A plant breeder employ technique of emasculation
1. To cover the flower with a bag
2. To prevent self-pollination
3. To prevent stamen from being contaminated
4. To produce female plant
In wheat, stem sawfly unable to cause destruction of crop due to
1. Low nitrogen and sugar content
2. Hollow stem
3. Solid stem
4. Nectarless flowers
Read the following statement and select the right choice
a. Semi-dwarf varieties, Jaya and Ratna were developed in IRRI, Phillippines
b. Classical plant breeding involves crossing or hybridization of pure lines
c. Saccharum barberi was originally growth in South India
d. Genetic variability is the root of any breeding programme
1. Only a is correct
2. b and d are correct
3. a, b and c are correct
4. b, c and d are correct
In nature, a given habitat has enough resources to support a maximum possible number, beyond which no further growth is possible. The limit is known as
1. Environmental resistance
2. Intrinsic rate of natural increase
3. Carrying capacity
4. Exponentially growth
Tropical environments, unlike temperate ones, are less seasonal, relatively more constant and predictable. Such constant environment promote
1. Deforestation 2. Mass extinctions
3. Over-exploitation 4. Niche specialization
In general, loss of biodiversity in a region may lead to
| 1. | Increase in plant production |
| 2. | Decreased variability in certain ecosystem processes such as plant productivity |
| 3. | Increase in species richness |
| 4. | Lowered resistance to environmental perturbations such as drought |
Exploring molecular, genetic and species - level diversity for products of economic importance is known as
1. Endemism
2. Bioinformatics
3. Tissue culture
4. Bioprospecting
It has been recommended that storage of nuclear waste, after sufficient pre-treatment, should be done in suitably shielded containers buried within rocks about _____ deep below the earth’s surface.
1. 100 m
2. 50 m
3. 500 m
4. 200 m
Find the correct match with respect to crop variety for their disease resistant
Column I Column II
(a) Pusa Komal (i) Tobaccor mosaic virus
(b) Pusa Sadabahar (ii) Black rot
(c) Pusa Shubhra (iii) White rust
(d) Pusa Swarnim (iv) Bacterial Blight
1. a(iv), b(i), c(ii), d(iii)
2. a(iv), b(i), c(iii), d(ii)
3. a(i), b(iv), c(ii), d(iii)
4. a(ii), b(iii), c(iv), d(i)
Rate of increase in biomass by heterotrophs or consumers per unit time and area is known as
1. Gross primary productivity
2. Net primary productivity
3. Secondary productivity
4. Community productivity
The concept of Joint Forest Management was introduced by the Government of India in
1. 1974
2. 1980
3. 1987
4. 1990
Which one of the following statement is false in respect of flowering plants?
1. Parthenocarpy can be induced through the application of growth hormones
2. Integuments encircle the ovule except at the tip where a small opening called the germ pore is organized
3. Endosperm development precedes embryo development
4. Apomicts have several advantages in horticulture and agriculture
Find incorrect statement about reproduction :-
1. Type of reproduction depends upon habitat and internal physiology of organisms also
2. Enables continuity of species generation after generation
3. May be monoparental or biparental
4. Asexual reproduction never involves gametes formation
Which group of plants show clear cut vegetative, reproductive and senescent phases in their life cycle?
1. Mustard, Radish, Mango
2. Peepal, Mango, Banyan
3. Wheat, Rice, Cabbage
4. Peepal, Bryophyllum, Rice
In a 7 celled and 8 nucleate embryo sac :
1. The polar nuclei are situated below the egg apparatus in central cell
2. Egg apparatus has two synergids and one egg cell at micropylar end
3. Filiform apparatus in synergid direct the pollen tube to enter the ovule
4. Antipodal cells are haploid but have different genetic composition to that of egg cell
How many of above statements are true for statement given in the question ?
1. Two
2. Three
3. One
4. Four
To form four fully developed Polygonum type of embryosacs in angiosperms total number of meiosis and mitosis divisions generations required are :-
1. 4, 12
2. 1, 3
3. 4, 4
4. 1, 12
Choose unrelated for embryo development in flowering plants :-
1. Develops at micropyle end of ovule
2. Early stages of embryo development differs in monocots and dicots
3. In most cases zygotes divide first by transverse division
4. Most zygote divide only after certain amount of endosperm is formed
How many of the structures (A to D) in the following diagram are diploid
1. Four
2. Two
3. Three
4. One
A: All organisms have to reach a certain stage of growth and maturity in their life, before they can reproduce sexually.
