When generic name is repeated in specific name of a plant it is called:
1. Synonym
2. Autonym
3. Tautonym
4. None above
Which one of the following statements about mycoplasma is wrong?
1. They are pleomorphic
2. They are sensitive to Penicillin
3. They cause diseases in plants
4. They are called PPLO
In the following diagram A, B, C and D represent
1. A - Seta, B - Leaves, C- Rhizoids, D - Capsule
2. A - Leaf, B - Rhizoids, C- Scales, D - Root hair
3. A - Capsule, B - Seta, C- Leaves, D - Rhizoids
4. A - Capsule, B - Foot, C- Leaves, D - Rhizoids
The incorrect match for chlorophyll type is
1. Chlorophyll ‘a’ .... Green algae
2. Chlorophyll ‘d’ ... Diatoms
3. Chlorophyll ‘c’ ... Dinoflagellates and Brown algae
4. Chlorophyll ‘d’ ...Red algae
Lichens are pioneer vegetation in
1. Xerosere
2. Hydroponics
3. Marine life
4. Hydrosere
Gametangial copulation is characteristic to
1. Ascomycetes
2. Zygomycetes
3. Deuteromycetes
4. Phycomycetes
The middle central part of moss capsule is sterile and is known
1. Columella
2. Spore sac
3. Paraphyses
4. Calyptra
Protonema is a characteristic feature of
1. Fern
2. Marchantia
3. Moss
4. Cycas
In Pteridophytes reduction division occurs during
| 1. | spore formation |
| 2. | gamete formation |
| 3. | sporophyte formation |
| 4. | gametophyte germination |
Endosperm in gymnosperm is
1. Haploid
2. Diploid
3. Triploid
4. All of the above
Match items in column I with those in column II
Column I Column II
A. Coralloid roots 1. Cycas
B. Monoxylic wood 2. Mass of seeds dispersed
C. Schizogenous canals 3. Single cambium ring
D. Sulphur shower 4. Stores resin
5. Mass of pollen
A B C D
1. 1 3 5 4
2. 1 2 4 5
3. 3 4 1 5
4. 1 3 4 5
Sweet potato is a modified
1. Rhizoid
2. Stem
3. Adventitious Root
4. Leaf
The diagram given below belongs to -
1. Dicot stem having conjoint and collateral vascular bundles
2. Monocot stem having conjoint and collateral vascular bundles
3. Dicot root with polyarch condition
4. Monocot root with radial vascular bundles
Syngenecious condition means
1. anthers fused filaments free
2. both anthers and filaments fused
3. filaments fused and anthers free
4. filaments fused with carpels
Consider the following four statements A, B, C and D and select the right option for two correct statements.
(A) In vexillary aestivation, the large posterior petal is called standard, two lateral ones are wings and two small anterior petals are termed keel
(B) The floral formula for Lillaceae is
C) In pea flower the stamens are monadelphous
(D)The floral formula for Solanaceae is
The correct statements are
1. (A) and (C)
2. (A) and (B)
3. (B) and (C)
4. (C) and (D)
Placentations given below in the diagrams are
1. a - Basal, b- Axile, c- Free central, d - Parietal
2. a - Marginal, b- Axile, c- Basal, d - Superfecial
3. a - Marginal, b- Free central, c- Basal, d - Axile
4. a - Marginal, b- Axile, c- Parietal, d - Free central
What type of influrescence is found in Ficus?
1. Cyathium
2. Umbel
3. Hypanthodium
4. Racemose
Coir of coconut is obtained from
1. Endocarp
2. Mesocarp
3. Pericarp
4. Placenta
The fruit of wheat is a
1. Nut
2. Caryopsis
3. Pome
4. Cypsella
Collenchyma differs from sclerenchyma in
| 1. | retaining protoplasm at maturity |
| 2. | having thick wall |
| 3. | having crystals inside lumen |
| 4. | being dead |
The ballon like growth of parenchyma in the lumen of a vessel, is called
1. Phellogen
2. Histogen
3. Tyloses
4. Tunica
Match the items in column I with column II and choose the correct option
| Column I | Column II | ||
| (1) | Radial vascuar bundle | a. | Cucurbita pepo |
| (2) | Collateral vascular bundle | b. | Dracaena |
| (3) | Bicollateral vascular bundle | c. | Roots of angiosperms |
| (4) | Amphicribral vascular bundle | d. | Sunflower stem |
| (5) | Amphivasal vascular bundle | e. | Fern |
1. 1-c, 2-d, 3-a, 4-e, 5-b
2. 1-b, 2-c, 3-a, 4-e, 5-d
3. 1-c, 2-d, 3-e, 4-a, 5-b
4. 1-d, 2-e, 3-a, 4-b, 5-c
Hypodermis in dicot stem is composed of
1. Paraenchyma
2. Collenchyma
3. Sclerenchyma
4. Both 1. and 3.
Osmotic pressure of a solution is
1. more than that of pure solvent
2. less than that of pure solvent
3. variable depending upon concentration
4. equal to that of pure solvent
Wilting occurs, when there is
1. higher relative humidity of atmosphere
2. absorption higher than transpiration
3. transpiration higher than absorption
4. excess root pressure
Which one of the following elements in plants is not remobilized?
