Which part of the human GIT has an innermost oblique muscle layer in its wall?
1. Stomach
2. Duodenum
3. Ileum
4. Rectum
The layer in the wall of the alimentary canal responsible for peristalsis, the basic propulsive movement in the gut, would be:
1. Mucosa
2. Submucosa
3. Muscularis
4. Serosa
The common bile duct is formed by the joining of:
1. Right and left hepatic ducts
2. Common hepatic duct and cystic duct
3. Common hepatic duct and the pancreatic duct
4. Stenson’s duct and Wharton’s duct
Gastric secretions do not include:
1. Pepsinogen
2. HCl
3. Lipase
4. Nuclease
Fructose is absorbed in the small intestine by:
1. Simple diffusion
2. Facilitated diffusion
3. Active transport
4. Active co-transport with sodium
The amount of air that is left in the lungs after maximum exhalation is called as:
1. Expiratory reserve volume
2. Functional residual capacity
3. Expiratory capacity
4. Residual volume
The partial pressure of oxygen in systemic circulation [oxygenated blood pumped by aorta] is about:
1. 104 mm Hg
2. 100 mm Hg
3. 95 mm Hg
4. 40 mm Hg
The oxy-hemoglobin dissociation curve will shift to right in all the following conditions except:
1. Increased pH
2. Increased carbon dioxide
3. Increased temperature
4. Presence of 2,3 DPG
The maximum amount of carbon dioxide in the blood is transported as:
1. Carbaminohemoglobin
2. Carboxyhemoglobin
3. Bicarbonate
4. Dissolved state in plasma
The respiratory rhythm center in humans in present in:
1. Pons varolii
2. Aortic and carotid bodies
3. Apex of the lungs
4. Medulla oblongata
The vitamin that plays an important role in erythropoiesis is:
1. Vitamin A
2. Vitamin B 12
3. Vitamin C
4. Vitamin K
Which of the following is not an anticoagulant?
1. Heparin
2. Warfarin
3. Oxalates and citrates
4. Plasmin
The chamber of the human heart with thickest musculature is the:
1. Right atrium
2. Right ventricle
3. Left atrium
4. Left ventricle
A ‘conduction delay’ in the human heart is associated with:
1. SA node
2. AV node
3. Bundle of His
4. Purkinje fibers
During the cardiac cycle, the AV vales close during:
1. Early ventricular systole
2. Late ventricular systole
3. Early ventricular diastole
4. Late ventricular diastole
A chronic condition in which the heart doesn’t pump blood as well as it should is called:
1. Angina pectoris
2. Heart attack
3. Heart failure
4. Heart block
Which of the following is not a function of the human kidney?
1. Osmoregulation
2. Regulation of acid base balance
3. Production of nitrogenous waste products
4. Regulation of erythropoiesis
The part of the nephron that is responsible for maximum reabsorption of the filtrate is:
1. Proximal convoluted tubule
2. Loop of Henle
3. Distal convoluted tubule
4. Collecting duct
The part of the nephron that plays most important role in the concentration of urine is:
1. Proximal convoluted tubule
2. Thin segment of ascending limb of loop of Henle
3. Thick segment of ascending limb of loop of Henle
4. Collecting duct
The mictuirition reflex is coordinated at the level of:
1. Pontine center
2. Sacral spinal cord
3. Medulla oblongata
4. Hypothalamus
A person having glycosuria and ketonuria is most likely suffering from:
1. Coronary artery disease
2. Chronic renal failure
3. Diabetes mellitus
4. Acute viral hepatitis
Angiotensinogen is synthesized by:
1. Liver
2. Macula densa
3. Juxta glomerular cells
4. Glial cells
What masks the active sites for myosin head present on the actin myofilaments?
1. Light meromyosin
2. Heavy meromyosin
3. G – actin
4. Troponin
A motor unit consists of:
1. A motor neuron and all the muscle fibers innervated by it
2. All the motor neurons innervating one muscle fiber
3. All the muscle fibers in a fasciculus
4. All the muscle fibers in a skeletal muscle
The muscle can directly use which of the following as a source of energy?