B: Male and female gametes must be physically brought together to facilitate syngamy :
1. A - correct B – incorrect
2. Both A and B are correct
3. Both A and B are incorrect
4. A - incorrect B - correct
Which one of the following plant does not show dioecious condition :-
1. Marchantia
2. Papaya
3. Date palm
4. Coconut
Read the following statements :-
(A) Desert lizard lack physiological ability that mammals have to deal with high temperature of their habitat.
(B) Kangaroo rat has the ability to concentrate its urine.
1. Only (A) is correct
2. Both statements are incorrect
3. Only (B) is correct
4. Both statements are correct
In how many population interactions only one species get harmed ?
Predation, Mutualism, Commensalism, Parasitism, competition, Amensalism, Protocooperation
1. Three
2. Five
3. Seven
4. Four
How many statements are correct about decomposition ?
(A) Oxygen independent process
(B) Faster if detritus is rich in lignin and chitin
(C) Light is most important climatic factor for decomposition
(D Warm and moist environment favour decomposition
1. One
2. Two
3. Three
4. Four
Identify the correct option for diagrammatic representation of organismic response in overwhelming majority (99%) animals :-
(EL = External level) (IL = Internal level)
Which one of the following belongs to more than one trophic level ?
1. Grasshopper
2. Insectivorous plant
3. Cow
4. Lion
Which one of the following is wrong about primary succession ?
1. Lichen is pioneer community in lithosere
2. Formation of soil does not take place
3. The establishment of new biotic community is generally slow
4. With time water body is converted into land
According to Robert Constanza, the most expensive ecological service is :-
1. Purification of air
2. Soil formation
3. Nutrient cycling
4. Climate regulation
In 1981 the r-value for human population in India was
1. 0.015
2. 0.12
3. 0.205
4. 0.0205
Find odd one out about levels of biological organisation :-
1. Biome
2. Community
3. Species
4. Population
Which of the following alcoholic drinks are produced by distillation of the fermented broth:
1. Beer and wine
2. Whisky, brandy, rum
3. Beer and rum
4. Wine and brandy
Which of the following bioactive moleule is used as a blood cholesterol lowering agent :
1. Cyclosporin-A
2. Streptokinase
3. Statins
4. All the above
In mycorrhiza association the fungal symbiont helpful in :
1. Phosphorus nutrition
2. Resistance to root borne pathogen
3. Tolerance to salinity and drought
4. All the above
A major component of gobar gas is :
1. Butane
2. Ammonia
3. Methane
4. Ethane
Which of the following is not used as a biofertilizers :
1. Rhizobium
2. Fungi-Glomus
3. Cyano bacteria
4. Yeast
Virus free plants can be obtained by :-
1. Only apical meristem
2. Only axillary meristem
3. Apical and axillary meristem
4. Embryo culture
Which of the following in not the objective of bio fortification :-
1. Improvement of protein content and quality.
2. Increase oil content & quality
3. Reduction in micro nutrient and mineral content.
4. Improvement of vitamin content.
Atlas 66 is a variety of :-
1. Wheat
2. Bhindi
3. Rice
4. Sugar cane
Practical, hygienic, efficient and cost effective solution to human waste disposal is :-
1. Natural toilets
2. Eco-friendly toilets
3. Eco-san toilets
4. Biological sanitation
In which device the exhaust is passed through a spray of water or lime for removal of gases as well as particles ?