1. Calcium
2. Potassium
3. Zinc
4. Phosphorus
Manganese is required in
1. Nucleic acid synthesis
2. Plant cell wall formation
3. Photolysis of water during photosynthesis
4. Chlorophyll synthesis
Micronutrients are
1. less important than macronutrients
2. as important as macronutrients
3. having a minor role in plant nutrition
4. omitted from culture medium without any detrimental effect
For yielding one molecule of glucose, the Calvin cycle turns
1. two times
2. four times
3. six times
4. eight times
In C3-plants, the first stable product of photosynthesis during dark reaction is
1. PGALD
2. RuBP
3. PGA
4. OAA
The basic feature of typical C4 plants showing Kranz anatomy is the presence of
| 1. | chloroplasts in mesophyll and epidermal cells |
| 2. | chloroplasts in bundle sheath cells |
| 3. | rudimentary chloroplasts in bundle sheath cells and typical chloroplasts with well-formed grana in mesophyll cells |
| 4. | typical chloroplasts with well formed grana in bundle sheath cells and rudimentary chloroplasts in the mesophyll cells |
Which of the following is wrong in relation to photorespiration?
1. It is a characteristic of C4-plants
2. Phospho-glycolate is the initial substrate
3. It occurs in chloroplast
4. It occurs during daytime only
In glycolysis, electrons are removed by
1. ATP
2. glyceraldehydes-3-phosphate
3. NAD+
4. molecular oxygen
Which of the following is correct for the diagram given below-
| 1. | D is involved in the creation of proton gradient across the membrane |
| 2. | A is involved in the photolysis of water |
| 3. | C is also called PS II |
| 4. | F0-F1 are a part of B |
Last electron acceptor in respiration is
1. Oxygen
2. Hydrogen
3. Carbon dioxide
4. NADH
Which one of the following pairs is not correctly matched?
| 1. | Adenine derivative | — Kinetin |
| 2. | Carotenoid derivative | — ABA |
| 3. | Terpenes | — IAA |
| 4. | Indole compounds | — IBA |
Choose the correct sequence of stages of growth curve for bacteria
1. lag, log, stationary, decline phase
2. log, lag, stationary phase
3. stationary, lag, log decline phase
4. decline, lag, log phase
What is true about phytochrome?
| 1. | PR absorbs red light and becomes PFR |
| 2. | PR absorbs yellow light and becomes PF |
| 3. | PFR absorbs yellow light and becomes PR |
| 4. | PFR absorbs red light and becomes PR |
Bakanae disease is caused by
1. Virus
2. Alga
3. Bacterium
4. Fungus
Hormone that promotes growth of lateral buds and has a negative effect on apical dominance is
1. Cytokinin
2. Gibberellin
3. Auxin
4. Both 2 and 3
Pick up the correct explanation
1. Tobacco — Long-day plant
2. Spinach — Short-day plant
3. Wheat — Short-day plant
4. Tomato — Day-neutral plant
Match the following and choose the correct combination
| Column – I | Column – II | ||
| A. | Zeatin | 1. | Flowering hormone |
| B. | Florigen | 2. | Natural auxin |
| C. | Ethylene | 3. | Cytokinin |
| D. | IAA | 4. | Gaseous hormone |
| A | B | C | D | |
| 1. | 3 | 4 | 1 | 2 |
| 2. | 3 | 1 | 2 | 4 |
| 3. | 1 | 2 | 3 | 4 |
| 4. | 3 | 1 | 4 | 2 |
Match the Column I and Column II and select the correct option.
| Column – I | Column – II | ||
| A. | Auxin | 1. | Herring sperm DNA |
| B. | Cytokinin | 2. | Inhibitor of growth |
| C. | Gibberellin | 3. | Apical dominance |
| D. | Ethylene | 4. | Closure of stomata |
| E. | Abscisic acid | 5. | Induces amylase synthesis |
| A | B | C | D | E | |
| 1. | 3 | 1 | 5 | 4 | 2 |
| 2. | 3 | 1 | 5 | 2 | 4 |
| 3. | 2 | 1 | 5 | 3 | 4 |
| 4. | 4 | 5 | 1 | 3 | 2 |
Cell elongation in the internodal regions of the green plants takes place due to
1. Indole acetic acid
2. Cytokinins
3. Gibberellins
4. Ethylene
The number of correct statements amongst the given statements is:
| I. | The digestive system in Platyhelminthes is incomplete. |
| II. | Coelenterates, Ctenophores, and Echinoderm larvae are radially symmetrical. |
| III. | Bilateral symmetry is seen in annelids and arthropods but not in mollusks. |
| IV. | Notochord is a mesodermally derived structure formed on the dorsal side in some animals. |
| V. | Aschelminthes are pseudocoelomates |
1. 2
2. 3
3. 4
4. 5
The number of correct statements, regarding cockroaches, among the given statements is:
| I. | Dorsal sclerites are called sternites and ventral sclerites are called tergites. |
| II. | While the labrum is the upper lip and the labium is the lower lip, the hypopharynx acts as the tongue. |
| III. | Anal cerci are seen in both sexes but anal styles are found in male cockroaches only. |
| IV. | Each thoracic segment bears a pair of walking legs. |
| V. | Forewings called tegmina are used in flight while the hind wings are opaque, dark, and leathery. |
