombinant transformants:
| 1. |
will be killed in a medium containing ampicillin but not in a medium containing tetracycline |
| 2. |
will be killed in a medium containing tetracycline but not in a medium containing ampicillin |
| 3. |
will be killed in a medium containing ampicillin and in a medium containing tetracycline |
| 4. |
will not be killed in a medium containing ampicillin and in a medium containing tetracycline |
3. In fermentation carried by yeast:
| 1. |
Two ATP molecules are produced when pyruvic acid is converted to lactic acid |
| 2. |
NAD+ is reduced to NADH |
| 3. |
Yeast poison themselves when the concentration of alcohol reaches about 13 %. |
| 4. |
Carbon dioxide is not produced. |
4. The term used to refer to the use of bio-resources by multinational companies and other organisations without proper authorisation from the countries and people concerned without compensatory payment is:
1. Bioprospecting
2. Biopiracy
3. Biomagnification
4. Bioaccumulation
5. The most important cause of biodiversity loss that the world is facing right now is:
| 1. |
Habitat loss and fragmentation |
| 2. |
Over-exploitation |
| 3. |
Invasion from alien species |
| 4. |
Co-extinctions |
6. Consider the following statements:
| I: |
The reaction centre chlorophyll a in PS I is called P700 |
| II: |
Oxygen evolving complex is associated with PS II |
| III. |
Both ATP and NADPH are made during cyclic photophosphorylation |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
7. Splitting of centromere occurs in:
| 1. |
Mitotic anaphase and Anaphase II |
| 2. |
Mitotic anaphase and Anaphase I |
| 3. |
Mitotic metaphase and Metaphase II |
| 4. |
Mitotic metaphase and Metaphase I |
8. In Polymerase chain reaction:
| 1. |
the correct chronological sequence of steps is: Annealing of primer → Denaturation → Extension. |
| 2. |
Taq polymerase is an RNA polymerase derived from a fungus. |
| 3. |
millions to billions of copies of a specific DNA sample are made. |
| 4. |
viral antigens cannot be detected in a given sample. |
9. Which of the following is not correctly matched?
|
Vegetative propagule |
Example |
| 1. |
Eye |
Ginger |
| 2. |
Bulbil |
Agave |
| 3. |
Leaf buds |
Bryophyllum |
| 4. |
Offset |
Water hyacinth |
10. In a monocot seed, the aleurone layer describes:
1. a proteinaceous outermost layer of the endosperm
2. the shield shaped cotyledon
3. sheath enclosing the plumule
4. sheath enclosing the radicle
11. Degradation of ozone continues for a very long time when CFCs are added to the stratosphere because of the formation of which of the following catalyst?
1. Carbon
2. Cl
3. Fe
4. Oxygen
12. The somatic cell of an organism has a diploid chromosome number of 16 [2N = 16]. The number of the chromosomes in this cell at the end of the S-phase will be:
13. Who popularised the term ‘Biodiversity’ to describe the combined diversity at all levels of biological organisation?
1. Robert May
2. Paul Ehrlich
3. Edward Wilson
4. Alexander von Humboldt
14. Match each item in Column I with one in Column II and choose the correct option from the codes given below:
|
Column I |
|
Column II |
| A. |
Primary Succession |
P. |
Takes place in dry areas and the series progress from xeric to mesic conditions |
| B. |
Secondary Succession |
Q. |
Occurs in areas like newly cooled lava, bare rock, newly created ponds or reservoirs and is generally slow |
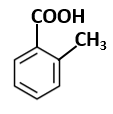
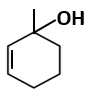
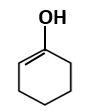
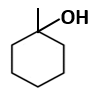
| C. |
Hydrarch Succession |
R. |
Takes place in wet areas and the series progress from hydric to mesic conditions |
| D. |
Xerarch Succession |
S. |
Occurs in areas where natural biotic communities have been destroyed like abandoned farmlands, burned or cut forests, lands that have been flooded and is faster |
|
A |
B |
C |
D |
| 1. |
Q |
S |
R |
P |
| 2. |
S |
Q |
P |
R |
| 3. |
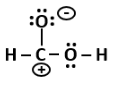
P |
R |
Q |
S |
| 4. |
R |
P |
S |
Q |
15. What is the correct order of the steps involved in the sexual cycle of organisms belonging to the Kingdom Fungi?
| 1. |
Meiosis in zygote — Plasmogamy — Karyogamy |
| 2. |
Karyogamy — Meiosis in zygote — Plasmogamy |
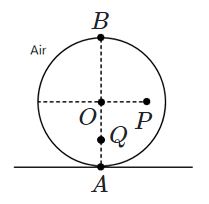
| 3. |
Plasmogamy — Karyogamy — Meiosis in zygote |
| 4. |
Karyogamy — Plasmogamy — Meiosis in zygote |
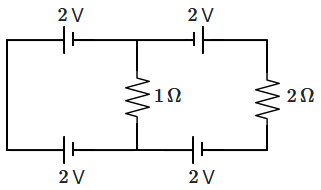
16. Agrobacterium tumefaciens infects the plant through its:
| 1. |
Chromosomal DNA |
2. |
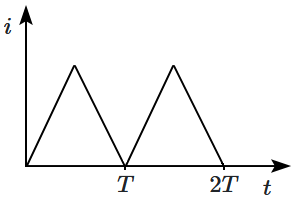
Complementary DNA |
| 3. |
F plasmid |
4. |
Ti plasmid |
17. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Cork cambium |
P. |
Phellogen |
| B. |
Cork |
Q. |
Phellem |
| C. |
Secondary cortex |
R. |
Phelloderm |
| D. |
Bark |
S. |
All tissues exterior to the vascular cambium |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
Q |
R |
S |
P |
| 3. |
R |
S |
P |
Q |
| 4. |
S |
P |
Q |
R |
18. Which of the following is true regarding the classes of Bryophytes?
| 1. |
The thallus of mosses is dorsiventral and closely appressed to the surface |
| 2. |
In the life cycle of liverworts, the predominant gametophytic stage is divided into two stages - the protonema and the leafy stage |
| 3. |
The sporophyte in liverworts is more elaborate than in mosses |
| 4. |
Asexual reproduction in liverworts takes place by fragmentation of thalli, or by the formation of specialised structures called gemmae |
19. Regarding nitrogen cycle:
| I: |
Nitrosomanas and/or Nitrococcus oxidize ammonia to nitrite. |
| II: |
Nitrite is oxidized to nitrate by Nitrobacter. |
| III. |
Denitrification is carried out by Pseudomonas and Thiobacillus. |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
20. Consider the two statements:
| I: |
Intercalary meristem is a primary meristem and contributes to the formation of the primary plant body. |
| II: |
Cork cambium is a lateral meristem and produces secondary tissues. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
21. Regarding factors affecting enzyme activity:
| I: |
Low temperature preserves the enzyme in a temporarily inactive state whereas high temperature destroys enzymatic activity. |
| II: |
With the increase in substrate concentration, the velocity of the enzymatic reaction rises at first. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
22. Identify the correct statement:
| 1. |
α amino acids are substituted methanes |
| 2. |
Only triglycerides are lipids that are both macromolecules as well as polymers |
| 3. |
Cellulose forms secondary helical structures and can hold iodine molecules in the helical portion |
| 4. |
Every virus will have both DNA and RNA as its genetic material |
23. Match each item in Column-I with one in Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Auxins |
P. |
Indole compounds |
| B. |
Cytokinins |
Q. |
Skoog and Miller |
| C. |
Gibberellins |
R. |
Terpenes |
| D. |
Ethylene |
S. |
Cousins |
Codes:
|
A |
B |
C |
D |
| 1. |
P |
Q |
R |
S |
| 2. |
P |
R |
S |
Q |
| 3. |
R |
P |
Q |
S |
| 4. |
R |
S |
P |
Q |
24. Two of the most common gases responsible for the ‘greenhouse effect’ are:
1. Carbon dioxide and methane
2. Ozone and Ammonia
3. Oxygen and Nitrogen
4. Nitrogen and Sulphur dioxide
25. In an angiosperm, the ovule is attached to the placenta by a stalk called:
1. Funicle
2. Raphe
3. Hilum
4. Chalaza
26. In which of the following class of fungi, sex organs are absent and plasmogamy is brought about by fusion of two vegetative or somatic cells of different strains or genotypes?