1. ESP
2. Scrubber
3. Catalytic converter
4. Both (1) and (2)
Select the correct match with respect to pollutants :-
I II
1. Itai-Itai i.Cd
2. Methaemoglobinemia ii. Hg
3. Acid Rain iii. CFC
4. Most effective green house gas iv. Stone cancer
Which phenomenon is indicated by below reaction:-
1. Acid rain
2. Photochemical smog
3. Ozone depletion
4. Sulphur smog
Which is not an example of in situ conservation:-
1. National park, wild life sanctuaries
2. Biosphere reserve
3. Sacred groves
4. Botanical gardens
A molecular technique in which DNA sequences between two oligonucleotide primers can be amplified is known as:
1. southern blotting
2. northern blotting
3. polymerase chain reaction
4. DNA replication
Agrobacterium tumefaciens is
1. a disease in humans that causes loss of sight
2. a bacterium that can be used to introduce DNA into plants
3. a fungi that is used to produce antibiotics in large amounts
4. a disease in humans that causes loss of weight
Vectors are
1. molecules that degrade nucleic acids
2. molecules that help in replication
3. molecules that are able to covalently bond to and carry foreign DNA into cells
4. molecules that protect host cells from invasion by foreign DNA
Bacterial cells protect their own DNA from degradation by restriction endonucleases by
1. methylating the DNA at the sites that the enzyme recognizes
2. deleting all recognition sites from the genome
3. not producing any restriction endonucleases
4. having anti restriction endonucleases
Which of the following enzyme is used to covalently bond foreign DNA to a vector plasmid?
1. DNA polymerase
2. Restriction endonuclease
3. DNA ligase
4. DNA helicase
The piece of equipment, that introduces DNA into cells via DNA-coated microprojectiles is known as
1. laser
2. DNA probe
3. gene gun
4. inoculating needle
How is human insulin synthesized using recombinant DNA technology?
| 1. | By using chemically synthesized DNA sequences for the two chains separately |
| 2. | By isolating DNA from the islets of Langerhans of the pancreas |
| 3. | By using cDNA for insulin |
| 4. | By using chemically synthesized DNA sequences for the complete insulin protein |
The mammary glands are modified
1. endocrine glands
2. sebaceous glands
3. vascular plexuses
4. sweat glands
Menstrual bleeding is the result of:
1. low levels of ovarian hormones
2. high levels of ovarian hormones
3. high levels of LH
4. high levels of FSH
The oral steroid contraceptive "the Pill" works by directly preventing
| 1. | ovulation | 2. | menstruation |
| 3. | fertilization | 4. | implantation |
Amenorrhea
| 1. | will cause overdevelopment of the secondary sex characteristics |
| 2. | is never normal |
| 3. | is rare in women athletes due to their good physical conditioning |
| 4. | is the absence of menstruation |
Which of the following is the correct sequence for the development and regression of an ovarian follicle?
| 1. | primordial follicle, mature vesicular ovarian follicle, corpus albicans, and corpus luteum |
| 2. | primordial follicle, mature vesicular ovarian follicle, corpus luteum, and corpus albicans |
| 3. | primordial follicle, corpus luteum, corpus albicans, and mature vesicular ovarian follicle |
| 4. | primordial follicle, corpus luteum, mature vesicular ovarian follicle, and corpus albicans |
Electroporation is:
| 1. | the process of separating charged molecules through a gel maintained in an electric field |
| 2. | the process of combining foreign DNA to an electrically charged vector molecule |
| 3. | the process of introducing DNA into cells by the application of high voltage pulses |
| 4. | the process of introducing DNA into plant cells by the application of PEG |
Which of the following was presented as evidence in favor of “the RNA world” hypothesis?
1. The fact that DNA can encode genetic information
2. The fact that enzymes, made of protein, can catalyze biological reactions
3. The discovery that some enzymes are composed of RNA rather than protein
4. The discovery of new life forms that encode their genetic information in RNA rather than DNA
The wobble effect is the:
1. Lack of precision with regard to the third base in the codon and anticodon
2. Instability of the DNA molecule when unwound
3. Instability of pairing when a purine pairs with another purine
4. Undulating movements of mRNA
About how much of the DNA in a eukaryotic cell is expressed at any one time?
1. 1 %
2. 20 %
3. 50 %
4. 80 %
In general, bacterial genes are regulated at the time of:
1. Transcription
2. Post transcription
3. Translation
4. Post translation
An operon is a:
1. Protein that suppresses gene expression
2. Protein that accelerates gene expression
3. Genes that switch other genes on or off
4. Cluster of structural genes with related functions
The activity of a repressor depends on whether:
1. The repressor is positioned next to the operon
2. The repressor is positioned next to the promoter
3. A key substance in the metabolic pathway is present
4. There is enough RNA polymerase present
A gene carried by recombinant DNA is cloned when:
1. The host bacterium divides by binary fission
2. It is transcribed
3. It is fragmented by restriction enzymes
4. It is hybridized
A piece of nucleic acid used to find a gene, by forming a hybrid with it, is called a:
1. Probe
2. Vector
3. Restriction sequence
4. Retrovirus
Restriction enzymes are synthesized by:
1. Bacteria only
2. Yeast and Bacteria only
3. Eukaryotic cells only
4. All kinds of cells
Bacterial resistance to antibiotics is a genetic trait carried in the bacterial:
1. Intron
2. Chromosome
3. Plasmid
4. Centromere
In oogenesis, when a diploid cell undergoes meiosis, how many ova result?