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
I. Amino acids are substitute methanes.
II. The R group in serine is hydroxyl-methyl.
III. Arachidonic acid has 20 carbon atoms including the carboxyl carbon.
IV. Glycerol is trihydroxypropane.
V. Lecithin is a phospholipid.
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
| I. | Yeast can progress through the cell cycle in only about 90 minutes. |
| II. | The interphase lasts more than 95% of the duration of the cell cycle. |
| III. | During G1 phase the cell is metabolically active, continuously grows and replicates its DNA. |
| IV. | Cells in G0 phase remain metabolically active bit no longer proliferate unless called to do so |
| V. | In animals, mitotic cell division is seen only in the diploid somatic cells. |
1. 2
2. 3
3. 4
4. 5
The number of incorrect statements amongst the given statements is:
I. The tongue is attached to the roof of the oral cavity by the frenulum.
II. The structural and functional units of the human liver are the hepatic lobules.
III. Lysozyme present in saliva acts as an antibacterial agent that prevents infection.
IV. Brunner’s glands are located in the submucosa of the duodenum.
V. No significant digestive activity occurs in the large intestine.
1. 1
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
I. Gills are used by most of the aquatic arthropods and mollusks.
II. Expiration takes place when intra-pulmonary pressure is higher than the atmospheric pressure.
III. Total volume of air a person can expire after normal inspiration is called as expiratory capacity.
IV. About 7 % of carbon dioxide is carried in dissolved state through plasma.
V. Every 100 ml of blood delivers approximately 4 ml of carbon dioxide to the alveoli.
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
I. Leucocytes are nucleated, lesser in number and generally short lived.
II. Rh incompatibility becomes significant when an Rh –ve mothers carries an Rh +ve fetus.
III. An enzyme complex, thrombokinase, is required for clot retraction and clot dissolution.
IV. Fats are absorbed through lymph in the lacteals present in the intestinal villi.
V. Heart is derived from the embryonic mesoderm.
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
I. Blood from the glomerulus is carried away by an afferent arteriole.
II. Vasa recta is absent or highly reduced in cortical nephrons.
III. Conditional reabsorption of sodium ions and water takes place in distal convoluted tubule.
IV. Human kidneys can produces urine nearly 5 times concentrated than the initial filtrate formed.
V. Glycosuria and ketonuria are indicative of diabetes mellitus.
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
| I. | Cardiac muscles are striated and involuntary. |
| II. | Myosin head is an active ATPase enzyme. |
| III. | The neurotransmitter released at the neuromuscular junction is acetylcholine. |
| IV. | White muscle fibers depend on anaerobic process for energy. |
| V. | Functional unit of muscle contraction is the sarcomere. |
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
I. Somatic neural system relays impulses from the CNS to smooth muscles of the body.
II. Myelinated nerve fibers are found in spinal nerves but not in cranial nerves.
III. Sodium-potassium pump transports 3 sodium outwards and 2 potassium into the cell.
IV. Electrical synapses are rare in our system.
V. Efferent neurons carry signals from the CNS to the effector.
1. 2
2. 3
3. 4
4. 5
The number of correct statements amongst the given statements is:
I. Invertebrates do not possess endocrine systems.
II. Vasopressin, synthesized by hypothalamus, is transported axonally to the adenohypophysis.
III. In males, FSH and androgens regulate spermatogenesis.
IV. Melanin is secreted by pineal and regulates diurnal rhythm.
V. Old persons have weak immune response due to degeneration of thymus in old persons.
1. 2
2. 3
3. 4
4. 5
Which of the following would be a characteristic of the animal shown in the given diagram?
| 1. | Excretory organ is the proboscis gland. |
| 2. | They migrate for spawning to fresh water. |
| 3. | Notochord is present only in the larval tail. |
| 4. | Parapodia are present for swimming. |
The type of epithelium shown in the given diagram is found in:

1. Proximal convoluted tubule
2. Later part of the small intestine
3. Bronchioles
4. Oviduct
The following diagram shows the molecular structure of:
| 1. | A hexose sugar |
| 2. | An aromatic amino acid |
| 3. | A nitrogenous base found in RNA |
| 4. | An important constituent of a DNA nucleotide. |
What stage of mitosis is shown in the diagram given below?
| 1. | Late prophase |
| 2. | Transition to metaphase |
| 3. | Anaphase |
| 4. | Early telophase |
In the given diagram of the transverse section of the gut, the longitudinal muscles are represented by the letter:
1. A
2. B
3. C
4. D
In the given diagram of a section of an alveolus with a pulmonary capillary, the basement substance would be present in the region shown by the letter:
1. A
2. B
3. C
4. D
What is true about the cell shown in the given diagram?