1. Ascomycetes
2. Phycomycetes
3. Basidiomycetes
4. Deuteromycetes
27. In the root, the movement of water is symplastic in the:
1. Pericycle
2. Cortex
3. Endodermis
4. Epidermis
28. Identify the incorrect statement:
| 1. |
Somatic hybridization has been used to produce pomato, though it is not used commercially |
| 2. |
Methylophilus methylotropus [bacterium] and Spirulina [blue-green algae] are sources of single-cell protein |
| 3. |
Plant cells do not show cellular totipotency and hence cannot be cultured |
| 4. |
Gamma radiations are used to induce mutations in plants |
29. Nucellar polyembryony is seen in:
1. Citrus and Mango
2. Castor and Pea
3. Grasses and Mustard
4. Orchids and China rose
30. A persistent nutritive tissue of a seed derived from the nucellus and deposited external to the embryo sac —distinguished from endosperm is known as:
1. Tegmen
2. Chalaza
3. Perisperm
4. Hilum
31. Match each item in Column I with one in Column II and choose the correct option from the codes given below:
|
Column I |
|
Column II |
| A. |
Psilopsida |
P. |
Selanginella |
| B. |
Lycopsida |
Q. |
Adiantum |
| C. |
Sphenopsida |
R. |
Equisetum |
| D. |
Pteropsida |
S. |
Psilotum |
|
A |
B |
C |
D |
| 1. |
R |
Q |
S |
P |
| 2. |
S |
P |
R |
Q |
| 3. |
P |
S |
Q |
R |
| 4. |
Q |
R |
P |
S |
32. Match the stage of Prophase I of Meiosis I given in
Column-I with the corresponding event in
Column-II and select the correct match from the codes given below:
|
Column-I |
|
Column-II |
| A. |
Zygotene |
P. |
Crossing over |
| B. |
Pachytene |
Q. |
Synapsis |
| C. |
Diplotene |
R. |
Terminalization of chiasmata |
| D. |
Diakinesis |
S. |
Appearance of chiasmata |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
P |
S |
R |
| 2. |
P |
Q |
S |
R |
| 3. |
P |
Q |
R |
S |
| 4. |
Q |
P |
R |
S |
33. Which of the following is a correct match between the biological name and the order of the organism?
1. Homo sapiens – Hominidae
2. Mangifera indica - Anacardiaceae
3. Triticum aestivum - Poaceae
4. Mangifera indica - Sapindales
34. Identify the incorrect match:
|
mRNA codon |
Amino acid |
| 1. |
AUG |
Methionine |
| 2. |
GGG |
Glycine |
| 3. |
UGA |
Tryptophan |
| 4. |
UAG |
None; it is a stop codon |
35. Identify the incorrectly matched pair:
| 1. |
Primary constriction |
Satellite chromosome |
| 2. |
Cilia/flagella |
9 + 2 arrangement of microtubules |
| 3. |
Centriole |
9 + 0 arrangement of microtubules |
| 4. |
Lysosomes |
Acid hydrolases |
Botany - Section B
36. In ‘competitive release’:
| 1. |
differences among similar species whose distributions overlap geographically are accentuated in regions where the species co-occur, but are minimized or lost where the species' distributions do not overlap. |
| 2. |
two species which compete for the same limited resource cannot coexist at constant population values. |
| 3. |
there is an expansion of the range of a species when a competitor for its niche is removed. |
| 4. |
there is a division of limited resources by species to help avoid competition in an ecological niche. |
37. After vernalization:
| 1. |
flowering is always immediately induced in the plants |
| 2. |
may require additional seasonal cues or weeks of growth before they will actually flower |
| 3. |
seeds cannot germinate in spite of favorable environmental conditions |
| 4. |
flowering is suppressed in the plants |
38. In glycolysis [EMP pathway], the number of ATP molecules directly synthesized from one glucose molecule are:
1. 2
2. 4
3. 6
4. 10
39. Identify the incorrectly matched pair:
| 1. |
Mung bean |
Resistance to powdery mildew transferred from a wild species |
| 2. |
Atlas 66 |
Protein enriched wheat |
| 3. |
Golden rice |
Vitamin A enriched rice |
| 4. |
Pusa sadabahar |
Chilli variety resistant to chilli mosaic virus |
40. Identify the option where the name of the scientist is not correctly matched with discovery related to understanding of the process of photosynthesis:
| 1. |
Cornelius van Neil |
Oxygen evolved comes from water |
| 2. |
T W
Engelmann |
First absorption spectrum of chlorophyll a |
| 3. |
Joseph Priestley |
Plants restore to the air whatever breathing animals remove |
| 4. |
Jan Ingenhousz |
Sunlight is essential for photosynthesis |
41. The physical property that describes attraction of water molecules to polar surface [such as the surface of tracheary elements] is known as:
1. Cohesion
2. Adhesion
3. Surface tension
4. Tensile strength
42. Regarding cell membrane:
| I: |
The lipid content mainly is phosphoglycerides |
| II: |
Proteins can move laterally within the overall bilayer |
| III: |
The membrane is selectively permeable |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
43. Identify the incorrect statement:
| 1. |
Robert May estimates the global species diversity to be about 7 million |
| 2. |
India is one of the 12 mega diversity countries in the world |
| 3. |
Amazon rainforests produce 20 percent of total oxygen in the Earth’s atmosphere |
| 4. |
The Earth Summit was held in Rio de Janeiro in 2002 |
44. Consider the given two statements:
| I: |
Lipids come under acid insoluble fraction. |
| II: |
Lipids are macromolecules. |
| 1. |
Both I and II are correct and II explains I |
| 2. |
Both I and II are correct but II does not explain I |
| 3. |
I is correct but II is incorrect |
| 4. |
I is incorrect but II is correct |
45. Kingdom Protista has brought together Chlamydomonas, Chlorella with Paramoecium and Amoeba. On what basis were these organisms separated under previous classification systems?
1. Cell wall
2. Cell type
3. Body organisation
4. Mode of nutrition
46. The tRNA molecule:
| 1. |
looks like an inverted L in its secondary structure |
| 2. |
is the largest of the major cellular RNAs |
| 3. |
acts as an adapter molecule |
| 4. |
has catalytic functions |
47. Glomus is:
| 1. |
the genus of fungus, the members of which help symbiont plants get phosphorus |
| 2. |
the genus of fungus, the members of which help symbiont plants get nitrogen |
| 3. |
the genus of plants, the members of which are symbionts of mycorrhizae |
| 4. |
the genus of fungus, the members of which help processing of certain varieties of cheese |
48. Which of the following biomes has a mean annual precipitation of 150-420 cm and a mean annual temperature of 200- 240 C?
