According to the Central Pollution Control Board, particles that are responsible for causing great harm to human health are of diameter
1. 2.50 µm
2. 5.00 µm
3. 10.00 µm
4. 7.5 µm
Catalytic converters are fitted into automobiles for reducing emission of poisonous gases. The converters are made up of metals namely
1. Fe, Cu and Rh
2. Pt, Pd and Rh
3. Al, Pd and Th
4. Fe, Cu and Pd
Identify 1., 2., 3. and 4. in the diagram
1. 1.–Dirty air; 2.– Water/lime spray ; 3.– Clean air; 4. – Particulate matter
2. 1.–Clean air; 2.– Water/lime spray ; 3.– Particulate matter; 4. – Dirty air
3. 1.–Water/lime spray; 2.– Particulate matter; 3.– Clean air; 4. – Dirty air
4. 1.–Particulate matter; 2.– Dirty air ; 3.– Clean air; 4. – Water/lime spray
Which is incorrect about electrostatic precipitator (ESP)?
1. Corona produces electrons
2. Collecting plates are grounded, so use to attract the charged dust particle
3. Velocity of air between the plates must be low
4. Electrode wires that are maintained at several thousand volts produces corona
What is/are done by catalytic converters?
(1) Unburnt hydrocarbons are converted to CO2 and H2O
(2) CO is converted to CO2
(3) Nitric oxide is changed to N2 gas
(4) All of these
According to Euro-III norms, sulphur should be controlled at ______ in diesel and _____ in petrol
1. 350 ppm; 50 ppm
2. 50 ppm; 150 ppm
3. 50 ppm; 50 ppm
4. 350 ppm; 150 ppm
Which of the following causes thinning of fish eating bird’s egg shell and their premature breaking?
(1) DDT
(2) Mercury
(3) Faecal matter
(4) Paper fibres
Find the correct match
(1) Black foot disease - Cadmium
(2) Itai-itai - Arsenic
(3) Minimata - Nitrate
(4) Hardened bones - Fluoride
Cultural (Accelerated) eutrophication is
(1) Natural ageing of lake
(2) Aging process of water body due to pollutants from man’s activities like effluents from industries and homes
(3) Natural conversion of water body into land
(4) Less algal growth in water body
Select the correct statements from the following
a. Thermal waste water eliminates or reduces the number of organisms sensitive to high temperature
b. In a young lake, the water is cold and clear supporting the little life
c. DDT disturbs calcium anabolism to birds, which causes thinning of eggshell and their premature
breaking, eventually causing decline in bird’s population
d. Terror of Bengal grows abundantly in eutrophic water bodies
1. a and b 2. c and d
3. a and d 4. All of these
The presence of large amount of nutrients in water causes excessive growth of planktonic (free floating) algae, which gives a distinct colour to water. This is called
(1) Red tide
(2) Biomagnification
(3) Algal bloom
(4) Biofortification
Increase in concentration of the toxic substances at successive trophic levels is known as
(1) Biofortification
(2) Biowar
(3) Bioinformatics
(4) Biomagnification
Which of the following statements about eutrophication are true?
a. It is a naturally occurring process
b. It is commonly found in standing rather than running water
c. It can lead to oxygen depletion
d. It is commonly associated with high levels of nitrates and sulphates
e. It is commonly associated with high levels of phosphates and nitrates
1. All of these 2. a, b, c and d
3. b, c and d 4. a, b, c and e
Measuring biochemical oxygen demand (BOD) is a method used for
(1) Estimating the amount of organic matter in sewage water
(2) Working out the efficiency of oil-driven automobiles engines
(3) Measuring the activity of Saccharomyces cerevisae in producing curd on a commercial scale
(4) Working out the efficiency of RBCs about their capacity to carry oxygen
Integrated organic farming is
(1) Cyclical process
(2) Zero waste procedure
(3) Maximum utilisation of resource
(4) All of these
Treatment of e-wastes includes
(1) Buried in landfills
(2) Incineration
(3) Recycling
(4) All of these
Find the incorrect statement
(1) Sanitary landfills were adopted as the substitute for open burning dumps
(2) Seepage of chemical from landfills pollutes the underground water resources
(3) All waste that we generate can be categorised into three types, that is, bio-degradable, recyclable and non-biodegradable
(4) Open dumps inhibitsbreeding of rats and flies
How deep should nuclear waste after sufficient pre-treatment be buried (in shielded containers) within the rocks below the earth’s surface?