1. It is the rarest WBC found in the blood.
2. It is the largest WBC.
3. Its number is increased in acute bacterial infections.
4. It is involved in the transport of blood gases.
In the given diagram, the columns of Bertini are represented by the letter:
1. A
2. B
3. C
4. D
In the given diagram of the human skull, the bones represented by the letters A, B, and C are respectively:
1. Sphenoid, Ethmoid and Lacrimal
2. Ethmoid, Sphenoid and Lacrimal
3. Sphenoid, Lacrimal and Ethmoid
4. Ethmoid, Lacrimal and Sphenoid
The structure marked as X in the following diagram showing a sagittal section of the human brain would be:
1. Corpora quadrigemina
2. Limbic system
3. Corpus callosum
4. Hippocampal gyrus
In the given diagram of cochlea, the tectorial membrane is represented by the letter:
1. A
2. B
3. C
4. D
Match each item in Column I with one in Column II and choose your answer from the codes given below:
Animal Feature
A. Obelia a. Water vascular system
B. Antedon b. Air bladder
C. Pterophyllum c. Metagenesis
D. Icthyophis d. Limbless amphibian
A. B. C. D.
1. c a b d
2. c b a d
3. b a c d
4. d b a c
Match each item in Column I with one in Column II and choose your answer from the codes given below:
| Tissue | Location | ||
| (a) | Simple Squamous | (i) | Blood vessels epithelium |
| (b) | Cuboidal brush | (ii) | PCT of nephron-bordered epithelium |
| (c) | Dense irregular | (iii) | In the skin connective |
| (d) | Areolar | (iv) | Beneath the skin |
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (i) | (iii) | (iv) |
| 2. | (i) | (iii) | (ii) | (iv) |
| 3. | (i) | (ii) | (iv) | (iii) |
| 4. | (i) | (ii) | (iii) | (iv) |
Match each item in Column I with one in Column II and choose your answer from the codes given below:
| Cockroach anatomy | Features | ||
| A. | Tests | a. | 6th - 7th abdominal segments |
| B. | Mushroom gland | b. | 4th - 6th abdominal segments |
| C. | Ovaries | c. | 2nd - 6th abdominal segments |
| D. | Spermathecae | d. | 6th abdominal segment |
| A | B | C | D | |
| 1. | c | a | b | d |
| 2. | c | b | a | d |
| 3. | b | a | c | d |
| 4. | d | b | a | c |
Match each item in Column I with one in Column II and choose your answer from the codes given below:
| GI secretion | Secreted by | ||
| (a) | Lysozyme | (i) | Upper small intestinal mucosa |
| (b) | Rennin | (ii) | Stomach |
| (c) | Enterokinase | (iii) | Saliva |
| (d) | Nucleases | (iv) | Pancreas |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (ii) | (iv) |
| 2. | (iii) | (ii) | (i) | (iv) |
| 3. | (ii) | (i) | (iii) | (iv) |
| 4. | (iv) | (ii) | (i) | (iii) |
Match each item in Column I with one in Column II and choose your answer from the codes given below:
Location Partial pressure of oxygen
A. Atmospheric air a. 159 mm Hg
B. Alveoli b. 95 mm Hg
C. Deoxygenated blood c. 104 mm Hg
D. Oxygenated blood d. 40 mmHg
A. B. C. D.
1. a c b d
2. b c a d
3. a b c d
4. a c d b
Match each item in Column I with one in Column II and choose your answer from the codes given below:
A. Generation a. AV node of impulse
B. Conduction delay b. SA node
C. Lub c. Closure of semilunar valves
D. Dub d. Closure of AV valves
A. B. C. D.
1. a b c d
2. b a d c
3. a b d c
4. b a c d
Match each item in Column I with one in Column II and choose your answer from the codes given below:
Organism Excretory structure
A. Rotifers a. Protonephridia
B. Earthworms b. Nephridia
C. Cockroach c. Antennal glands
D. Prawns d. Malpighian tubules
A. B. C. D.
1. b a c d
2. a c b d
3. a b d c
4. a b c d
Match each item in Column I with one in Column II and choose your answer from the codes given below:
Type of joint Example
A. Hinge a. First carpo-metacarpal
B. Pivot b. Between the carpals
C. Gliding c. Atlanto-axial
D. Saddle d. Knee joint
A. B. C. D.
1. d c b a
2. c d b a
3. c b d a
4. d b a c
Match each item in Column I with one in Column II and choose your answer from the codes given below:
Column I Column II
A. Limbic system a. Behavior
B. Thalamus b. Sensory and motor signal coordination
C. Medulla c. Gastric secretions
D. Hypothalamus d. Body temperature
A. B. C. D.
1. b a c d
2. a c b d
3. a b d c
4. a b c d
Match each item in Column I with one in Column II and choose your answer from the codes given below:
| Column I | Column II | ||
| (a) | Estrogen | (i) | Supports pregnancy |
| (b) | Progesterone | (ii) | Secondary sexual characters in females |
| (c) | FSH | (iii) | Regulates spermatogenesis |
| (d) | LH | (iv) | Ovulation |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iii) | (iv) |
| 2. | (i) | (iii) | (ii) | (iv) |
| 3. | (i) | (ii) | (iv) | (iii) |
| 4. | (i) | (ii) | (iii) | (iv) |
All cyclostomes are ___________ on some fishes; paired fins are _________, cranium is ________ and circulation is _______ type.
| 1. | Ectoparasites, Absent, Cartilaginous, Open |
| 2. | Endoparasites, Present, Bony, Closed |
| 3. | Ectoparasites, Absent, Cartilaginous, Closed |
| 4. | Ectoparasites, Present, Cartilaginous, Open |
Bone is a _______ connective tissue, bone cells called ________ are located in spaces called ______.