1. Creatine phosphate
2. Glucose
3. ATP
4. Fatty acids
With respect to the red muscle fiber, the white muscle fiber has higher:
1. Density of capillary network
2. Number of mitochondria
3. Amount of sarcoplasmic reticulum
4. Glycogen reserve
Myasthenia gravis is an autoimmune disease in which antibodies are formed against:
1. Myelin sheath
2. Articular cartilage
3. Acetylcholine receptor
4. Thyroid follicle
A glenoid cavity is seen in:
| 1. | Scapula | 2. | Coxa |
| 3. | Humerus | 4. | Clavicle |
The first carpometacarpal joint is a/an:
1. Saddle joint
2. Ellipsoidal joint
3. Pivot joint
4. Fibrous joint
Osteoporosis, in a post-menopausal woman, is most directly due to the deficiency of:
1. Progesterone
2. Estrogen
3. LH
4. FSH
In the resting stage, the axonal membrane:
1. is more permeable to sodium than to potassium ions
2. is more permeable to potassium than to sodium ions
3. is equally permeable to both sodium and potassium ions
4. is impermeable to both sodium and potassium ions
In a chemical synapse what activates the synaptic vesicles to release its neurotransmitter into the synaptic cleft?
1. Sodium
2. Potassium
3. Chloride
4. Calcium
Damage to which of the following will result in ataxia and dysarthria?
1. Medulla oblongata
2. Cerebellum
3. Mid brain
4. Hypothalamus
Sympathetic stimulation would lead to all of the following except:
1. Dilation of pupil
2. Bronchoconstriction
3. Increased heart rate
4. Decreased gland secretions
Which layer of human retina stores Vitamin A?
1. Pigment layer
2. Rods
3. Layer of bipolar cells
4. Inner plexiform layer
The biomolecule that cannot act as a chemical messenger [hormone] is:
1. Proteins
2. Steroids
3. Fatty acids
4. Carbohydrate
Which of the following is a storage and release center for neurohormones?
1. Anterior pituitary
2. Posterior pituitary
3. Adrenal medulla
4. Pineal
The hormone melatonin is a derivative of:
1. Cholesterol
2. Tyrosine
3. Tryptophan
4. Niacin
Find the incorrect match:
1. Conn’s syndrome: Hypoaldosteronism
2. Cushing’s disease: Hypercortisolism
3. Hashimoto’s disease: Hypothyroidism
4. Osteitisfibrosacystica: Hyperparathyroidism
Modified fatty acids, prostaglandins, act as localized hormones that regulate neighbouring cells. Such a regulation is called as:
1. endocrine
2. paracrine
3. eccrine
4. glycoprotein
Which hormones stimulate the production of pancreatic juice and bicarbonate?
1. angiotensin and epinephrine
2. gastrin and insulin
3. cholecystokinin and secretin
4. insulin and glucagon
All the following regarding the regulation of a physiological function by either a neurotransmitter or a hormone are true except:
| 1. | Target cells must have specific receptor proteins with which these molecules combine. |
| 2. | When these molecules bind to target cells, specific sequences of changes must occur. |
| 3. | There must be an “off-switch” that will stop the induced changes. |
| 4. | Second messengers will always be involved in the response of the target cell. |
Which digestive organ mechanically and chemically transforms a food bolus into chyme?
1. esophagus
2. stomach
3. small intestine
4. large intestine
From deep to superficial, what are the tunics of the intraperitoneal portions of the GI tract?
1. serosa, muscularis, submucosa, and mucosa
2. mucosa, submucosa, muscularis, and serosa
3. adventitia, muscularis, submucosa, and mucosa
4. mucosa, submucosa, muscularis, and adventitia
Which selection includes only accessory digestive organs?
1. salivary glands, thyroid gland, pancreas, liver
2. stomach, duodenum, pancreas, gallbladder
3. gallbladder, liver, pancreas, salivary glands
4. liver, thyroid gland, gallbladder, spleen
In prematurely born infants, hyaline membrane disease is associated with inadequate production of _________ by _________ cells.
1. lysozyme; mucous gland
2. mucin; goblet
3. glycoproteins; alveolar type I
4. surfactant; alveolar type II
Which of the following result from the presence of free air in the pleural cavity?
1. pneumothorax and atelectasis
2. hydrothorax and hemothorax
3. empyema and emphysema
4. all of the above
Asthma is most similar to ____________ localized in the bronchi and bronchioles.
1. an immune response
2. a pulmonary embolism
3. acute bronchitis
4. pneumonia
The epithelium lining the alveolus is composed of
1. a simple squamous epithelium.
2. a pseudostratified ciliated columnar epithelium.
3. a simple cuboidal epithelium.
4. a transitional epithelium.
Which of the following factors favors a release of oxygen from hemoglobin?
1. decreased
2. decreased pH
3. lower blood temperature
4. increased pH
The cells that produce the smallest formed elements of the blood are called
1. erythroblasts
2. megakaryocytes
3. myeloblasts
4. thrombocytes
Persons with Rh ____________ blood never exhibit Rh ____________.