1. Tropical Forest
2. Grassland
3. Temperate Forest
4. Coniferous Forest
49. The amount of energy which the plants can capture is:
| 1. |
2-10 per cent of the incident solar radiation |
| 2. |
5-10 per cent of the photosynthetically active radiation |
| 3. |
5-10 per cent of the incident solar radiation |
| 4. |
2-10 per cent of the photosynthetically active radiation |
50. In a plant cell:
| 1. |
there are multiple vacuoles, each small in size |
| 2. |
centrioles play prominent role in organization of microtubules |
| 3. |
cytokinesis occurs through cleavage furrow |
| 4. |
Secondary cell wall is formed on the inner [towards membrane] side of the cell |
Zoology - Section A
51. Which of the following sexually transmitted infection is caused by a bacterium and is curable if diagnosed early?
| 1. |
Syphilis |
2. |
HIV |
| 3. |
Genital herpes |
4. |
Trichomoniasis |
52. The karyotype of a person suffering from Down’s syndrome will show:
| 1. |
44 Autosomes + XXY |
| 2. |
44 Autosomes + XO |
| 3. |
45 Autosomes [trisomy #21] + XY |
| 4. |
43 Autosomes [monosomy #21] + XY |
53. Identify the incorrect statement:
| 1. |
Electrostatic precipitator can remove over 99 percent of particulate matter present in the exhaust from a thermal power plant. |
| 2. |
According to CPCB, particulate sizes of more than 10 micrometers are responsible for causing the greatest harm to human health. |
| 3. |
Motor vehicles equipped with catalytic converters should use unleaded petrol. |
| 4. |
CNG burns more efficiently than petrol or diesel. |
54. Identify the incorrectly matched pair regarding the life cycle of malaria parasite:
| 1. |
Sporozoite |
Infective stage for humans |
| 2. |
Liver cell |
Asexual reproduction |
| 3. |
RBC |
Sexual reproduction |
| 4. |
Fertilization |
In the mosquito gut |
55. Which of the following is not aneuploidy?
1. Down’s syndrome
2. Klinefelter’s syndrome
3. Turner’s syndrome
4. Phenylketonuria
56. Match each item in Column I with one in COLUMN II and select the correct match from the codes given:
|
COLUMN I |
|
COLUMN II |
| A |
Insulin |
P |
Increases blood glucose levels |
| B |
Glucagon |
Q |
Decreases blood glucose levels |
| C |
Parathyroid
hormone |
R |
Increases blood calcium levels |
| D |
Thyrocalcitonin |
S |
Decreases blood calcium levels |
Codes
|
A |
B |
C |
D |
| 1. |
Q |
P |
S |
R |
| 2. |
Q |
P |
R |
S |
| 3. |
P |
Q |
R |
S |
| 4. |
P |
Q |
S |
R |
57. Identify the incorrect statement:
| 1. |
Metamerism appeared for the first time in annelida |
| 2. |
Arthropods have jointed appendages and chitinous exoskeleton |
| 3. |
Reptiles are endotherms |
| 4. |
Forelimbs of birds are modifies into wings |
58. On an average a female cockroach produces:
1. 9-10 oothecae , each containing 14-16 eggs
2. 14-16 oothecae , each containing 9-10 eggs
3. 5-6 oothecae , each containing 9-10 eggs
4. 9-10 oothecae , each containing 5-6 eggs
59. Generally, the shape not seen in a bacterial cell is:
1. Rod like
2. Comma shaped
3. Spherical
4. Icosahedral
60. Identify the incorrectly matched pair:
| 1. |
Limbic system |
Behaviour |
| 2. |
Medulla oblongata |
Gastric secretions |
| 3. |
Hypothalamus |
Thermoregulation |
| 4. |
Corpus callosum |
Connects cerebellum with rest of the brain |
61. Angiotensin:
| I: |
is a vasoconstrictor |
| II: |
stimulates the adrenal cortex to secrete aldosterone |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
62. All the following statements are correct except:
| 1. |
Neutrophils and monocytes are the main phagocytic cells in human blood. |
| 2. |
Mature RBC does not have a nucleus and is biconcave disc-shaped. |
| 3. |
A person with type A blood will have anti-B antibodies in his plasma. |
| 4. |
Erythroblastosis foetal occurs when an Rh +ve mother is carrying an Rh –ve foetus. |
63. Identify the incorrect statement regarding thalassemia:
| 1. |
There is reduced synthesis of one of the globin chains. |
| 2. |
It is an autosome-linked dominant blood disease. |
| 3. |
Alpha thalassemia is controlled by two closely linked genes on chromosome 16. |
| 4. |
Beta thalassemia is controlled by a single gene on chromosome 11. |
64. Foetal ejection reflex is initiated by:
| 1. |
Foetal hypothalamus |
| 2. |
Maternal pituitary |
| 3. |
Fully developed foetus and placenta |
| 4. |
Maternal hypothalamus |
65. If you draw the age pyramid for a population that is expanding in size, you would expect that:
| 1. |
the number of individuals in the pre-reproductive age group are more than the number of individuals in the reproductive age group. |
| 2. |
the number of individuals in the reproductive age group are less than the number of individuals in the post-reproductive age group. |
| 3. |
the number of individuals in the reproductive age group are equal to the number of individuals in the reproductive age group. |
| 4. |
the number of individuals in the pre-reproductive age group are less than the number of individuals in the reproductive age group. |
66. Identify the incorrect statement regarding transgenic animals:
| 1. |
α-1 antitrypsin is being produced in a transgenic sheep |
| 2. |
The first transgenic cow, Rosie, produced human protein-enriched milk (2.4 grams per litre). |
| 3. |
Transgenic mice are being used to test the safety of the polio vaccine. |
| 4. |
Over 95 percent of all existing transgenic animals are cattle. |
67. A characteristic 9 + 2 arrangement of axonemal microtubules is seen in:
1. Cilia and flagella
2. Centriole
3. Filaments in axons of neurons
4. Actin myofilaments
68. The first restriction endonuclease, whose functioning depended on a specific DNA nucleotide sequence was:
| 1. |
EcoR I |
2. |
BamH I |
| 3. |
Hind II |
4. |
Sma I |
69. Which of the following is incorrect regarding acquired immunity?
| 1. |
It is a function of lymphocytes |
| 2. |
It retains memory of previous encounters with pathogens |
| 3. |
It is non-specific and present since birth |
| 4. |
It has the ability to distinguish between self and non-self |
70. Urea can be used in humans to increase the osmolarity primarily in:
| 1. |
Blood |
2. |
Renal cortex |
| 3. |
Renal medulla |
4. |
Urinary bladder |
71. In a continuous culture system:
| I: |
The used medium is drained out from one side while the fresh medium is added from the other. |
| II: |
The cells are maintained in their physiologically most active log/exponential phase. |
| III: |
A larger biomass is produced leading to higher yields of desired proteins. |
| 1. |
Only I and II are correct |
| 2. |
Only I and III are correct |
| 3. |
Only II and III are correct |
| 4. |
I, II and III are correct |
72. Out-cross is very important in animal breeding programs because:
| I: |
It is the best method for animals that are below average in productivity in milk production, growth rate of beef cattle etc. |
| II: |
A single outcross often helps to overcome inbreeding depression. |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
73. Select the incorrectly matched pair from the following options:
| 1. |
Fragmentation - breakdown of detritus into smaller particles by detrivores |
| 2. |
Leaching - water soluble inorganic nutrients go down into the soil horizon and get precipitated as unavailable salts |
| 3. |
Catabolism - bacterial and fungal enzymes degrade simple inorganic substances into detritus |
| 4. |
Humification - accumulation of a dark coloured amorphous substance that is highly resistant to microbial action and undergoes decomposition at an extremely slow rate |
74. The length of which of the following structures remains same during muscle contraction by sliding filament mechanism?