1. Dense, Osteocytes, Lacunae
2. Specialized, Osteocytes, Lacunae
3. Dense, Osteoblasts, Haversian canal
4. Specialized, Osteoclasts, Haversian canal
The sequence of amino acids forms the _________ structure of the protein; the left end amino acid is ___ terminal amino acid and the right end amino acid is ____ terminal amino acid.
1. Primary, N, C
2. Primary, C, N
3. Tertiary, N, C
4. Tertiary, C, N
In telophase, _______, ________ and _________ reform.
| 1. | Chromosomes, Nucleolus, Endoplasmic reticulum |
| 2. | Mitochondria, Nucleolus, Endoplasmic reticulum |
| 3. | Mitochondria, Chromosomes, Endoplasmic reticulum |
| 4. | Golgi complex, Nucleolus, Endoplasmic reticulum |
The __________ duct and the _______ duct open together into the duodenum as the ________ duct which is guarded by _____.
1. Bile, Hepatic, Common hepto-pancreatic, Sphincter of Oddi
2. Bile, Pacreatic, Common hepto-pancreatic, Sphincter of Oddi
3. Bile, Pancreatic, Common bile, Cardiac sphincter
4. Bile, Hepatic, Common hepto-pancreatic, Lower esophageal
The thoracic chamber is formed dorsally by the ______, ventrally by the _______, laterally by the ______ and inferiorly by the ________.
1. Sternum, Vertebral Column, Ribs, Diaphragm
2. Sternum, Vertebral Column, Diaphragm, Ribs
3. Vertebral Column, Sternum, Ribs, Diaphragm
4. Vertebral Column, Sternum, Diaphragm, Ribs
Atrial systole increases ventricular filling by about ___ %, Ventricular systole leads to closure of ______ valves and ventricular diastole leads to closure of __________ valves and opening of ______ valves.
1. 30, Atrioventricular, Semilunar, Atrioventricular
2. 70, Atrioventricular, Semilunar, Atrioventricular
3. 30, Semilunar, Atrioventricular, Semilunar
4. 70, Semilunar, Atrioventricular, Semilunar
A _____ in GFR can activate ______ to release _______ which converts angiotensinogen to angiotensin I and further to angiotensin II which is a powerful _________ and restores GFR.
1. Rise, JG cells, Renin, Vasoconstrictor
2. Fall, JG cells, Renin, Vasoconstrictor
3. Fall, JGA, Renin, Vasodilator
4. Rise, JGA, Renin, Vasodilator
During skeletal muscle contraction, increase in calcium levels leads to binding of calcium to a subunit of _______ on ______ filaments and thereby removes the masking of active sites for ______.
1. Troponin, Tropomysin, Myosin
2. Calmodulin, Actin, Myosin
3. Troponin, Actin, Myosin
4. Calmodulin, Actin, Myosin
The inner layer of the eyeball, the retina, contains three layers of cells – from inside to outside - ______ cells, ______ cells and ______ cells.
1. Photoreceptor, Bipolar, Ganglion
2. Bipolar, Photoreceptor, Ganglion
3. Ganglion, Photoreceptor, Bipolar
4. Ganglion, Bipolar, Photoreceptor
The adrenal cortex secretes hormones which are chemically ______ and while ______ is the main glucocorticoid, ______ is the main mineralocorticoid.
1. Peptides, Cortisol, Aldosterone
2. Steroids, Cortisol, Aldosterone
3. Peptides, Aldosterone, Cortisol
4. Steroids, Aldosterone, Cortisol
Consider the following two statements:
I. Hormones which interact with intracellular receptors mostly regulate gene expression or chromosome function.
II. Steroid hormones and iodothyronines interact with intracellular receptors.
1. Both I and II are correct and II explains I
2. Both I and II are correct but II does not explain I
3. I is correct but II is incorrect
4. I is incorrect but II is correct
Consider the following two statements:
I. Co-factors play a crucial role in the catalytic activity of the enzyme.
II. If co-factor is removed from the enzyme, the catalytic activity of the enzyme is slightly reduced.
1. Both I and II are correct and II explains I
2. Both I and II are correct but II does not explain I
3. I is correct but II is incorrect
4. I is incorrect but II is correct
Which of the following pairs of substances illustrate the law of multiple proportions
1.
2.
3.
4.
Boron has two stable isotopes, (19%) and (81%). The atomic mass that should appear for boron in the periodic table is
1. 10.8
2. 10.2
3. 11.2
4. 10.0
Molarity of liquid HCl with density equal to 1.17 g/cc is
1. 36.5
2. 18.25
3. 32.05
4. 4.65
Hydrochloric acid solutions A and B have concentration of 0.5 N and 0.1 N respectively. The volume of solutions A and B required to make 2 litres of 0.2 N hydrochloric are
1. 0.5 I of A + 1.5 l of B
2. 1.5 l of A + 0.5 l of B
3. 1.0 l of A + 1.0 l of B
4. 0.75 l of A + 1.25 l of B
Densities of two gases are in the ratio 1 : 2 and their temperatures are in the ratio 2 : 1, then the ratio of their respective pressures is
1. 1 : 1
2. 1 : 2
3. 2 : 1
4. 4 : 1
If 4 g of oxygen diffuse through a very narrow hole, how much hydrogen would have diffused under identical conditions
1. 16 g
2. 1 g
3. 1/4 g
4. 64 g
The Vander Waal’s equation reduces itself to the ideal gas equation at
1. High pressure and low temperature
2. Low pressure and low temperature
3. Low pressure and high temperature
4. High pressure and high temperature
Favourable conditions for electrovalency are
1. Low charge on ions, large cation, small anion
2. High charge on ions, small cation, large anion
3. High charge on ions, large cation, small anion
4. Low charge on ions, small cation, large anion
Which of the following statement is not correct ?