1. negative; incompatibility
2. negative; antibodies
3. positive; surface antigens
4. positive; antibodies
In an ECG tracing, what might an unusually strong QRS complex indicate?
1. damage to the sinoatrial node
2. acute coronary ischemia
3. hypertrophy of the ventricular myocardium
4. damage to the AV bundle or Purkinje fibers
What happens during ventricular diastole?
1. both ventricles fill with blood
2. both semilunar valves remain open
3. both AV valves remain closed
4. both ventricles contract
Which structures prevent the AV valves from everting and flipping into the atria when the ventricles contract?
1. chordae tendineae and pectinate muscles
2. chordae tendineae and papillary muscles
3. conus arteriosus and interventricular septum
4. foramen ovale and fossa ovalis
What is the functional filtration unit in the kidney?
1. renal tubule
2. renal corpuscle
3. nephron
4. glomerulus
A blockage in a glomerulus would directly obstruct blood flow into which vessel?
1. afferent arteriole
2. efferent arteriole
3. interlobular artery
4. interlobar artery
What three physical barriers must filtrate cross within the renal corpuscle?
1. podocytes, pedicels, and filtration slits
2. macula densa, vascular pole, and parietal layer of glomerular capsule
3. glomerular endothelium, basement membrane, and podocytes
4. proximal convoluted tubules, connecting tubules, and papillary ducts
Which statement does not correctly describe the path of urine flow?
1. ducts in each renal papilla drain into a minor calyx
2. the minor calyces merge to form a major calyx
3. the major calyces combine to form the renal pelvis
4. the renal pelvis conveys urine directly to the urethra
The direct consequence of inadequate antidiuretic hormone (ADH) secretion would most likely be
1. dehydration
2. incontinence
3. dysuria
4. urolithiasis
Ribs 11 and 12 are called floating ribs because they
1. do not articulate with the vertebrae
2. attach only indirectly to the sternum
3. have no connection to the sternum
4. do not articulate directly with any other bone
Fill in the blanks with appropriate letters from the given diagram and choose the correct answer from the options given: ____ pumps oxygenated blood into ____ and ______ prevents its backflow into ______.
1. m, j, d, f
2. f, j, d, f
3. m, h, n, m
4. f, h, d, m
Which part of the specialized conducting system of the human heart is auto-excitable?
| I. | SA Node | II. | AV node |
| III. | AV bundle | IV. | Purkinje fibers |
1. I and II only
2. I, II and III only
3. II, III and IV only
4. I, II, III and IV
Which among the following is correct during each cardiac cycle?
1. The volume of blood pumped out by the Rt and Lt ventricles is same.
2. The volume of blood pumped out by the Rt and Lt ventricles is different
3. The volume of blood received by each atrium is different
4. The volume of blood received by the aorta and pulmonary artery is different
If due to some injury the chordae tendinae of the bicuspid valve of the human heart is partially non-functional, what would be the immediate effect?
1. Heart beat would become irregular
2. Blood will tend to move back into the right atrium.
3. The flow of blood into the aorta will be slowed down.
4. The flow of blood into pulmonary artery will be reduced.
What is the state of heart in the interval after end of second heart sound and before beginning of first heart sound again?
1. Ventricle remain systole auricles undergo first diastole and the systole
2. Ventricle remain in diastole, auricles undergoes first diastole and then systole
3. Ventricles undergo first systole and then diastole, auricles remain in diastole.
4. Ventricles remain in diastole, auricles remain in systole.
Which of the following statements is correct for cardiac cycle?
1. The ventricular systole is stimulated by the SA node.
2. The first heart sound coincides with the closure of semilunar valves and relaxation of ventricles.
3. The blood flows from auricles to ventricles during the atrial systole via bicuspid and tricuspid valves.
4. The fall in ventricular pressure causes the closure of semilunar valve to prevent the back-flow of blood into ventricles.
If type A agglutinogens are present on the surface of RBCs of a person but type B are absent, then what type of agglutinin will be present in the plasma of this person?
1. Anti-A
2. Anti-B
3. Nil
4. Both Anti-A and Anti-B
The thalamus does all of the following except
1. processes and relays sensory information to the primary sensory cortex
2. filters incoming information and forwards relatively little to the cerebrum
3. receives and processes sensory input from olfactory receptors in the nose
4. sends signals about conscious awareness of emotional states to frontal lobes
Which region of the diencephalon controls the ANS and endocrine system and body temperature?
1. epithalamus
2. hypothalamus
3. thalamus
4. infundibulum
Reissner’s membrane separates the cochlear duct from the _____.