| 1. |
H zone |
2. |
A band |
| 3. |
I band |
4. |
Sarcomere |
75. In the human genome:
| 1. |
the total number of genes are about 60000 |
| 2. |
less than 2 percent of genome does not code for proteins |
| 3. |
repeated sequences are rarely found |
| 4. |
SNPa occur at about 1.4 million locations |
76. Communication junctions called as intercalated discs are seen in:
1. Cardiac muscle
2. Bone
3. Cartilage
4. Neuron
77. Match each item in
Column-I with one in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| A. |
Dengue and Chikungunya |
P. |
Aedes mosquito |
| B. |
Ringworm |
Q. |
Microsporum, Epidermophyton, and Trichophyton |
| C. |
Amoebic dysentery |
R. |
Wuchereria bancrofti |
| D. |
Filariasis |
S. |
Entamoeba histolytica |
Codes:
|
A |
B |
C |
D |
| 1. |
Q |
P |
R |
S |
| 2. |
P |
Q |
S |
R |
| 3. |
R |
S |
Q |
P |
| 4. |
S |
R |
P |
Q |
78. Fimbriae in bacterial cells:
| 1. |
play an important role in motility |
| 2. |
attach the cell to the rocks in a stream and also to the host tissues |
| 3. |
provide surface area for the location of enzymes |
| 4. |
form a tube like structure for passage of DNA during bacterial conjugation |
79. If the gene coding for permease enzyme in the lac operon is inactivated by a mutation:
| 1. |
the cell will not be able to metabolize glucose |
| 2. |
the cell will not be able to breakdown lactose |
| 3. |
lactose will not be able to enter the cell |
| 4. |
the catabolite activator protein will increase the rate of transcription of lac operon |
80. The members of which of the following phylum are bilaterally symmetrical, triploblastic and pseudocoelomates?
| 1. |
Platyhelminthes |
2. |
Annelids |
| 3. |
Mollusc |
4. |
Aschelminthes |
81. Identify the incorrectly matched pair:
| 1. |
Tidal volume in a healthy human adult |
About 500 ml |
| 2. |
Partial pressure of oxygen in oxygenated blood |
159 mm Hg |
| 3. |
Resting respiratory rate in a healthy adult |
12 to 16 per minute |
| 4. |
Percent of carbon dioxide transported as bicarbonate in the blood |
70 |
82. After absorption, the end products of digestion of which of the following biomolecules are transported by central lacteals?
1. Lipids
2. Carbohydrates
3. Proteins
4. Nucleic acids
83. Identify the correct statement:
| 1. |
In spermiogenesis, spermatids are formed, while in spermiation, spermatozoa are formed. |
| 2. |
Spermatogonia are diploid and primary spermatocytes are haploid. |
| 3. |
Testes descend into the scrotum after birth but before puberty while ovaries do not descend at all. |
| 4. |
The Infundibulum of fallopian tube possesses fimbriae while fertilization occurs in the ampulla of the fallopian tube. |
84. Which vitamin, important for effective erythropoiesis, is added by LAB when it is used to convert milk into curd?
1. Vitamin D
2. Vitamin A
3. Vitamin B12
4. Vitamin E
85. A method used in molecular biology for the detection of a specific DNA sequence in DNA samples is:
| 1. |
Northern blotting |
2. |
Western blotting |
| 3. |
Southern blotting |
4. |
Eastern blotting |
Zoology - Section B
86. The bacterium
Escherichia coli was not used by:
| 1. |
Fred Griffith, when he discovered bacterial transformation |
| 2. |
Alfred Hershey and Martha Chase, when they conclusively proved that DNA is the molecule that stores genetic information |
| 3. |
Matthew Meselson and F. Stahl, when they experimentally proved that DNA replicates semi-conservatively |
| 4. |
Francois Jacob and Jacques Monod, when they proposed the working of lactose operon |
87. Consider the following two statements and choose the option which best describes the relation between them:
| Statement 1: |
Thermoregulation is energetically expensive for small animals like shrews and humming birds |
| Statement 2: |
Small animals have a larger volume as compared to their surface are so they tend to lose body heat very fast when it is cold outside |
| 1. |
Both 1 and 2 are true and 2 is the correct explanation for 1 |
| 2. |
Both 1 and 2 are true but 2 does not explain 1 |
| 3. |
1 is true but 2 is false |
| 4. |
2 is true but 1 is false |
88. Identify the incorrect statement:
| 1. |
The muscularis layer in the wall of gut has smooth muscles |
| 2. |
Enterokinase is secreted by the pancreatic acini |
| 3. |
Brunner’s glands are present in the submucosa of gut wall |
| 4. |
Sphincter of Oddi is present at the junction of oesophagus and stomach |
89. What prevents the development of new follicles during the post-ovulatory luteal phase of the menstrual cycle?
| 1. |
A very low level of progesterone and oestrogen secretion by the corpus luteum |
| 2. |
Secretion of high levels of FSH by the corpus luteum |
| 3. |
LH surge just before ovulation |
| 4. |
Low levels of both FSH and LH levels |
90. Identify the incorrectly matched pair:
| 1. |
Pleiotropy |
Sickle cells anaemia |
| 2. |
Polygenic inheritance |
Human skin colour |
| 3. |
Multiple allelism |
ABO blood group |
| 4. |
Gene/point mutation |
Down’s syndrome |
91. The coxal bone in the human skeleton:
| 1. |
is a part of the pectoral girdle, has a glenoid cavity that articulates with the head of the femur. |
| 2. |
is a part of the pelvic girdle, has an acetabulum that articulates with the head of the humerus. |
| 3. |
is a part of the pelvic girdle and is formed by the fusion of three bones. |
| 4. |
is a part of pectoral girdle and is formed by the fusion of three bones. |
92. Match the pathogens that can cause disease in humans in
Column-I with their correct biological names in
Column-II and select the correct match from the codes given:
|
Column-I |
|
Column-II |
| a. |
Filariasis |
(i) |
Haemophilus influenza |
| b. |
Amoebiasis |
(ii) |
Trichophyton |
| c. |
Pneumonia |
(iii) |
Wuchereria bancrofti |
| d. |
Ringworm |
(iv) |
Entamoeba histolytica |
|
(a) |
(b) |
(c) |
(d) |
| 1. |
(i) |
(ii) |
(iv) |
(iii) |
| 2. |
(ii) |
(iii) |
(i) |
(iv) |
| 3. |
(iv) |
(i) |
(iii) |
(ii) |
| 4. |
(iii) |
(iv) |
(i) |
(ii) |
93. Two heart sounds are normally produced during a cardiac cycle. The first of these is produced due to the:
| 1. |
simultaneous opening of atrio-ventricular valves |
| 2. |
simultaneous opening of semilunar valves |
| 3. |
simultaneous closure of semilunar valves |
| 4. |
simultaneous closure of atrio-ventricular valves |
94. In an operon:
| I: |
a polycistronic structural gene is regulated by a common promoter and regulatory genes |
| II: |
the regulation is always negative and inducible |
| 1. |
Only I is correct |
| 2. |
Only II is correct |
| 3. |
Both I and II are correct |
| 4. |
Both I and II are incorrect |
95. Identify the incorrect statement regarding fallopian tubes:
| 1. |
Infundibulum possesses fimbriae |
| 2. |
Fertilization takes place in the narrowest part of fallopian tube called ampulla |
| 3. |
Isthmus joins the uterus |
| 4. |
It is lined with ciliated epithelium |
96. Which of the following hormones does not act on its target cells through a second messenger?
1. Adrenaline
2. Insulin
3. Thyrocalcitonin
4. Oestrogen
97. Identify the incorrectly match pair:
| 1. |
RuBisCO |
Most abundant protein in the whole of the biosphere |
| 2. |
Insulin |
A polymer of fructose |
| 3. |
Vinblastin |
A secondary metabolite used as a drug |
| 4. |
Adenine |
A substituted purine |
98. The trachea is surrounded by 16 to 20 incomplete and C-shaped rings of:
1. Hyaline cartilage
2. Elastic cartilage
3. Fibrous cartilage
4. Calcified cartilage
99. Dysfunction of which of the following parts of the human brain is most likely to affect learning and memory?
1. Pons
2. Hippocampus
3. Cerebellum
4. Hypothalamus
100. According to the 2017 MTP Amendment, the maximum gestation age till which MTP is allowed is:
1. 16 weeks
2. 20 weeks
3. 24 weeks
4. 28 weeks
Chemistry - Section A
101. The strongest conjugate base is
1.