1. Hybridization is the mixing of atomic orbitals prior to their combining into molecular orbitals
2. hybrid orbitals are formed from two p atomic orbitals and one s atomic orbital
3. hybrid orbitals are directed towards the corners of a regular octahedron
4. hybrid orbitals are all at 90o to one another
The bond order is maximum in
In the reaction , if 2 moles of A, 3.0 moles of B and 2.0 moles of C are placed in a 2.0l flask and the equilibrium concentration of C is 0.5 mole/l. The equilibrium constant () for the reaction is
1. 0.073
2. 0.147
3. 0.05
4. 0.026
A solution of in water acts as acidic due to
1. Hydrolysis of
2. Acidic impurities
3. Dissociation
4. Ionisation
Which is the correct representation of the solubility product constant of
1.
2.
3.
4.
At 25°C, the dissociation constant of a base BOH is . The concentration of Hydroxyl ions in 0.01 M aqueous solution of the base would be
1.
2.
3.
4.
Which equilibrium can be described as an acid-base reaction using the Lewis acid-base definition but not using the Bronsted-Lowry definition
1.
2.
3.
4.
50mL of 2.0 M (molar mass = 158g mol–1) is dilluted to 500mL. Molarity of the resulting solution and the number of moles of are :
1. 2.0 M and 2.0
2. 0.1 M and 0.2
3. 0.2 M and 0.1
4. 0.1 M and 0.1
0.63 g of a diabasic acid is available in 100 mL solution. 20 mL of this solution required 20 mL of M/10 NaOH solution for complete neutralization. The molar mass of the acid is
1. 63 g
2. 126 g
3. 31.5 g
4. 94.5 g
50 mL of 0.2 M NaOH is mixed with 25 mL of 0.2 M . The resulting solution is :
1. acidic, 0.2 M
2. basic, 0.2 M
3. basic, 0.4 M
4. neutral
A fixed mass of a gas is subjected to transformation of states from K to L to M to N and back to K as shown
The successive operation that enable this transformation of states are:
1. heating, cooling, heating, cooling
2. cooling, heating, cooling, heating
3. heating, cooling, cooling, heating
4. cooling, heating, heating, cooling
Equal masses of , He and gasses mixed in vessel recorded a pressure of 7.5 atm. The partial pressure of is:
1. 4.8 atm
2. 2.4 atm
3. 0.3 atm
4. 7.5 atm
In the van der Waals, equation of states about gas law, the constant ‘b’ is a measure of:
1. intermolecular collision per unit volume
2. intermolecular attraction
3. volume occupied by the molecules
4. intermolecular repulsions
The number of radial nodes of 3s and 2p orbitals are respectively:
1. 2, 0
2. 0, 2
3. 1, 2
4. 2, 1
The electrons identified by quantum numbers n and l
(1) n = 4, l = 1
(2) n = 4, l = 0
(3) n = 3, l = 2
(4) n = 3, l = 1
1. 4 < 2 < 3 < 1
2. 2 < 4 < 1 < 3
3. 1 < 3 < 2 < 4
4. 3 < 4 < 2 < 1
Correct order of electron affinities of halogen is:
1. F > Cl > Br > I
2. Cl > Br > I > F
3. Cl > F > Br > I
4. Br > F > Cl > I
The increasing order of the ionic radii of the given isoelectronic species is:
1.
2.
3.
4.
Which of the following has shortest bond length?
1.
2.
3.
4.
has dipole moment x Debye. Which of the following is correctly matched for its dipole moment?
1.
2.
3.
4.
If the heats of neutralisation of HCl and HCN with NaOH are – 56 kJ and –12 kJ respectively, the enthalpy of ionisation of HCN is:
1. – 44 kJ
2. 44 kJ
3. 78 kJ
4. – 78 kJ
The bond energies of C – C, C = C, H – H and C – H linkages are 350, 600, 400 and 410 kJ mol–1 respectively. The heat of hydorgenation of ethylene is
1. – 170 kJ mol–1
2. – 260 kJ mol–1
3. – 400 kJ mol–1
4. – 450 kJ mol–1
Ammonium carbamate is in equilibrium with and as:
If total pressure at equilibrium is 3 atm, is:
1. 4
2. 12
3. 36
4. 27
of a weak acid HA which is 0.01% ionised is . Its pH is
1. 1
2. 2
3. 4
4. 5
At 25°C, the solubility product of is . At which pH, will ion start precipitating in the form of , from a solution of 0.001 M ions?
1. 8
2. 9
3. 10
4. 11
As per the structure of , the two Cl-atoms have oxidation numbers:
1. 0, 0
2. 0, –1
3. + 1, – 1
4. 0, +1
oxidation product of X. X cannot be:
1.
2.
3.
4.
The oxidation states of the most electronegative element in the products of the reaction of and ice cold dilute are
1. 0, 0
2. 0, – 1
3. – 1, – 2
4. 0, – 2
Which of the following does not exist as solid?