1. round window
2. scalavestibuli
3. scala tympani
4. basilar membrane
The autonomic division of the peripheral nervous system does not innervate:
1. visceral organs
2. exocrine glands
3. skeletal muscle
4. endocrine glands
What is true if an object that is more than 20 feet from the eye and is clearly focused on the retina?
1. lens is most strong and refractory
2. lens is not refracting the light
3. ciliary muscle is relaxed
4. zonular fibers of the suspensory ligament are relaxed
Catecholamines like adrenaline and noradrenaline are derived from the amino acid:
1. glycine
2. tyrosine
3. tryptophan
4. glutamic acid
The reflex arc ends at:
1. sensory neuron
2. motor neuron
3. effector
4. receptor
The examination of CSF from a healthy person should not have:
1. glucose
2. electrolytes
3.
4. Erythrocytes
The third ventricle, filled with CSF, is the cavity near:
1. hippocampus
2. thalamus
3. cerebellum
4. medulla oblongata
The brain stem does not include:
1. medulla
2. pons
3. midbrain
4. cerebral cortex
Mammals have evolved brains that are particularly large relative to their body mass. This is due to the enlargement of which part of the mammalian brain?
1. the cerebellum
2. the cerebrum
3. the whole brain
4. the mesencephalon
If the enzyme acetylcholinesterase were inhibited in postsynaptic membranes, what would be the consequence?
1. accumulation of acetylcholine in the synaptic cleft
2. accumulation of acetylcholinesterase in the synaptic clef
3. muscle relaxation
4. repeated action potentials
An injury to the cerebellum would most probably lead to:
1. hyperphagia
2. coma
3. aphasia
4. ataxia
All the following statements regarding the action potential are true except:
1. Only a relatively small number of and ions actually diffuse across the membrane.
2. This event includes both positive and negative feedback loops.
3. The / pumps are directly involved in creating the action potential.
4. During an action potential, the and total concentration are not significantly changed.
Adenohypophysis is an endocrine gland that secretes all of the following except:
| 1. | corticotropin | 2. | luteinizing hormone |
| 3. | somatostatin | 4. | somatotropin |
Identify the precursor from the given list that can result in the synthesis of all others:
1. aldosterone
2. cholesterol
3. cholecalciferol
4. estrogen
A hormonal disorder that develops when pituitary gland produces too much growth hormone during adulthood is known as:
1. cretinism
2. gigantism
3. Conn’s syndrome
4. Acromegaly
The thyroid hormone that has the most potent effect on increasing metabolic rate would be:
1. thyroxine
2. thyroglobulin
3. triiodothyronine
4. thyrocalcitonin
All the following would be true for the mineralocorticoid, aldosterone, except:
1. secreted from the zona glomerulosa
2. decreases blood levels
3. stimulated by angiotensin II
4. raises blood volume
A patient suspected of having hypothyroidism is investigated and has low blood levels of thyroxin and triiodothyronine. He is given thyrotropin that causes the hormone levels to return to normal. What would such a condition be called?
1. Primary hyposecretion
2. Secondary hyposecretion
3. Primary hypersecretion
4. Secondary hypersecretion
Identify the incorrect statement regarding the main glucocorticoid cortisol in the human body:
1. Cortisol is important during fetal development for the proper differentiation of many tissues
2. Cortisol levels increase during stress and act to increase the plasma concentrations of glucose, amino acids, and fatty acids
3. Cortisol is a potent anti-inflammatory agent and is useful for treating diseases such as arthritis
4. Cortisol promotes the action of the immune system
The human placenta has endocrine functions that does not secrete:
1. prolactin
2. estrogens
3. progesterones
4. human chorionic gonadotropin (HCG)
The ionisation energy of hydrogen is high as compared to alkali metals because of
1. One electron in outermost shell
2. Small size
3. One proton in its nucleus
4. No Neutron
High purity dihydrogen is obtained by electrolysing
1. Warm aqueous barium hydroxide
2. Brine solution
3. Acidified sulphate solution
4. Water gas
Hardness of water cannot be removed by
1. Treating with washing soda
2. Boiling
3. Adding Calgon
4. Addition of chlorine
gas along with solid (Y) is obtained when sodium salt (X) is heated. (X) is again obtained when gas is passed into aqueous solution (Y). (X) and (Y) are
1.
2.
3
4.
A compound which can be used in space vehicles both to absorb and liberate is
1.
2.
3.
4.
An orange coloured solution of acidified with and treated with gives a blue coloured solution. The blue colour is due to the formation of
1.
2.
3.
4.
can be obtained when following reacts with except with
1.
2.
3.
4.
Which nitrate cannot give light brown gas on heating?
1.
2.
3.