\(Cl^-\)
2.
\(NO^-_3\)
3.
\(HCO^-_3\)
4.
\(HSO^-_4\)
102. In a compound, atoms of element Y form ccp lattice and those of element X occupy \({ 2 \over 3}rd\) of tetrahedral voids. The formula of the compound will be
1. \(X_3Y_4\)
2. \(X_2Y_3\)
3. \(X_2Y\)
4. \(X_4Y_3\)
103. An ideal solution is made by mixing 2 moles of benzene
\((P^o = 266 ~mm) \) and 3 moles of another liquid
\((P^o= 236 ~mm) \).
The total vapor pressure of the solution at the same temperature would be:
| 1. |
502 mm |
2. |
248 mm |
| 3. |
600 mm |
4. |
298 mm |
104. Benzene ring contains
1. \(12 \sigma\) bonds and \(3 \pi\) bonds
2. \(9\sigma\) bonds and \(3 \pi\) bonds
3. \(6\sigma\) bonds and \(3 \pi\) bonds
4. \(6\sigma\) bonds and \(6 \pi\) bonds
105. Which, of the following, has the least sweetness value among artificial sweeteners?
1. Aspartame
2. Saccharin
3. Sucralose
4. Alitame
106. In which of the following molecule is the bond angle maximum?
1. \(NF_3\)
2. \(NH_3\)
3. \(PH_3\)
4. \(H_2O\)
107. Benzene on ozonolysis gives
1. Acetone
2. Glyoxal
3. Propanol
4. Butanone
108. The number of moles of
\(K_2Cr_2O_7\) required to oxidise one mole of
\(FeSO_4\) in acidic medium is:
| 1. |
6 |
2. |
\(1 \over 2\) |
| 3. |
\(1 \over 3\) |
4. |
\(1 \over 6\) |
109. The best reducing agent among the following is:
1. \(GeCl_2\)
2. \(SnCl_2\)
3. \(PbCl_2\)
4. \(GeCl_4\)
110. Which of the following is an amorphous solid?
1. \(Zn\)
2. \(NaCl\)
3. Glass
4. \(CaF_2\)
111. \(R-COOH\xrightarrow[(2)~Br_2/KOH]{(1)~ND_3,~\Delta}A~\text(major).\)
The compound \('A'\) is:
1. \(R-COND_2\)
2. \(R-NH_2\)
3. \(R-ND_2\)
4. \(R-CONH_2\)
112. The intensive property among the following is:
1. Volume
2. Enthalpy
3. Molar heat capacity
4. Internal energy
113. Bredig's Arc method for the preparation of metallic sol is
1. Dispersion method
2. Condensation method
3. Dispersion as well as a condensation method
4. None of the above
114. Which of the following element does not show a variable oxidation state?
1. \(Zn\)
2. \(Fe\)
3. \(Mn\)
4. \(Cu\)
115. If the volume of 2 moles of a gas at 546 K is 44.8 liters, the pressure of the gas will be
1. 1 atm
2. 2 atm
3. 3 atm
4. 4 atm
116. Given the reaction:
R-OH + PCl
5 \( \longrightarrow \) R-Cl + POCl
3 + HCl
The most reactive alcohol in the above reaction will be:
| 1. |
1o Alcohol |
2. |
2o Alcohol |
| 3. |
3o Alcohol |
4. |
Cannot be predicted |
117. Which, of the following, ores is concentrated by froth flotation method?
1. Cuprite
2. Haematite
3. Chalcopyrite
4. Alum stone
118. The hardness of water cannot be removed by
1. Treating with washing soda
2. Boiling
3. Adding permutit
4. Adding chlorine
119. Consider the following reaction:
\(2SO_2 + O_2 \rightarrow 2SO_3 .\)
The rate of disappearance of \(O_2\) is \(2 \times 10^{-4}~mole~ l^{-1} ~min^{-1}\). The rate of appearance of \(SO_3\) will be
1. \(2 \times 10^{-4} ~mole~l^{-1}~min^{-1}\)
2. \(4 \times 10^{-4} ~mole~l^{-1}~min^{-1}\)
3. \(6 \times 10^{-4} ~mole~l^{-1}~min^{-1}\)
4. \(1 \times 10^{-4} ~mole~l^{-1}~min^{-1}\)
120. Which compound is most reactive towards electrophilic substitution reaction?
121. How many nodal planes are present in \(4~dz^2\) ?
1. 1
2. 2
3. 3
4. Zero
122. When
\(CuSO_4\) solution is electrolysed using inert electrodes, the incorrect statement regarding this electrolysis is:
| 1. |
weight of cathode increases |
| 2. |
pH of the solution remains unchanged |
| 3. |
product at the anode is \(O_2\) gas |
| 4. |
weight of the anode is unchanged |
123. When sodium thiosulphate is reacted with I2 , then the product of sulphur will be:
1.
2.
3.
4.
124. Excess of \(AgNO_3\) solution is added to 100 ml of 0.024 M solution of dichloridobis(ethylene diamine)cobalt(III) chloride. How many moles of \(AgCl\) will be precipitated?
1. 0.0012
2. 0.0024
3. 0.0036
4. 0.0048
125. Which of the following, nitrates cannot give light brown gas on heating?
1. \(LiNO_3\)
2. \(NaNO_3\)
3. \(Mg(NO_3)_2\)
4. \(Ca(NO_3)_2\)
126. Which lanthanoid symbol is incorrectly matched with its atomic number?
1. Cerium(Ce) (Z =58)
2. Promethium(Pm) (Z= 61)
3. Europium(Eu) (Z= 64)
4. Dysprosium(Dy) (Z = 66)
127. The step growth polymer is
1. Neoprene
2. Bakelite
3. PVC
4. Polystyrene
128. When toluene is oxidised with chromyl chloride, benzaldehyde is formed. This reaction is known as:
1. Gattermann Koch Synthesis
2. Etard's reaction
3. Schmidt's reaction
4. Friedel Craft reaction
129. 10 gm carbon reacts with 100 gm \(Cl_2\) to form \(CCl_4.\) The maximum weight of \(CCl_4\) formed will be:
1. 128 gm
2. 94.22 gm
3. 108.45 gm
4. 54.22 gm
130. The first electron affinity values of O, S, Se and Te are given correctly as :
1. \(S > O> Se > Te\)
2. \(O > S > Se > Te\)
3. \(S > Se > Te >O\)
4. \(S > Se> O > Te\)
131. 3-Methylpentane forms all the possible monohalogenated products. Among all monohalogenated products, how many products can show optical isomerism?