1.
2.
3.
4.
pH of aqueous solution of is more than 7 because of the formation of ions due to
1. hydrolysis of
2. hydrolysis of
3. hydrolysis of both and
4. hydrolysis of
The correct acidic character shown by boron halides is
1.
2.
3.
4.
Graphite is a soft solid lubricant, extremely difficult to melt. The reason for this anomalous behaviour is the graphite:
1. has molecules of variable molecular masses, like polymers
2. is an allotropic form of diomond
3. has carbon atoms arranged in large plates of rings of strongly bound carbon atoms with weak interplanar bonds
4. is a non-crystalline substance
Which of the following amines is the most basic
1.
2.
3.
4.
Which of the following is the most stable carbocation?
1.
2.
3.
4.
The non-aromatic compound among the following is:
1.
2.
3.
4.
Which of the following does not show tautomerism?
1.
2.
3.
4. All of these
When phenyl magnesium bromide reacts with t-butanol, the product would be:
1. benzene
2. phenol
3. t-butyle benzene
4. t-butyle phenyl ether
In the following reaction:
The end product is
1. m-bromotoluene
2. o-bromotoluene
3. 3-bromo-2, 4, 6-trichlorotoluene
4. p-bromotoluene
Displacement of a particle is given by , where t denotes the time. The unit of k is
1. hertz
2. metre
3. radian
4. second
The percentage errors in a measurement of mass and speed are 2% and 3% respectively. What will be the maximum error in the measurement of kinetic energy?
1. 11%
2. 5%
3. 8%
4. 4%
A unit vector is represented by , then value of c is
1.
2.
3.
4.
The velocity-time relation of an electron starting from rest is given by where . The distance traveled in 3 seconds is
1. 9 m
2. 27 m
3. 36 m
4. 16 m
Two particles P and Q are moving with same acceleration at time , velocity of particles are and respectively. When velocity of P is the velocity of Q is
1.
2.
3.
4.
The maximum height reached by the projectile is 4 metres and the horizontal range is 12 metres. The velocity of projection in is:
(\(g\) is the acceleration due to gravity)
1.
2.
3.
4.
The tangential component of acceleration of a particle in circular motion is due to:
| 1. | speed of the particle |
| 2. | change in the direction of the velocity |
| 3. | change in the magnitude of the velocity |
| 4. | rate of change of acceleration |
Traffic is moving at 60 km/hr along a circular track of radius 0.2 km, the correct angle of banking is
1.
2.
3.
4.
A massless, frictionless rope is attached at point \(A\) on the wall, then passes under a movable pulley \(P_2,\) goes up and over a fixed ceiling pulley \(P_1,\) and supports a hanging weight \(W_1.\) The movable pulley \(P_2\) carries a second weight \(W_2\) hanging directly beneath it. If \(W_1=W_2,\) what is the angle \((AP_2P_1)\) between the two rope segments meeting at \(P_2\text{?}\)
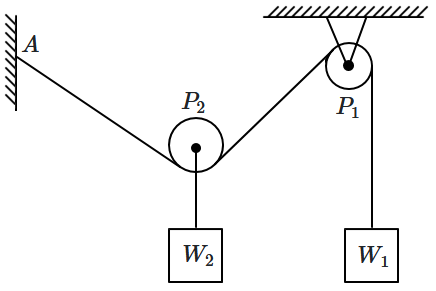
1. \(30^\circ\)
2. \(60^\circ\)
3. \(150^\circ\)
4. \(120^\circ\)
A block of mass 2 kg is placed on the floor. The co-efficient of static friction is 0.4. A force of 2.8 N is applied on the block as shown in figure. The force of friction between the block and the floor is
1. 10 N
2. 8 N
3. 16 N
4. 2.8 N
A body is falling under gravity from rest. It loses 200 J of gravitational potential energy when its speed is Mass of the body is:
1. 3 kg
2. 4 kg
3. 5 kg
4. 7 kg
A body starts from rest and acquire a velocity V in time T. The work done on the body in time t will be proportional to
1.
2.
3.
4.
Ball A collide with another identical ball B at rest. For what value of coefficient of restitution e, the velocity of second ball becomes twice that of A after one collision?
1.
2.
3.
4.
A particle of mass m moving with a velocity v strikes a stationary particle of mass 2 m and sticks to it. The speed of the system will be
1.
2. 2 v
3.
4. 3 v
The values of the acceleration on the surfaces of two planets are equal, the planets must have same
1. Mass
2. Radius
3.
4.
A body is projected up with a velocity equal to escape velocity of earth at angle of 60° with the horizontal, then
1. Body comes back to the surface
2. Body first go up than returns back after 5 second
3. Never come back
4. Depends upon mass of the body
Two blocks placed on an inclined plane having unequal mass are connected by thread. Initially the thread is loose as shown in the figure. When the block are released
1. Thread will certainlly remain loose if
2. Thread will remain loose if is smooth and is rough
3. Thread will remain loose of is rough and is smooth
4. Depends upon angle of inclination
Infinite number of masses each of 1.5 kg are placed along a straight line at distance of 1 m, 2 m, 4 m, 8 m ..... from a point O on the same line. What is the magnitude of gravitational field at O?