4.
can be manufactured by Solvay’s process but cannot be prepared because
1. is more soluble
2. is less soluble
3. is more soluble than
4. is less soluble than
Which of the following metal bicarbonates does not exist as solid?
1.
2.
3.
4.
Which of the following metal is most commonly used in photochemical cells?
1. Lithium
2. Calcium
3. Caesium
4. Francium
Which of the following is ‘dead burnt plaster’?
1.
2.
3.
4.
A pair of metals which dissolves in sodium hydroxide solution is
1. Cu, K
2. Fe, Mg
3. Ag, Cu
4. Sn, Zn
Which of the following substances can be used for drying gas?
1. Calcium carbonate
2. Sodium carbonate
3. Sodium bicarbonate
4. Calcium oxide
Alkali metals in liquid produce a blue coloured solution, due to
1. Polarisability of
2. Charge transfer
3. dd transition
4. Ammoniated electrons
Lewis acid character of boron trihalides follows the order
1.
2.
3.
4.
Which one of the following alkali metal does not form alums?
1. Li
2. Na
3. K
4. All of these
When we heat borax strongly then it will yield the compound
1.
2.
3.
4. Both (1) and (2)
Which one of the following compounds has similar structure to that of graphite?
1. Boron nitride
2. Boron carbide
3. Aluminium oxide
4. Aluminium carbide
is known as
1. Chain silicate
2. Pyrosilicate
3. Sheet silicate
4. Ring silicate
In borax, the hybridised state of boron is
1.
2.
3. as well as
4. sp
The best reducing agent is
1.
2.
3.
4.
When zeolite, which is hydrated sodium aluminum silicate, is treated with hard water the sodium ions are exchanged with
1. H+ ions
2. Ca2+ ions
3. Mg2+ ions
4. Both Ca2+ and Mg2+
Polyphosphates are used as water softening agents because the
1. Form soluble complexes with anionic species
2. Precipitate anionic species
3. Forms soluble complexes with cationic species
4. Precipitate cationic species
One mole of calcium phosphide on reaction with excess water gives
1. One mole of phosphine
2. Two moles of phosphoric acid
3. Two moles of phosphine
4. One mole of phosphorus pentaoxide
Saline hydrides react explosively with water, such fires can be extinguished by
1. Water
2. Carbon dioxide
3. Sand
4. None of these
Out of the two allotropic forms of dihydrogen, the form with lesser molecular energy is
1. Ortho
2. Meta
3. Para
4. All have same energy
D2O is preferred to H2O, as a moderator, in nuclear reactors because
1. D2O slows down fast neutrons better
2. D2O has a high specific heat
3. D2O is cheaper
4. None of these
Permanent hardness due to Mg2+ ions is best removed by
1. Ca(OH)2
2. Na2CO3
3. Na2CO3 + Ca(OH)2
4. None of these
Hydrogen can be obtained from water, by the action of water on
1. Calcium carbide
2. Calcium hydride
3. Calcium oxide
4. Calcium
The amount of H2O2 present in 1 L of 1.5 N H2O2 is
1. 2.5 g
2. 25.5 g
3. 3.0 g
4. 8.0 g
Hydrogen can behave as a metal
1. At a very high temperature
2. At very low temperature
3. At very high pressure
4. At very low pressure
HCl is added to the following oxides. Which one would give H2O2
1. MnO2
2. PbO2
3. BaO
4. None of these
Metals of groups 7, 8, and 9 do not form metallic hydrides. This is termed as
1. Hydride gap
2. Hydride shift
3. Anhydride
4. Dehydride
Hydrogen can be produced by heating
1. Cu with H2SO4
2. Sodium formate
3. Sodium oxalate
4. None of these
Plumbosolvency is a health hazard in the transportation of
1. Hard water only
2. Soft water only
3. Both 1 and 2
4. Water containing plum juice
When temporary hard water containing Mg(HCO)3 is boiled the ppt. formed is of
1. MgCO3
2. MgO
3. Mg(OH)2
4. None of these
A sample of water contains sodium chloride. It is
1. Hard water
2. Soft water
3. Moderately hard
4. None of these
The most abundant element in the universe is
1. Carbon
2. Silicon
3. Hydrogen
4. Helium
Pick out the correct statement
1. By decreasing the temperature pure para-hydrogen can be obtained
2. By increasing the temperature pure ortho-hydrogen can be obtained
3. By decreasing the temperature pure ortho-hydrogen can be obtained
4. By increasing the temperature pure para-hydrogen can be obtained
A commercial sample of hydrogen peroxide is labeled as 10 volume. Its percentage strength is nearly
1. 1%
2. 3%
3. 10%
4. 90%
Which o f the following is/are hard water(s)
1. Water containing some potash alum
2. Water containing a few drops of HCl
3. Water containing common salt
4. Water containing calcium nitrate
Hydrogen is evolved by the action of cold dil. HNO3 on
1. Fe
2. Mn
3. Cu
4. Al
A sample of water contains sodium chloride. It is
1. Hard water
2. Soft water
3. Moderately water
4. None of these
Hardness producing salt, whose solubility in water decreases with rise of temperature is
1. CaCl2
2. CaSO4
3. Ca(HCO3)2
4. MgSO4
A long cylindrical vessel of volume V and linear coefficient of expansion contains a liquid. The level of liquid has not changed on heating. The volume coefficient or real expansion of liquid is :
1.