1. 1
2. 2
3. 3
4. 4
132. Which one of the following, gases is colourless, highly toxic, and reduces oxygen carrying capacity of blood?
1. \(SO_2\)
2. \(CO \)
3. \(NO \)
4. \(SO_3\)
133. Which of the following compounds is the most acidic?
134. The most basic hydride of Group 15 is:
1. \(NH_3 \)
2. \(PH_3 \)
3. \(AsH_3 \)
4. \(BiH_3 \)
135. Which of the following ions has the maximum magnetic moment?
1. \(Mn^{3+}\)
2. \(Cu^{2+}\)
3. \(Fe^{3+}\)
4. \(V^{3+}\)
Chemistry - Section B
136. Alkali metals in liquid
\(NH_3\) produce a blue coloured solution. It is due to
1. Polarisability of
\(NH_3\)
2. Charge transfer
3. Ammoniated ions
4. Ammoniated electrons
137. Consider the following cell:
\(Pt, H_2|H^+||H^+|H_2, Pt~~, \)
If a solution of pH = 3 is kept at the cathode and a solution of pH = 5 is kept at the anode, then \(E_{cell} \) at 25 °C will be:
1. –0.059 V
2. +0.059V
3. +0.118 V
4. –0.118 V
138. For an exothermic reaction, the value of activation energy for forward reaction is 40 Joule and \(\Delta H\) is -60 Joule. The activation energy for backward reaction will be:
1. 20 Joule
2. 40 Joule
3. 80 Joule
4. 100 Joule
139. For the reversible reaction
\(N_2(g)+3H_2(g)⇌2NH_3(g)\), what is the effect on the equilibrium position when an inert gas is added at constant volume?
| 1. |
Shifts in the forward reaction |
| 2. |
Shifts in backward reaction |
| 3. |
Remains unaffected |
| 4. |
Initially in the forward direction and then in the backward direction |
140. Which, of the following compounds, can give iodoform reaction?
| 1. |
\(~~~~~~~~~~~~O\\ ~~~~~~~~~~~~||\\ HO-C-CH_3\) |
| 2. |
\(~~~~~~~~~~~~~O\\ ~~~~~~~~~~~~~~||\\ H_2N-C-CH_3\) |
| 3. |
\(~~~~~~~~~~~~~O\\ ~~~~~~~~~~~~~||\\ CH_3 -C - CH_3\) |
| 4. |
All of the above |
141. When
\(NH_3\) is treated with HCl,
\(H - N - H\) bond angle:
| 1. |
Decreases |
2. |
Increases |
| 3. |
Remains the same |
4. |
Can't be predicted |
142. Which, of the following alcohols, is most reactive toward dehydration?
143. Given the reaction:
\(A + B \rightarrow C+D - q~ cal. \) is found to have a positive entropy change. The reaction will be:
| 1. |
Possible at high temperature. |
| 2. |
Possible only at low temperatures. |
| 3. |
Not possible at any temperature. |
| 4. |
Possible at any temperature. |
144. The most stable resonating structure of HCOOH is:
145. Which of the following mixtures can act as an acidic buffer?
1. 50 ml 0.1 M CH3COOH + 50 ml 0.1 M NaOH
2. 100 ml 0.1 M CH3COOH + 50 ml 0.1 M NaOH
3. 50 ml 0.1 M CH3COOH + 100 ml 0.1 M NaOH
4. None of the above
146. The number of lone pairs of electrons on the central atom in \(XeF^+_5 ~, ~SO_2 \) and \(XeF_4\) , respectively, are:
1. 2, 1, 2
2. 1, 1, 1
3. 1, 0, 2
4. 1, 1, 2
147. A solution that has the highest osmotic pressure among the following is:
| 1. |
0.1 M NaCl solution |
2. |
0.5 M Glucose solution |
| 3. |
0.1 M MgCl2 solution |
4. |
0.2 M KCl solution |
148. The most stable carbocation is:
149. \([Fe(H_2O)_6]^{3+}\) and \([Fe(CN)_6]^{3-} \) ions differ in:
1. Oxidation number of the central atom
2. Magnetic nature
3. Co-ordination number
4. Structure
150. 2 gm Iron Pyrite \((FeS_2) \) is burnt with \(O_2\) to form \(Fe_2O_3\) and \(SO_2\). The mass of \(SO_2\) produced is:
[Fe = 56, S=32, O=16]
1. 2 gm
2. 2.13 gm
3. 4 gm
4. 4.26 gm
Physics - Section A
151. A calorimeter contains \(270\) g of ice at \(0^\circ\)C (specific latent heat \(80\) cal/g). Steam (specific latent heat \(540\) cal/g) at \(100^\circ\)C is continuously passed through it, and the excess steam is allowed to escape. Assume negligible loss of heat to the surroundings, except due to excess steam being allowed to escape. Also, ignore the heat capacity of the calorimeter. The final mass of water in the calorimeter is:
1. \(40\) g
2. \(90\) g
3. \(310\) g
4. \(360\) g
152. The product of the angular momentum and the kinetic energy of an electron in the
\(n^\text{th}\) Bohr orbit in a hydrogen atom is proportional to:
| 1. |
\(n\) |
2. |
\(n^2\) |
| 3. |
\(\dfrac1n\) |
4. |
\(\dfrac{1}{n^3}\) |
153. Three charged particles having charges equal to
\(+1~\text C,\) \(-2~\text C\) and
\(+3~\text C\) are placed on the
\(x\text-\)axis at
\(x=1~\text{cm},\) \(2~\text{cm},\) \(3~\text{cm}\) respectively. Consider a sphere of radius
\(10~\text{cm}\) centred at the origin. The magnitude of the flux of the electric field of these charges through the sphere is, numerically:
| 1. |
slightly greater than \(\dfrac{2}{\varepsilon_0}\) |
| 2. |
slightly less than \(\dfrac{2}{\varepsilon_0}\) |
| 3. |
exactly equal to \(\dfrac{2}{\varepsilon_0}\) |
| 4. |
exactly equal to \(-\dfrac{2}{\varepsilon_0}\) |
154. Two long solenoids have the same total length and the same total number of turns, but their cross-sectional areas are different: \(A_1,A_2\) where \(A_1>A_2.\) Their self-inductances \(L_1,L_2\) are in the ratio (approximately):
1. \(A_1:A_2\)
2. \(A^2_1:A_2^2\)
3. \(A_2:A_1\)
4. \(A^2_2:A_1^2\)
155. Assume that the earth is perfectly spherical and its magnetic field is due to a dipole at its centre. The angle of dip at a magnetic latitude of \(90^\circ\) in the northern hemisphere will be equal to
1. \(0^\circ\)
2. \(45^\circ\)
3. \(60^\circ\)
4. \(90^\circ\)
156. Given below are two statements:
| Assertion (A): |
If two particles move with uniform accelerations in different directions, then their relative velocity changes in direction. |
| Reason (R): |
Since the acceleration are in different directions, there is a relative acceleration and hence the relative velocity changes. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
157. A block is placed on a smooth horizontal surface, and forces are applied to it as shown in the diagram. Take
\(g=10~\text{m/s}^2.\) The normal reaction acting on the block is:

1.
\(100~\text N\)
2.
\(60~\text N\)
3.
\(40~\text N\)
4.
\(20~\text N\)
158. A solid sphere (centre
\(O\)) of homogeneous transparent material kept upright has rays parallel to its vertical diameter falling on it. A ray that is aimed at
\(P,\) where the horizontal distance
\(OP=\dfrac{\sqrt3}{2}R\) (
\(R\) being the sphere's radius), undergoes refraction at the sphere's surface and strikes the lowest point
\(A.\)
Rays falling close to the highest point
\((B)\) of the sphere are directed towards a point
\(Q\) on the vertical diameter
\(AB,\) after refraction. The distance
\(BQ\) is:
| 1. |
\(\dfrac{R}{2}\) |
2. |
\(\dfrac{R\sqrt3}{\sqrt3 +1}\) |
| 3. |
\(\dfrac{R\sqrt3}{2}\) |
4. |
\(\dfrac{R\sqrt3}{\sqrt3 -1}\) |
159. The output
\((Y)\) of the logic circuit given above is:

1.
\(A\) AND
\(C\)
2.
\(A\) OR
\(C\)
3. always TRUE
4. always FALSE
160. One mole of an equimolar mixture of monoatomic \((He)\) and diatomic \((H_2)\) gases is heated to raise the temperature by \(1\) K under constant pressure. The amount of heat used in this process is (nearly):
1. \(8.3\) J
2. \(16.6\) J
3. \(25\) J
4. \(29\) J
161. Given below are two statements:
| Statement I: |
If the acceleration of a particle is directed towards a fixed point, and proportional to the distance from that point – the motion is SHM. |
| Statement II: |
During SHM, the kinetic energy of the particle oscillates at twice the frequency of the SHM. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
162. The circuit shown in the figure has ideal batteries wired up along with the resistances.
The current through the
\(2~\Omega\) resistance is:
1.