(1) 100 m
(2) 200 m
(3) 300 m
(4) 500 m
Eco-san toilets are not associated with which of the following?
(1) Working is Sri Lanka and Kerala
(2) Composting method for recycling of human excreta
(3) Recycled material forms natural fertiliser
(4) Enhance the need for chemical fertilisers
What did Chernobyl,Three mile Island and Bhopal all have in common?
(1) They all qualified as technological disasters
(2) They all are involved in environmental racism
(3) Environmental problems were caused by economic development in all these places
(4) Nuclear disasters took place in all these places
Prolonged water logging in agricultural fields is likely to create the problem of
(1) Poor aeration and low salinity
(2) Poor aeration and high salinity
(3) Poor aeration and high acidity
(4) Metal toxicity and proper aeration
Which of the following figures shows correct relative contribution of greenhouse gases to global warming?
(1)
(2)
(3)
(4)
In human eye, cornea absorbs UV-B radiation, and a high dose of UV-B causes inflammation of cornea, a disease known as
(1) Night-blindness
(2) Xerophthalmia
(3) Epicanthus
(4) Snow-blindness
The presence of ozone (O3) in the stratosphere of atmosphere
(1) Hinders higher rate of photosynthesis
(2) Helps in checking the penetration of ultra-violet rays to earth
(3) Has been responsible for increasing the average global temperature in recent years
(4) Is advantageous since it supplies O2 for people travelling in jets
Government of India has recently instituted which award for individuals or communities from rural areas that have shown extra ordinary courage and dedication in protecting wildlife?
(1) Padma Bhushan
(2) Chipko Award
(3) Bharat Ratna
(4) Amrita Devi Bishnoi-Wildlife Protection Award
The Earth Summit, 1992, was held at
(1) Rio de Janeiro
(2) Kyoto
(3) St. Petersburg
(4) Washington
Match column-I with column-II and select the correct option
Column-I Column-II
a. Air (prevention and control of (i) 1987
pollution) Act
b. Water (prevention and control of (ii) 1981
pollution) Act
c. Noise added as air pollutant (iii) 1974
d. Environment (Protection) Act (iv) 1986
1. a – (ii) ;b – (iii) ; c – (i) ; d – (iv)
2. a – (i) ;b – (iii) ; c – (ii) ; d – (iv)
3. a – (iv) ;b – (iii) ; c – (ii) ; d – (i)
4. a – (iv) ;b – (iii) ; c – (i) ; d – (ii)
Select the incorrect statement
1. India has more than 50,000 genetically different strains of rice
2. India has 100 varieties of mango
3. The genetic variation in Rauwolfia vomitoria can be in terms of concentration and potency of reserpine
4. The tropical rainforest initially covered 14% of the land surface of earth, but now they cover only 6% of the land area
Identify a, b, c and d in the given figure.
The pie-chart for the number of species of major taxa of invertebrates represent respectively
1. a – Insects; b – Crustaceans; c. – Molluscs; d – Other animal groups
2. a – Other animal groups; b – Crustaceans; c – Molluscs; d –Insects
3. a –Molluscs; b – Insects; c – Other animal groups; d – Crustaceans
4. a – Insects; b – Molluscs; c – Crustaceans; d – Other animal groups
Which of the following rainforest is home to more than 40,000 species of plants, 3000 of fishes, 1300 of birds, 427 of mammals, 427 of amphibians, 378 of reptiles and more than 1,25,000 invertebrates?
1. Amazonian
2. Tropical
3. Arctic tundra
4. Temperate
Find the false statement
| 1. | The number of fungi species is more than the combined total of the species of fishes, amphibians, reptiles and mammals |
| 2. | Conventional taxonomic methods are not suitable for identifying microbial species. |
| 3. | For many taxonomic groups, species inventories are more complete in temperate than in tropical countries. |
| 4. | Insects form 70% part of all the species recorded |
Find the true statement
a. If we consider biochemical or molecular criteria for microorganisms, their diversity may run into
millions.
b. There are 12 mega diversity centres in the world.
c. If we accept May’s global estimates, only 8.1 per cent of the total species have been recorded so far.
d. Western Ghats have greater amphibian species diversity than Eastern Ghats.