2.
3.
4.
A small quantity of water of mass m at a temperature is poured on to a large mass M of ice which is at melting point. If c is the specific heat capacity of water and L the specific latent heat of fusion of ice, the mass of the ice melted is given by :
1.
2.
3.
4.
Two straight metallic strips each of thickness t and length l are riveted together. Their coefficients of linear expansion are and . If they are heated through a temperature , the bimetallic strip will bend to form an arc of radius :
1.
2.
3.
4.
A constant pressure air thermometer gave a reading of 47.5 units of volume when immersed in ice-cold water, and 67.5 units in boiling water and a reading of 74.5 units in a boiling liquid. The boiling point of the liquid is :
1.
2.
3.
4.
If P represents the pressure exerted by an ideal gas and E represents the translational kinetic energy per unit volume of the gas, then
1.
2.
3.
4.
Which of the following graph best represents the relationship between absolute temperature of a gas and the average kinetic energy of the gas molecules?
1.
2.
3.
4.
The latent heat of vaporization of water is 2240 J/gm. If the work done in the process of vaporization of 1g of water at is 168 J, then an increase in internal energy is :
1. 2408 J
2. 2240 J
3. 2072 J
4. 1904 J
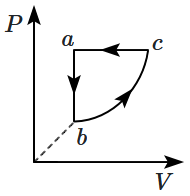
An ideal gas changes from state a to state b as shown in figure. What is the work done by the gas in the process?
1. Zero
2. Positive
3. Negative
4. Infinite
The loss in weight of a solid when immersed in a liquid at is and at is . If the cubical coefficient of expansion of the solid and the liquid be and respectively, then is equal to:
1.
2.
3.
4.
If two rods of length L and 2L having coefficients of linear expansion and respectively are connected so that their total length becomes 3L, the average coefficient of linear expansion of the composition of rods equals:
1.
2.
3.
4. none of these
Two identical containers A and B have frictionless pistons. They contain the same volume of an ideal gas at the same temperature. The mass of the gas in A is and that in B is . The gas in each cylinder is now allowed to expand isothermally to double the initial volume. The changes in the pressure in A and B are found to be and respectively.
1.
2.
3.
4.
A gas undergoes a process in which its pressure P and volume V are related as constant. The bulk modulus for the gas in this process is :
1.
2.
3.
4.
In the cyclic process shown on the V-P diagram, the magnitude of the work done is :
1.
2.
3.
4.
A cyclic process is shown in the P-T diagram. Which of the curves show the same process on a V-T diagram?
1.
2.
3.
4.
The molar heat capacity for an ideal gas
1. cannot be negative
2. must be equal to either or
3. must lie in the range
4. may have any value between and
A cyclic process is shown on the P-T diagram. Which of the curves show the same process on a V-T diagram?
1.
2.
3.
4.
A cyclic process is shown on the P-T diagram. Which of the curves show the same process on a P-V diagram?
1.
2.
3.
4.
Three rods of the same dimensions have thermal conductivities 3k, 2k, and k. They are arranged as shown, with their ends at and The temperature of their junction is
1.
2.
3.
4.
Three rods A, B and C have the same dimensions. Their thermal conductivities are and respectively. A and B are placed end to end, with their free ends kept at a certain temperature difference. C is placed separately, with its end kept at the same temperature difference. The two arrangements conduct heat at the same rate. must be equal to
1.
2.
3.
4.
A point source of heat of power p is placed at the centre of a spherical shell of mean radius R. The material of the shell has thermal conductivity k. If the temperature difference between the outer and inner surfaces of the shell is not to exceed T, the thickness of the shell should not be less than
1.
2.
3.
4.
The temperature of an isolated black body falls from to in time t. Let c be a constant.
1.
2.
3.
4.