1. 1
2. 2
3. 3
4. 4
An organism with two different alleles is:
1. Homozygous for that trait
2. Homologous for the allele
3. Heterozygous for the trait
4. Heterologous for the allele
How many different kinds of gametes can an organism with genotype A/a; B/B; C/c produce?
1. 4
2. 8
3. 9
4. 16
The vapour pressure of acetone at is 185 torr. When 1.2 g of a non-volatile substance was dissolved in 100 g of acetone at , its vapour pressure was 183 torr. The molar mass (g/mol) of the substance is
1. 32
2. 64
3. 128
4. 488
Two faraday of electricity is passed through a solution of . The mass of copper deposited at the cathode is (at. mass of Cu=63.5 amu)
1. 0
2. 63.5 g
3. 2 g
4. 127 g
Higher order (>3) reaction are rare due to
1. low probability of simultaneous collision of all the reacting species
2. increase the entropy and activation energy is more molecules are involved
3. shifting of equilibrium towards the reactants due to elastic collision
4. loss of active species on collision
The color of is due to
1. ML charge transfer transition
2. d d transition
3. LM charge transfer transition
4. transition
Which polymer is used in manufacture of paints and lacquers?
1. Bakelite
2. Glyptal
3. Polypropene
4. Polyvinyl chloride
Which of the following compounds is not an antacid?
1. Aluminium hydroxide
2. Cimetidine
3. Phenelzine
4. Ranitidine
In a face centered cubic lattice atoms A are at the corner points and atom B at the face centered points. If atom B is missing from one of the face centered points, the formula of the ionic compound is:
1.
2.
3.
4.
The half-life period of a first order reaction is 15 minutes. The amount of substance left after one hour will be:
1. 1/4 of the original amount
2. 1/8 of the original amount
3. 1/16 of the original amount
4. 1/32 of the original amount
The standard electrode potentials of four metals A, B, C and D are - 1.2 V, 0.6 V, 0.85 V and -0.76 V, respectively. The sequence of deposition of metals on applying potential is:
1. A, C, B, D
2. B, D, C, A
3. C, B, D, A
4. D, A, B, C
An octahedral complex of is diamagnetic. The hybridisation involved in the formation of the complex is:
1.
2.
3.
4.
The correct statement about the magnetic properties of and is (Z=26):
1. both are paramagnetic.
2. both are diamagnetic.
3. is diamagnetic, is paramagnetic.
4. is paramagnetic, is diamagnetic.
Which of the following compounds is metallic and ferromagnetic?
1.
2.
3.
4.
For a linear plot of log (x/m) versus log p in Freundlich adsorption isotherm, which of the following statements is correct? (k and n are constants)
1. 1/n appears as the intercept
2. Only 1/n appears as the slope
3. log (1/n) appears as the intercept
4. Both k and 1/n appear in the slope term
Which of the following is an anionic detergent?
1. Sodium lauryl sulphate
2. Cetyltrimethylammonium bromide
3. Glyceryl oleate
4. Sodium stearate
18 g glucose is added to 178.2 g water. The vapour pressure of water (in torr) for this aqueous solution is:
1. 76.0
2. 752.4
3. 759.0
4. 7.6
The volume of atoms present in a face centred cubic unit cell of a metal is:
1.
2.
3.
4.
A hexagonal unit cell has following characteristics:
1.
2.
3.
4.
A crystalline solid has a cubic structure in which tungsten (W) atoms are located at the cubic corners of the unit cell, oxygen (O) atoms at the cube edges and sodium (Na) atom at the centre of the cube. The molecular formula of the compound is:
1.
2.
3.
4.
The molarity of a solution obtained by mixing 750 mL of 0.5 M HCl with 250 ml of 2M HCl will be:
1. 0.875 M
2. 1.0 M
3. 1.75 M
4. 0.975 M
Which of the following will have highest measure of any colligative property?