1. a and b 2. Only d
3. a and d 4. a, b and d
India has only ______ per cent of the world’s land area; its share of the global species diversity is an impressive _____ per cent
Find the incorrect statements
(1) India has nearly 45,000 species of plants and twice as many of animals
(2) A forest in a tropical region like equator has 10 times as many species of vascular plants as a forest of equal area in a temperate region like the mid-west of the US.
(3) Greenland 710N has only 56 species of birds.
(4) The diversity of plant and animals is uniform throughout the world.
The graph formed by plotting species richness against area for a wide variety of taxa (angiosperm plants, birds, bats, fresh water fishes, etc.) turns out to be a
(1) Parabola
(2) Straight line
(3) Rectangular hyperbola
(4) Any of these
Species diversity _______ as we move away from the equator towards the poles
(1) Increase
(2) Decrease
(3) First increases than decreases
(4) First decreases then increases
Which of the following factor has not aided in high species diversity of tropics?
(1) Less seasonal variation promoting niche specialisation
(2) Availability of more solar energy
(3) Less competition
(4) Constant environmental condition
From the following statements you have to select the right option that mentions observations shown by David Tillman, who performed long term ecosystem experiment using outdoor plots?
a. Plots with more species showed less year-to-year variation in total biomass
b. Plots with less species showed less year-to-year variation in total biomass
c. Increased diversity contributed to higher productivity
d. Plots with more species showed year-to-year variation in total rainfall
1. a and c 2. a and d
3. b and d 4. a, c and d
Which hypothesis suggest ecosystems are like aeroplane where flight (ecosystem functioning) may or may not be compromised depending upon which species are lost?
(1) Rivet popper hypothesis
(2) Gaia hypothesis
(3) Gause exclusion hypothesis
(4) Odum’s hypothesis
Match the following Column-I (recently extinct animals) with Column-II (places from where they are extinct) and select the correct option
Column-I Column-II
a. Dodo (i) Mauritius
b. Quagga (ii) Africa
c. Thylacine (iii) Australia
d. Sea cow (iv) Russia
1. a – (i) ;b – (ii) ; c – (iii) ; d – (iv)
2. a – (i) ;b – (ii) ; c – (iv) ; d – (iii)
3. a – (ii) ;b – (i) ; c – (iii) ; d – (iv)
4. a – (ii) ;b – (i) ; c – (iv) ; d – (iii)
The Evil Quartet is the sobriquet used to describe
(1) Population growth
(2) Causes of biodiversity losses
(3) Water pollutants
(4) DO of water body
Find the false statement
| a. | In the last 27 years, there is disappearance of 27 species |
| b. | Three species (Bali, java and Caspian) of tiger are included in recently extinct animals |
| c. | If we apply May’s estimate, then there are probably more than 1,00,000 plants species in India that are yet to be discovered and described. |
| d. | There is more solar energy available in tropics, which contribute to higher productivity, this in turn contributes directly to greater diversity |
1. Only a
2. c and d
3. b and d
4. a and c
Loss of biodiversity in a region may lead to
(1) Decline in plants production
(2) Lowered resistance to environmental perturbations such as draught.
(3) Increased variability in certain ecosystem process such as plant productivity, water use, and pest and disease cycle
(4) All of these
Co-extinction means
(1) Introduction of alien species leading to decline or extinction of indigenous species
(2) Extinction due to over-exploitation
(3) Extinction due to habitat loss
(4) Extinction due to obligatory association of plant or animal
Sacred groves are present in
(1) Khasi and Jaintia hills in Meghalaya
(2) Aravalli hills of Rajasthan
(3) Western Ghat regions of Karnataka and Maharashtra
(4) All of these
Consider the following two statements regarding enzyme action:
| I: | Co-factors play a crucial role in the catalytic activity of the enzyme. |
| II: | Catalytic activity is reduced when the co-factor is removed from the enzyme. |
Of the above statements:
1. Both I and II are correct and II explains I
2. Both I and II are correct but II does not explain I
3. I is correct but II is incorrect
4. I is incorrect but II is correct
Enzymes, vitamins and hormones are common in:
(1) Being proteinaceous
(2) Being synthesized in the body of organisms
(3) Enhancing oxidative metabolism
(4) Regulating metabolism
The four elements making 99 % of living system are:
| 1. | CHOS | 2. | CHOP |
| 3. | CHON | 4. | CNOP |
Glycogen is a polymer of:
1. Galactose
2. Glucose
3. Fructose
4. Sucrose
Most of the functions of a cell membrane can be attributed to its:
1. Phospholipids
2. Cholesterol
3. Proteins
4. Carbohydrates
An enzyme brings about:
1. Decrease in reaction time
2. Increase in reaction time
3. Increase in activation energy
4. Reduction in activation energy
In competitive inhibition, if the concentration of the substrate is increased:
(1) the rate of the reaction is increased
(2) the rate of the reaction is decreased
(3) the rate of the reaction is not affected
(4) the reaction direction is reversed
The backbone of a DNA molecule is formed by:
| 1. | nitrogenous bases |
| 2. | deoxyribose sugar |
| 3. | phosphate groups |
| 4. | alternate sugar and phosphate moieties |
A glycosidic bond connects:
(1) two monosaccharides
(2) two amino acids
(3) two fatty acids
(4) a fatty acid and a glycerol
All of the following are true of protein denaturation except that it
| 1. | is a shape change |
| 2. | is always irreversible |
| 3. | may be caused by a pH change |
| 4. | could result from a temperature change |
Assuming they all had the same number of carbon atoms, which of the following has the most C-H bonds?