A black body is at a temperature of 2880 K. The energy of radiation emitted by this object with wavelength between 499 nm and 500 nm is , between 999 nm and 1000 nm is and between 1499 nm and 1500 nm is . The Wien's constant is.
1.
2.
3.
4.
A body with an initial temperature is allowed to cool in a surrounding which is at a constant temperature of . Assume that Newton's law of cooling is obeyed. Let k = constant. The temperature of the body after time t is best expressed by
1.
2.
3.
4.
A system S receives heat continuously from an electrical heater of power 10 W. The temperature of S becomes constant at when the surrounding temperature is . After the heater is switched off, S cools from to in 1 minute. The heat capacity of S is
1.
2.
3.
4.
A body cools in a surrounding which is at a constant temperature of . Assume that it obeys Newton's law of cooling. It's temperature is plotted against time t. Tangents are drawn to the curve at the points and . These tangents meet the time axis at angles of and , as shown.
1.
2.
3.
4.
An ideal gas expands according to the law = const. The molar heat capacity C is :
1.
2.
3.
4.
An empty pressure cooker with a volume of \(10\) liters contains air at atmospheric pressure \(10^5~\text{Pa}\) and temperature of \(27^\circ \text{C}.\) The cooker has a whistle with an area of \(0.1\) cm² and a mass of \(100~\text{grams}.\)
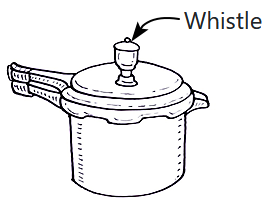
What temperature should the air inside the pressure cooker be, so that, the whistle is just lifted?
1. \(400^\circ\text{C}\)
2. \(227^\circ\text{C}\)
3. \(327^\circ\text{C}\)
4. \(600^\circ\text{C}\)
An air bubble of volume is released by a fish at a depth h in a lake. The bubble rises to the surface. Assume constant temperature and standard atmospheric pressure above the lake. The volume of the bubble just before touching the surface will be (density of water is ) :
1.
2.
3.
4.
Two containers of equal volume contain the same gas at pressures and and absolute temperatures and respectively. On joining the vessels, the gas reaches a common pressure P and a common temperature T. The ratio P/T is equal to –
1.
2.
3.
4.
N (< 100) molecules of a gas have velocities 1, 2, 3, ........ N km/s respectively. Then –
1. rms speed and average speed of molecules is same
2. ratio of rms speed of average speed is
3. ratio of rms speed of average speed is
4. ratio of rms speed of average speed of molecule is
The average translational energy and the rms speed of the molecules in a sample of oxygen gas at are and respectively. Assuming ideal gas behaviors, the corresponding values at are nearly
1.
2.
3.
4.
One mole of an ideal gas is taken from state A to state B by three different processes, (a) ACB (b) ADB (c) AEB as shown in the P-V diagram. The heat absorbed by the gas is–
1. greater in process (b) than in (a)
2. the least in process (b)
3. the same in (a) and (c)
4. less in (c) then in (b)
When a sample of a gas is taken from stable i to state f along the path ‘iaf’, heat supplied to the gas is 50 cal and work done by the gas is 20 cal. If it is taken by path ‘ibf’, then heat supplied is 36 cal.
(a) Work done by the gas along path ibf is 6 cal
(b) If work done upon the gas is 13 cal for the return path ‘fi’, then heat rejected by the gas along path ‘fi’ is 43 cal.
(c) If internal energy of the gas at state i is 10 cal, then internal energy at state ‘f’ is 40 cal
(d) If internal energy at stable ‘b’ is 22 cal and at ‘i’ is 10 cal then heat supplied to the gas along path ‘ib’ is 18 cal.
1. ab
2. ac
3. abc
4. abcd
The equation of state of a gas is where a, b, c and R are constants. The isotherms can be represented by , where A and B depend only on temperature. Hence :
1. m = c and n = –1
2. m = –c and n = –1
3. m = c and n = 1
4. m = –c and n = 1
mole of an ideal gas at constant temperature kept inside a cylinder of length L and cross-section area A closed by a massless piston.
The cylinder is attached with a conducting rod of length L , cross-section area and thermal conductivity k, whose other end is maintained at . If piston is moved such that rate of heat flow through the conducting rod is constant then velocity of piston when it is at height L/2 from the bottom of cylinder is : [ neglect any kind of heat loss from system ]
1.
2.
3.
4.