1. 1% sucrose solution
2. 1% glucose solution
3. 1% glycerol solution
4. 1% urea solution
The values for Cr, Mn, Fe and Co are -0.41, +1.57, +0.77 and +1.97V respectively. For which one of these metals the change in oxidation state from +2 and +3 is easiest?
1. Cr
2. Mn
3. Fe
4. Co
for the cell is at . The equilibrium constant for the cell reaction; is of the order of:
1.
2.
3.
4.
If the rate law of a reaction is , what would be the new rate in comparison to the original one when the volume of the vessel is reduced to half of the original?
1. 2
2. 1/8
3. 8
4. none of these
The rate of a reaction doubles when its temperature changes from 300K to 310K. Activation energy of such reaction will be:
1.
2.
3.
4.
The coagulation power of electrolytes having ions and for arsenic sulphide sol increases in the order:
1.
2.
3.
4.
From the adjacent graphs of oxide formation:
Which statement is correct?
1. can be reduced to by below
2. can be reduced to by above
3. Both (1) and (2) are correct
4. Both (1) and (2) are wrong
The method of zone refining of metal is based on the principle of:
1. greater solubility of the impurity in the molten state than in the solid
2. greater mobility of the pure metal than that of impurity
3. higher m.p. of the impurity than that of pure metal
4. greater noble character of the solid metal than that of impurity
Extra pure gas can be obtained by heating:
1.
2.
3.
4.
Which of the following is not oxidised by gas?
1.
2.
3.
4.
The values of x and y are:
1. 2, 10
2. 8, 4
3. 4, 8
4. 8, 10
In which of the following coordination compounds, does the central metal obeys the EAN rule?
1.
2.
3.
4. All of these
Which of the following is wrong?
1. and are tetrahedral
2. and are square planar
3. and are paramagnetic
4. and are paramagnetic
Geometrical shape of the complexes formed by the reaction of with and are respectively:
1. octahedral, tetrahedral, square planar
2. tetrahedral, square planar, octahedral
3. square planar, tetrahedral, octahedral
4. octahedral, square planar, tetrahedral
Which of the following is hydrolysed most easily?
1.
2.
3.
4.
How many chiral compounds are possible on monochlorination of 2-methyl butane?
1. 2
2. 4
3. 6
4. 8
Iodoform can be prepared from all except:
1. ethyl methyl ketone
2. isopropyl alcohol
3. 3-methyl-2-butanone
4. isobutyl alcohol
The compound D is
1. propanal
2. butanal
3. n-butyl alcohol
4. n-propyl alcohol
The number of organic products is:
1. 1
2. 2
3. 3
4. 4
Which of the following is most acidic?
1.
2.
3.
4.
The final product is:
1.
2.
3.
4.
Ethyl isocyanide on hydrolysis in acidic medium generates:
1. ethyl anime and methanoic acid
2. propanoic acid and ammonium salt
3. ethanoic acid and ammonium salt
4. methylamine salt and ethanoic acid
Free radical polymerization can occur in the pressure of:
1. benzoyl peroxide
2. protonic acid
3. electron donor
4. electron acceptor
oxides glucose to ...... moles of and ...... moles of .
1. 5 and 1
2. 1 and 5
3. 2 and 4
4. 4 and 2
The compound 
1. antiseptic
2. antibiotic
3. analgesic
4. pesticide
A particle of charge - q and mass m moves in a circle of radius r around an infinitely long line charge on linear charge density . Then time period will be -
1.
2.
3.
4.
The dipole moment of the given charge distribution is:
1.
2.
3.
4.
In a region of space the electric field is given by The electric flux through a surface of area of 100 units in x-y plane is:
1. 800 units
2. 300 units
3. 400 units
4. 1500 units
The variation of electric field between two points charges along the line joining the charge is given in figure. Then which is/are correct?
1.
2.
3.
4.