\(0\) A
2.
\(1\) A
3.
\(1.5\) A
4.
\(2\) A
163. Match the following quantities in
List-I with their dimensions in
List-II.
| List-I |
List-II |
| (a) |
acceleration |
(i) |
\([M^0L^0T^0 ]\) |
| (b) |
torque |
(ii) |
\([ML^{-1}T^{-2} ]\) |
| (c) |
absorptive power |
(iii) |
\([LT^{-2} ]\) |
| (d) |
pressure |
(iv) |
\([ML^2T^{-2} ]\) |
| 1. |
(a)-(iii), (b)-(iv), (c)-(i), (d)-(ii) |
| 2. |
(a)-(iii), (b)-(ii), (c)-(i), (d)-(iv) |
| 3. |
(a)-(iii), (b)-(i), (c)-(ii), (d)-(iv) |
| 4. |
(a)-(ii), (b)-(iv), (c)-(iii), (d)-(i) |
164. Given below are two statements:
| Statement I: |
If a galvanometer is connected with a high resistance in series with it, it can be used as an ammeter. |
| Statement II: |
If a galvanometer is connected with a low resistance in parallel with it, it can be used as a voltmeter. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
165. The moment of inertia of a uniform solid hemisphere, about a tangent to the circumference of the hemisphere's flat circular surface, is:
(mass of hemisphere: \(m,\) radius: \(R\))
| 1. |
\(\dfrac75mR^2\) |
2. |
\(\dfrac7{10}mR^2\) |
| 3. |
\(\dfrac15mR^2\) |
4. |
\(\dfrac65mR^2\) |
166. 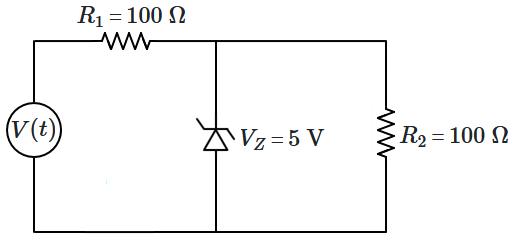
For the above circuit, the applied voltage
\(V(t)\) is periodic and linear, and is shown by the waveform below:

The current flowing through
\(R_2\) varies as:
167. Which, of the following, shows the correct graph of the de-Broglie wavelength
\((\lambda)\) of a particle and its kinetic energy
\((E_K)?\)
168. Given below are two statements:
| Assertion (A): |
Faraday's law of electromagnetic induction is not consistent with the law of conservation of energy. |
| Reason (R): |
Lenz's law is consistent with energy conservation. |
| 1. |
(A) is True but (R) is False. |
| 2. |
(A) is False but (R) is True. |
| 3. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. |
Both (A) and (R) are True but (R) is not the correct explanation of (A). |
169. An alternating emf of
\(V=20\) V
\(\mathrm{sin}(100\pi ~\text{s}^{-1}.t)\) is applied to an inductor and the peak current through the inductor is observed to be
\(\dfrac{20}{\pi}\) A. The inductance is:
| 1. |
\(\dfrac{1}{10}\) H |
2. |
\(\dfrac{1}{100}\) H |
| 3. |
\(\dfrac{1}{1000}\) H |
4. |
\(\dfrac{1}{10^4}\) H |
170. Given below are two statements:
| Statement I: |
The kinetic energy of a planet is maximum when it is closest to the sun. |
| Statement II: |
The time taken by a planet to move from the closest position (perihelion) to the farthest position (aphelion) is larger for a planet that is farther from the sun. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
171. An isotope with a mean life of \(1000\) s is produced in a reaction at a constant rate of \(R=5\times10^6/\)s. The maximum number of atoms of the isotope in the sample can be (in this case):
1. \(5\times10^3\)
2. \(5\times10^6\)
3. \(5\times10^9\)
4. \(5\times10^{12}\)
172. Three capacitors are connected in the configuration shown, with
\(C_1=C_3=C\) and
\(C_2=2C.\) If a charge
\(Q\) is passed through the circuit from
\(A\) to
\(B\) (with the capacitors initially uncharged), the energies stored in the capacitors,
\(C_1,C_2,C_3\) are in the ratio:

| 1. |
\(1:2:1\) |
2. |
\(1:\dfrac12:1\) |
| 3. |
\(1:4:1\) |
4. |
\(1:\dfrac14:1\) |
173. If a transistor has \(\beta=100,\) then the value of \(\alpha\) for the transistor is:
1. \(1.01\)
2. \(0.99\)
3. \(0.98\)
4. \(0.01\)
174. A uniform disc of mass
\(m\) and radius
\(r\) is rotating freely about its own axis, which is kept vertical. There is a small insect of mass
\(\dfrac m2\) sitting on the periphery of the disc, also rotating with the disc. The initial angular speed of the disc is
\(\omega_0.\) The insect moves radially inward and finally reaches the centre. The final angular speed of the disc is:
| 1. |
\(\omega_0\) |
2. |
\(2\omega_0\) |
| 3. |
\(\dfrac32\omega_0\) |
4. |
\(\dfrac52\omega_0\) |
175. In a Young's double-slit experimental setup,
\(240\) fringes are observed to be formed in a region of the screen when light of wavelength
\(450\) nm is used. If the wavelength of light is changed to
\(600\) nm, the number of fringes formed in the same region will be:
| 1. |
\(135\) |
2. |
\(180\) |
| 3. |
\(320\) |
4. |
\(428\) |
176. A plane electromagnetic waveform given by:
\(\vec {E}_1=E_0\hat j\sin(\omega t-kx)\)
propagates along the
\(x\)-axis. A second waveform given by:
\(\vec {E}_2=E_0\hat k\sin(\omega t-kx)\)
is also allowed to propagate. The magnetic field has the amplitude:
(Assume speed of light in vacuum is \(c\))
| 1. |
\(\dfrac{E_0}{c}\) |
2. |
\(\dfrac{E_0}{2c}\) |
| 3. |
\(\dfrac{\sqrt2E_0}{c}\) |
4. |
Zero |
177. A negative point charge
\((-q)\) is placed at the centre of a spherical ball of charge distributed uniformly over its volume. The radius of the ball is
\(R.\) The electric field on its surface is zero. The potential on its surface is:
| 1. |
zero |
2. |
\(\dfrac{kq }{2R}\) |
| 3. |
\(\dfrac{-kq }{ 2R}\) |
4. |
\(\dfrac{2kq }{ R}\) |
178. A gas
\((\gamma = 1.5)\) undergoes a process in which its volume is doubled, but the speed of sound in the gas remains unchanged. Then,
| 1. |
the pressure is halved |
| 2. |
the pressure decreases by a factor of \(2\sqrt 2\) |
| 3. |
the temperature is halved |
| 4. |
the temperature decreases by a factor of \(2 \sqrt 2\) |
179. The equation of vibration of a taut string, fixed at both ends, is given by;
\(y=(3~\text{mm})~\text{cos}\left(\dfrac{\pi x}{10~\text{cm}}\right)~\text{sin}(800\pi~\text{s}^{-1}{t}).\) The positions of the nodes are:
| 1. |
\(x= 0~\text{cm}, 10~\text{cm}, 20~\text{cm},....\) |
| 2. |
\(x= 0~\text{cm}, 20~\text{cm}, 40~\text{cm},....\) |
| 3. |
\(x= 5~\text{cm}, 10~\text{cm}, 15~\text{cm},....\) |
| 4. |
\(x= 5~\text{cm}, 15~\text{cm}, 25~\text{cm},....\) |
180. Given below are two statements:
| Statement I: |
The total energy of a particle falling freely under gravity increases with time. |
| Statement II: |
The law of conservation of mechanical energy does not apply to the motion of a projectile under gravity. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
181. The elastic energy density in a stretched wire is:
| 1. |
\(\text{(stress)}^2 × \text{strain}\) |
2. |
\(\text{stress} × \text{strain}\) |
| 3. |
\(\dfrac12\times \text{stress} × \text{strain}\) |
4. |
\(\text{stress} × \text{(strain)}^2\) |
182. The velocity-time graph of a particle moving along a straight line is shown in the adjacent figure. The average velocity is:

| 1. |
\(1~\text{ms}^{-1}\) |
2. |
\(2~\text{ms}^{-1}\) m/s |
| 3. |
less than \(1~\text{ms}^{-1}\) |
4. |
greater than \(2~\text{ms}^{-1}\) |
183. Given below are two statements:
| Statement I: |
In an elastic collision, the relative velocity of approach equals the relative velocity of separation. |
| Statement II: |
In an elastic collision, the kinetic energy before the collision is equal to the kinetic energy after the collision. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
184. The capacitance of the system of capacitors connected in the circuit, between
\(A\) and
\(B,\) equals:

1.