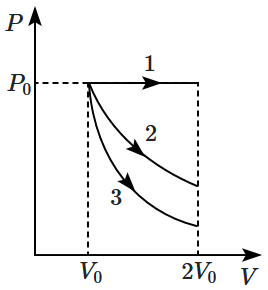
Let \(\Delta U_1, \Delta U_2~\text{and}~\Delta U_3\) be the change in internal energy of the gas is these three processes. Then:
1. \(\Delta U_1>\Delta U_2>\Delta U_3\)
2. \(\Delta U_1<\Delta U_2<\Delta U_3\)
3. \(\Delta U_2<\Delta U_1<\Delta U_3\)
4. \(\Delta U_2<\Delta U_3<\Delta U_1\)
The molar heat capacity C for an ideal gas going through a given process is given by C = a/T , where 'a' is a constant. If , the work done by one mole of gas during heating from to through the given process will be:
1.
2.
3.
4. none of these
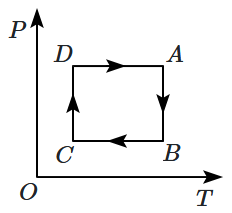
A cyclic process is shown on the \(V\text-T\) diagram. The same process on a \(P\text-T\) diagram is shown by:
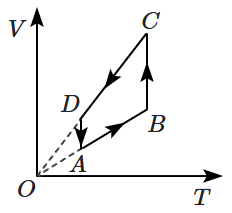
| 1. | 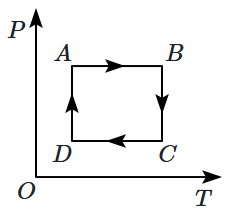 |
2. |  |
| 3. | 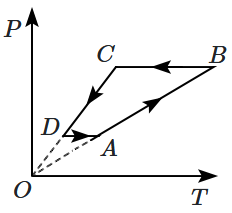 |
4. | 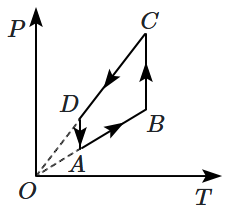 |
An ideal gas undergoes a thermodynamic cycle as shown in the figure. Which of the following graphs represents the same cycle:
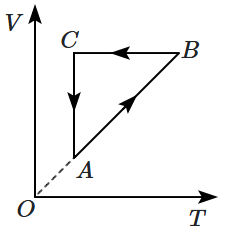
| 1. | 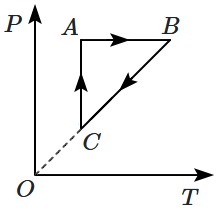 |
2. | 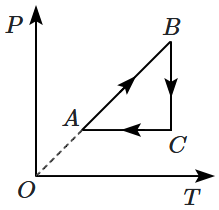 |
| 3. | 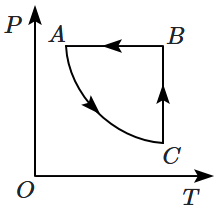 |
4. | 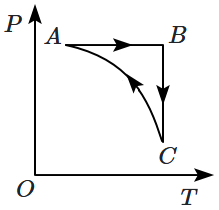 |
\(P\text-V\) curve for the process whose \(V\text-T\) curve is:
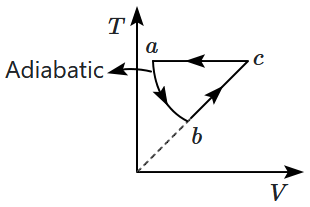
| 1. | 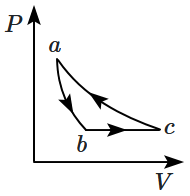 |
2. | 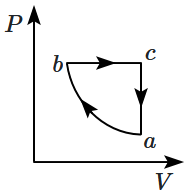 |
| 3. | 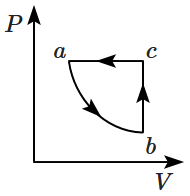 |
4. |  |
Two rods of lengths and are made of materials having coefficients of linear expansion and respectively. If is independent of temperature, which of the following relations is correct?
1.
2.
3.
4. none of these.
Two ends of a conducting rod of varying cross-section are maintained at and as shown in Figure. In steady state,
1. Temperature difference across AB and CD are equal
2. Temperature difference across AB is greater than that across CD
3. Temperature difference across AB is less than that across CD.
4. Data is not sufficient to predict any of the above statements.
When of ice at melts to water at , the resulting change in its entropy, taking latent heat of ice to be is
1.
2.
3.
4.
Three rods made of the same material and having the same cross-section have been joined as shown in figure. Each rod is of the same length. The left and right ends are kept at and respectively. The temperature of the junction of the three rods is
1.
2.
3.
4.
The power radiated by a black body is P, and it radiates maximum energy around the wavelength . If the temperature of black body is now changed so that it radiates maximum energy around a wavelength , the power radiated by it will increase by a factor of
1.
2.
3.
4.