The materials suitable for making electromagnets should have -
1. low retentivity and high coercivity
2. low retentivity and low coercivity
3. high retentivity and low coercivity
4. high retentivity and high coercivity
The period of oscillation of a freely suspended bar magnet is 4 second. If it is cut into two equal parts lengthwise then the time period of each part will be
1. 4 sec
2. 2 sec
3. 0.5 sec
4. 0.25 sec
The mutual inductance of a pair of coils is 2H. If the current in of the coil changed from 10A to zero in 0.1s, the emf induced in the other coil is -
1. 2V
2. 20V
3. 0.2V
4. 200V
A current-carrying wire is placed between a coil in its plane, with current flowing as shown. If the current increases-
1. no current will be induced in the coil
2. an anticlockwise current will be induced in the coil
3. a clockwise current will be induced in the coil
4. the current induced in the coil will be first anticlockwise and clockwise
A small magnet is along the axis of a coil and its distance from the coil is 80 cm. In this position the flux linked with the coil is weber turns. If the coil is displaced 40 cm towards the magnet in 0.08 second, then the induced emf produced in the coil will be-
1. 0.5 mV
2. 1 mV
3. 7 mV
4. 3.5 mV
A thin semi-circular conducting ring of radius R is falling with its plane vertical in a horizontal magnetic induction (see figure). At the position MNQ the speed of the ring is v and the potential difference developed across the ring is
1. zero
2. and M is at higher potential
3. and Q is at higher potential
4. 2RBv and Q is at higher potential
Current in an ac circuit is given by then -
1. rms value of current is 5 A
2. mean value of this current in one half period will be
3. if voltage applied is then the circuit must be containing resistance and capacitance.
4. if voltage applied is , the circuit may contain resistance and inductance.
Which of the following curves correctly represents the variation of capacitive reactance with frequency n -
1.
2.
3.
4.
In the LCR series circuit, the voltmeter and ammeter readings are -
1. E=100 volts, I=2 amp
2. E=100 volts, I=5 amp
3. E=300 volts, I=2 amp
4. E=300 volts, I=5 amp
Which of the following is the correct statement for the refractive index :
1.
2.
3.
4.
What is the magnitude of momentum that is contained in a unit volume for an electromagnetic wave:
1.
2.
3.
4.
Which of the following are not electromagnetic waves?
1. Cosmic-rays
2. -rays
3. -rays
4. X-rays
In the figure shown a point object O is placed in air. A spherical boundary separates two media. AB is principal axis. Given that the radius of curved surface is 1m. The refractive index above AB is 1.6 and below AB is 2.0. The separation between the image formed due to refraction at spherical surface is -
1. 12 m
2. 20 m
3. 14 m
4. 10 m
The given lens is broken into four parts and rearranged as shown. If the initial focal length is f then after rearrangement the equivalent focal length is -
1. f
2. f/2
3. f/4
4. 4f
When light is passed through a prism, the colour which deviates least is -
1. Red
2. Violet
3. Blue
4. Green
A ray of light falls on a transparent sphere as shown in the figure. If the final ray emerges from the sphere parallel to the horizontal diameter, then calculate the refractive index of the sphere. Consider that the sphere is kept in air -
1.
2.
3.
4.
In a YDSE experiment if a slab whose refraction index can be varied is placed in front of one of the slits then the variation of resultant intensity at mid-point of the screen with will be best represented by . [Assume slits of equal width and there is no absorption by slab; the midpoint of the screen is the point where waves interfere with zero phase difference in absence of slab]
1.
2.
3.
4.
In Young's double slit experiment, the two slits are illuminated by light of wavelength and the angular separation between the fringes obtained on the screen is . If the whole apparatus is immersed in water, then the angular fringe width will be, if the refractive index of water is 4/3-
1.
2.
3.
4.
The equations of two light waves are given by:
\(y_1=6~\text{cos}(\omega t)\) & \(y_2=8~\text{cos}(\omega t+\phi).\)
What is the ratio of the maximum to the minimum intensities produced by the superposition of these waves?
1. \(49:1\)
2. \(1:49\)
3. \(1:7\)
4. \(7:1\)
A stabilized voltage 12V across a load whose current varies from 5mA to 35mA with an unregulated d.c. supply of 18V is to be obtained with a Zener diode of Zener voltage 12V and . Calculate the value of the current limiting resistance required.
1.
2.
3.
4.