1. 0.5 G
2. 1.0 G
3. 1.5 G
4. 2 G
A bullet is fired on a wooden plank. In penetrating through the plank it loses 10% of its K.E. How many plank are required just to stop the bullet?
1. 20
2. 15
3. 10
4. 5
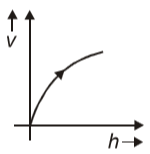
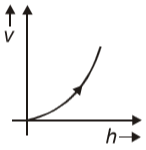
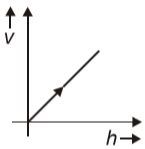
A ball is dropped from a certain height, the graph between its velocity and height h will be
1. 
2. 
3. 
4.
Moment of Inertia is a
1. Scalar
2. Vector
3. Tensor
4. Both 1. & 2.
Steel is preferred for making springs over copper because (Y = Young’s modulus)
1. Steel is cheaper
2.
3.
4. Steel is less likely to be oxidised
When a load of 5 kg is hung on a wire, then extension of 3 m take place, then work done will be
1. 7.5 J
2. 50 J
3. 100 J
4. 60 J
When a tube is dipped in liquid, the angle of contact is 79°. When tube is dipped at angle of 60° with the vertical then angle of contact will be
1. 60°
2. 79°
3. 54°
4. 120°
The surface tension of pure water is 0.061 N/m, the rise of water in the capillary of diameter 1 mm is (cos = 1)
1. 1.22 cm
2. 2.44 cm
3. 3.12 cm
4. 3.86 cm
Weight of a body is 8 N in the air and 3 N in the water. The buoyant force acting on the body in water is
1. 11 N
2. 3 N
3. 8 N
4. 5 N
There is a hole of area A at the bottom of cylindrical vessel, water is filled up to height h and water flow out in t second. If water is filled to a height 4h, water will flow out in time equal to
1. t
2.
3. 2t
4. 4t
At what temperature the molecules of nitrogen will have the same r.m.s speed as molecules of oxygen at 127°C?
1. 77°C
2. 350°C
3. 273°C
4. 457°C
Which graph represents behavior of ideal gas at constant temperature?
1. 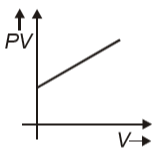
2. 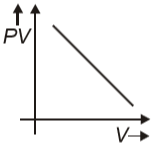
3. 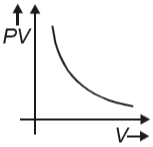
4.
| 1. | \(\dfrac{3}{2} kT\) | 2. | \(\dfrac{kT}{2}\) |
| 3. | \(\dfrac{3}{4} kT\) | 4. | \(kT\) |
Water flows through a frictionless tube with varying cross-section as shown in figure. The variation of pressure is best represented by
1. 
2. 
3. 
4.
The temperature of substance increases by 27°C this increases is equal to (in kelvin)
1. 300 K
2. 2.46 K
3. 27 K
4. 7 K
An anisotropic material has co-efficient of linear expansion 1unit, 3 unit and 2 unit along x, y and z respectively. Co-efficient of cubical expansion equal to
1. 4 unit
2. 2 unit
3. 6 unit
4. 7 unit
The speed of sound in a medium is \(v.\) If the density of the medium is doubled at constant pressure, what will be the new speed of sound?
| 1. | \(\sqrt{2} v \) | 2. | \(v \) |
| 3. | \(\frac{v}{\sqrt{2}} \) | 4. | \(2v\) |
Two sound sources when sounded simultaneously produce four beats in 0.50 second. The difference in their frequencies must be
1. 8
2. 4
3. 16
4. 1
The vibrations of four air columns are represented in the figure. The ratio of frequencies is
1. 12 : 6 : 3 : 4
2. 1 : 2 : 4 : 3
3. 4 : 2 : 3 : 1
4. 6 : 2 : 3 : 4
A light spring is compressed and placed horizontally between a vertical fixed wall and a toy car which is free to slide over a smooth horizontal table. If the system is released from rest, which graph best represents acceleration \(a\) and distance \(x\) covered by the car?

| 1. |  |
2. |  |
| 3. |  |
4. |  |
What is the angular frequency of the system? (as shown in figure)
1.
2.
3.
4.
At what point normal reaction will be maximum?
1. A
2. B
3. C
4. At both point A and B
When a source is going away from a stationary observer with the speed equal to that of speed of sound in air, then the frequency heard by the observer will be
1. Same
2. Double
3. Halved
4. One-third
A body executing SHM has time period T. The body complete oscillation by distance, starting from mean position in the time
1.
2.
3.
4.
If temperature of a black body increases from 7°C to 287°C, then the rate of energy radiation become
1.
2. 16 times
3. 4 times
4. 2 times
100 gm of ice at 0°C is mixed with 100 gm of water at 100°C. What will be the final temperature of the mixture?
1. 10°C
2. 20°C
3. 30°C
4. 40°C
A gas expands at constant pressure of , the work done is
1. 2.5 ergs
2. 250 J
3. 250 W
4. 250 N
Two bodies A and B have moment of interia and respectively, if both have same angular momenta, then their rotational kinetic energies are in the ratio
1.
2.
3.
4.
Common cold is not cured by antibiotics because it is
1. caused by a Gram-negaive bacterium
2. not an infectious disease
3. viral disease
4. caused by a Gram-positive bacterium