\(4~\mu\text{F}\)
2.
\(2.5~\mu \text{F}\)
3.
\(2.4~\mu \text{F}\)
4.
\(1.5~\mu \text{F}\)
185. If a convex lens
\((\mu=1.5)\) is immersed in water
\(\left(\mu=\dfrac{4}{3}\right)\) then its focal length in water is:
| 1. |
\(\dfrac14\) of that in air |
2. |
\(\dfrac89\) of that in air |
| 3. |
\(\dfrac98\) of that in air |
4. |
\(4\) times that in air |
Physics - Section B
186. The string and pulley are ideal, and there is no friction anywhere. If the block
\(A\) is moving down at
\(2\) m/s with the string taut, the rate at which work is done on the
\(2\) kg block
\(B\) is:
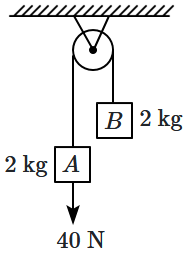
1.
\(80\) W
2.
\(40\) W
3.
\(20\) W
4.
\(0\) W
187. The resistance between
\(A\) and
\(B\) is

| 1. |
\(100~\Omega\) if \(V_A>V_B\) and \(200~\Omega\) if \(V_A<V_B\) |
| 2. |
\(100~\Omega\) if \(V_A<V_B\) and \(200~\Omega\) if \(V_A>V_B\) |
| 3. |
\(100~\Omega\) |
| 4. |
\(200~\Omega\) |
188. Select the correct option based on the statements given below:
| Statement I: |
When a projectile is at its highest point, its tangential acceleration is zero. |
| Statement II: |
When a projectile is at the highest point of its trajectory, its speed is minimum. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
189. Two blocks are joined by a light spring of spring constant,
\(k=1000\) N/m, and are placed at rest on a horizontal frictionless table. A horizontal force of
\(6\) N acts on the
\(2\) kg block. The acceleration of the centre-of-mass of the system is:

| 1. |
\(3\) m/s2 |
2. |
\(2\) m/s2 |
| 3. |
\(1.5\) m/s2 |
4. |
\(4.5\) m/s2 |
190. Consider a tank full of water with an outlet at the bottom. As the outlet valve is opened, and water flows out, the pressure inside the tank near the outlet:
| 1. |
increases. |
| 2. |
decreases. |
| 3. |
remains unchanged. |
| 4. |
increases and then quickly returns to normal. |
191. A block
\(A\) is pushed on a smooth horizontal plane by applying a horizontal force
\(F ,\) which causes an acceleration of
\({\dfrac g 4}\) (\(g\): acceleration due to gravity). The block does not topple, even though the force acts at its highest point. The normal reaction shifts forward by:

| 1. |
\({\dfrac b 2}\) |
2. |
\({ \dfrac b 4}\) |
| 3. |
\({\dfrac b 8}\) |
4. |
\(\dfrac b 3\) |
192. Photons of frequency
\(\nu\) fill a room. A metallic plate having a work function
\(W\) \((<h\nu)\) is moved with a velocity
\(v\), in this room. The maximum energy of the emitted photoelectrons: (in the plate's frame)
| 1. |
does not depend on \(v\) |
| 2. |
increases as \(v\) increases |
| 3. |
decreases as \(v\) increases |
| 4. |
first increases and then decreases as \(v\) is increased |
193. The surface tension of soapy water is
\(S.\) When bubbles are blown with soapy water, one bubble of radius
\(r\) is formed within another of radius
\(3r.\) The excess pressure within the smaller bubble, relative to the atmospheric pressure is:
| 1. |
\(\dfrac{4S}{r}\) |
2. |
\(\dfrac{8S}{3r}\) |
| 3. |
\(\dfrac{8S}{r}\) |
4. |
\(\dfrac{16S}{3r}\) |
194. The system of blocks is pulled up by force as shown in the figure. The force exerted on the
\(3\) kg block by the connecting string is:

| 1. |
\(80~\text{N}\) |
2. |
\(60~\text{N}\) |
| 3. |
\(40~\text{N}\) |
4. |
\(100~\text{N}\) |
195. A parallel beam is incident on to a lens of focal length
\(f\) (positive), parallel to its principal axis. A thin prism of vertex angle
\(A\) and refractive index
\(\mu\) is inserted in the path of the parallel beam before it falls on the lens. The focal point of the image:

| 1. |
remains unchanged |
| 2. |
is displaced along \(+y\) by \((\mu-1)Af\) |
| 3. |
is displaced along \(-y\) by \((\mu-1)Af\) |
| 4. |
is displaced along \(+x\) by \((\mu-1)Af\) |
196. In the Wheatstone Bridge arrangement shown, the resistance
\(P\) is
\(2~ \Omega.\) The bridge is initially balanced. If
P and
Q are interchanged: an amount equal to
\(S\) has to be added to
\(R\) to restore the balance. The resistance
\(Q\) equals (nearly):

1.
\(2 ~\Omega\)
2.
\(3.2 ~\Omega\)
3.
\(4 ~\Omega\)
4.
\(6.4 ~\Omega\)
197. A spring-mass system is undergoing horizontal oscillations on a frictionless surface. During the oscillation, the block picks up a particle, when it is at its extreme position. As a result of this,

| 1. |
the amplitude increases. |
| 2. |
the amplitude decreases. |
| 3. |
the frequency increases. |
| 4. |
the frequency decreases. |
198. Two long parallel wires carrying currents
\(I_1\) and
\(I_2\) give a magnetic field of
\(3\) G at a point exactly mid-way between the two wires. When one of the currents is reversed, the field becomes
\(5\) G. The ratio of the large current to the smaller one is:
| 1. |
\(2\) |
2. |
\(\dfrac43\) |
| 3. |
\(\dfrac32\) |
4. |
\(4\) |
199. A sinusoidal waveform is travelling along the \(x \)-axis. The phase difference between two particles separated by \(10~\text{cm}\) is \(\dfrac\pi{2}{}.\) The wavelength of the wave is:
1. \(20~\text{cm}\)
2. \(30~\text{cm}\)
3. \(40~\text{cm}\)
4. \(80~\text{cm}\)
200. Given below are two statements:
| Statement I: |
The efficiency of any thermodynamic engine can approach \(100\%\) if friction and all dissipative processes are reduced. |
| Statement II: |
The first law of thermodynamics is applicable only to non-living systems. |
| 1. |
Statement I is incorrect and Statement II is correct. |
| 2. |
Both Statement I and Statement II are correct. |
| 3. |
Both Statement I and Statement II are incorrect. |
| 4. |
Statement I is correct and Statement II is incorrect. |
*If above link doesn't work, please go to test link from where you got the pdf and fill OMR from there
CLICK HERE to get FREE ACCESS for 2 days of ANY NEETprep course