The stopping potential applied between a photocathode and respective anode is such that the fastest electron can fly only one half of the distance L between cathode and anode. For the same stopping potential, the distance between cathode and anode is reduced to L/2. The fastest electron can:
1. reach the anode
2. fly a distance greater than L/4
3. fly a distance less than L/4
4. fly a distance L/4
In the circuit given below, the value of the current is -
1. 0 amp
2. 10-2 amp
3. 102 amp
4. 10-3 amp
If the stationary proton and -particle are accelerated through same potential difference, the ratio of de Broglie's wavelength will be
1. :1
2. 1:1
3. :1
4. none of these
A hydrogen atom is in an excited state of principal quantum number (n), it emits a photon of wavelength , when it returns to the ground state. The value of n is
1.
2.
3.
4.
In a hydrogen atom, the electron is in nth excited state. It may come down to second excited state by emitting ten different wavelengths. What is the value of n?
1. 6
2. 7
3. 8
4. 5
The maximum wavelength of spectral line in Lyman series in terms of Rydberg constant R is:
1. R
2. 4/3R
3. Rc
4. 1/R
The ratio of to when is maximum is :
1. at no time this is possible
2. 2
3. 1/2
4.
A radioactive nucleus \(X\) decays to a stable nucleus \(Y.\) Then, the graph of the rate of formation of \(Y\) against time \(t\) will be:
| 1. | 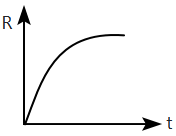 |
2. | 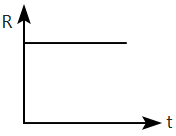 |
| 3. | 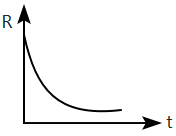 |
4. |  |
An npn transistor is biased to work as an amplifier. Which of the following statements is not correct?
1. The electrons go from the base region to collector region
2. The electrons go from emitter region to base region
3. The electrons go from the collector region to base region
4. The holes go from the base region to the emitter region
Zener diode work in -
1. zero bias
2. reverse bias
3. forward bias
4. infinite bias
By adding indium at the rate of one indium atom per germanium atoms, a sample of germanium atom is made p material. The donor density is assumed to be zero and per cubic metre at 300K. If there are germanium atoms per cubic metre, find the ratio .
1. 0.22
2. 0.51
3. 0.31
4. 0.78
The length, breadth and mass of two bar magnets are same but their magnetic moments are 3M and 2M respectively. These are joined pole to pole and are suspended by a string. When oscillated in a magnetic field of strength B, the time period obtained is 5s. If the poles of either of the magnets are reverse then the time period of the combination in the same magnetic filed will be
1.
2.
3.
4.
A bird is flying 3m above the surface of water. To a fish underwater, the height of the bird from the water surface appears to be
1. 2 m
2. 3.33 m
3. 4 m
4. (4/3) m
Which of the following represents the form of Plasmodium that is released from the erythrocyte by lysis?
1. Trophozoite
2. Schizont
3. Merozoite
4. Macrogametocyte
Dysarthria on consumption of alcohol is due to the fact that alcohol affects:
1. Cerebrum
2. Cerebellum
3. Hypothalamus
4. Medulla oblangata
Which disease has been completely eradicated?
1. Smallpox
2. Chickenpox
3. Measles
4. Mumps
Influenza viruses are classified into groups based on the _______ antigens of their protein coats.
1. A
2. B
3. C
4. N
Infection with HIV will result in a depletion of
1. CD1+ cells.
2. CD2+ cells.
3. CD3+ cells.
4. CD4+ cells.
Prion diseases share important clinical, neuropathological, and cell biological features with:
1. Kuru
2. Creutzfeldt-Jakob disease
3. Typhoid
4. Alzheimer's disease
At the time early life first formed on Earth, the atmosphere could best be described as:
1. oxidizing
2. reducing
3. rich in oxygen
4. low in hydrogen gas
Which of the following statements is TRUE?
| 1. | Prokaryotes evolved before eukaryotes. |
| 2. | Eukaryotes evolved before prokaryotes. |
| 3. | Prokaryotes and eukaryotes evolved at approximately the same time. |
| 4. | It is unclear whether prokaryotes or eukaryotes evolved first |
Reproductive isolation in sympatric speciation develops without a:
1. Geographic barrier
2. Barrier to gene flow
3. Change in chromosome number
4. Barrier to mating
Distantly related organisms with similar traits have experienced:
1. Coevolution
2. Convergent evolution
3. Divergent evolution
4. Parallel evolution
Natural selection acts on an organism's :
1. Dominant alleles
2. Recessive alleles
3. Phenotype
4. Combined genotype
Disruptive selection favors:
1. Both extreme forms of a trait
2. Intermediate forms of a trait
3. One of the extremes of a trait
4. Environmental differences
A deleterious allele decreases more rapidly in frequency if it is:
1. Recently mutated
2. Rare
3. Dominant
4. Recessive
Look at the diagram given below and answer appropriately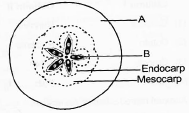
(i) Since ___A___ is associated with fruit, it is called as ___(i)___ .
(ii) Since ___B___ is present, fruit cannot be called as ___(ii)___ .
1. A - Pericarp, (i) - True fruits
2. B - Seed, (ii) - Ex-albuminous
3. A - Thalamus, (i) - True fruit
4. B - Seed, (ii) - Parthenocarpic
The extint reptile group from which mammals directly evolved is:
1. Sauropsids
2. Pelycosaur
3. Thecodonts
4. Therapsids
A typical anther shows
a. Two lobes with two theca each
b. Each lobe as dithecous
c. All cells in a given microsporangium as potential PMC
d. Thousands of microspores per microsporangium
Out of these statements :
1. b is incorrect
2. a and b are correct
3. c and d are incorrect
4. a, b, c and d are correct
Which one of the following would cause the Hardy-Weinberg principle to be inaccurate?
| 1. | The size of the population is very large. |
| 2. | Individuals mate with one another at random. |
| 3. | Natural selection is present. |
| 4. | There is no source of new copies of alleles from outside the population. |
Find correct match
| Column I | Column II |
| a. Thick and swollen | i. Castor cotyledons |
| b. Unused endosperm | ii. Legumes in mature seed |
| c. Unused nucleus in | iii. Cashew nut seed |
| d. Thalamus contributes to fruit formation | iv. Beet |
| a | b | c | d | |
| (1) | iii | iv | ii | i |
| (2) | i | iii | iv | iii |
| (3) | ii | i | iii | iv |
| (4) | ii | i | iv | iii |
Which of the following ideas was developed by Thomas Malthus?
1. Peppered moths living near English industrial cities provide a good example of evolution.
2. The diversity of Galápagos finch species is the result of natural selection.
3. Living things often produce far more offspring than are needed to maintain their numbers.
4. Reproductive isolation is pre-condition for speciation
Which of the following is not a vector borne disease?
1. African sleeping sickness
2. Dengue
3. Filariasis
4. Diptheria
Adaptive immunity depends on:
| 1. | traits common to groups of pathogens. |
| 2. | pathogen-specific recognition. |
| 3. | maternal provision of antibodies to offspring. |
| 4. | having exhausted all options for innate immunity responses. |
A patient who can produce antibodies against some bacterial pathogens, but not against viral infections, probably has a disorder in his
1. B cells.
2. plasma cells.
3. natural killer cells.
4. T cells.
Two concentric, thin metallic spheres of radii and bear changes and respectively. Then the potential at distance r between and will be
1.
2.
3.
4.
Find equivalent capacitance between X and Y if each capacitor is .
1.
2.
3.
4.
Maximum charge stored on a metal sphere of radius \(15\) cm may be \(7.5~\mu\text{C}\). The potential energy of the sphere in this case is:
1. \(9.67\) J
2. \(0.25\) J
3. \(3.25\) J
4. \(1.69\) J
Resistance of a material at temperature . Here is the temperature at . The temperature coefficient of resistance at temperature t is -
1.
2.
3.
4. constant
Three voltmeters A, B and C having resistance R, 1.5 R and 3R, respectively, are connected as shown. When some potential difference is applied between X and Y, the voltmeter readings are and respectively. Then -
1.
2.
3.
4.
An electric bulb rated for 500W at 100V is used in a circuit having a 200V supply. The resistance R that must be put in series with the bulb, so that the bulb draws 500W is -
1.
2.
3.
4.
The length of a potentiometer wire is l. A cell of emf E is balanced at a length l/3 from the positive end of the wire. If the length of the wire is increased by l/2. At what distance will be the same cell give a balance point.
1. 2l/3
2. l/2
3. l/6
4. 4l/3
A magnet of moment M is lying in a magnetic field of induction B. is the work done in turning it from to and is the work done in turning it from to . Then -
1.
2.
3.
4.