(1) an unsaturated fat
(2) a polyunsaturated fat
(3) a polysaccharide
(4) a saturated fat
Why is cellulose so difficult for most animals to digest?
1. they don’t have the proper enzyme to break the bonds between subunits
2. cellulose is made up of chitin, which is indigestible
3. the bonds holding cellulose subunits together are extremely strong, stronger than in any other macromolecule
4. there are many hydrogen bonds holding the subunits together
Catalytic efficiency of two different enzymes is compared by their:
(1) Product
(2) Molecular size
(3) Km value
(4) pH optimum value
Carbohydrates are commonly found as starch in plant storage organs. Which of the
following five properties of starch (a-e) make it useful as a storage material?
a. easily translocated
b. chemically non-reactive
c. easily digested by animals
d. osmotically inactive
e. synthesized during photosynthesis
The useful properties are:
1. (a), (b) and (e)
2. (a) and (e)
3. (b) and (c)
4. (b) and (d)
A molecule of ATP is structurally most similarto:
(1) RNA nucleotide
(2) DNA nucleotide
(3) amino acid
(4) fatty acid
In feedback inhibition, a metabolic pathway is switched off by:
1. a rise in temperature
2. lack of a substrate
3. accumulation of the end product
4. competitive inhibition
The unique properties of each amino acid is determined by its particular:
1. R group
2. amino group
3. kinds of peptide bond
4. number of bonds to other amino acids
The ion required as the co-factor for the proteolytic enzyme, carboxypeptidase, is:
(1) Zinc
(2) Copper
(3) Iron
(4) Manganese
The inhibition of succinic dehydrogenase by malonate is an example of:
1. Non-competitive reversible inhibition
2. Non-competitive irreversible inhibition
3. Competitive inhibition
4. Allosteric inhibition
The enzymes that catalyse the removal of groups from substrates by mechanisms other than hydrolysis leaving double bonds are called:
1. Oxidoreductases
2. Hydrolases
3. Ligases
4. Lyases
The property of the water molecule that is most important for the maintenance of homeostasis in living organisms is:
(1) High latent heat of vaporization
(2) High latent heat of fusion
(3) High specific heat
(4) Liquid form more dense than solid form
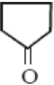
Given below is the structure of a monosaccharide. The homopolymer of this monosaccharide is used in: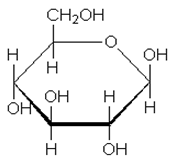
(1) Storage of carbohydrates in plants
(2) Storage of carbohydrates in animals
(3) Making the medium for microbial culture
(4) Formation of the structure of the plant cell wall
Given below is the structure of a lipid. The excess of this compound in human blood can lead to:
Shown below is a level of structural organization of a protein. Which of the following proteins attains its final form as this structure? 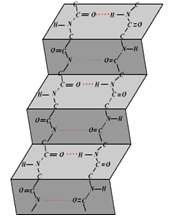
| 1. | Keratin |
| 2. | Fibroin |
| 3. | Tropo-collagen |
| 4. | Myoglobin |
The following diagram depicts the structure of a:
1. Nucleoside
2. Nucleotide
3. Nitrogen containing Monosaccharide
4. Glycoprotein
Which of these is under voluntary control?
1. urethra
2. detrusor muscle
3. internal urethral sphincter
4. external urethral sphincter
The mucosa of the bladder is comprised of _____.
(1) smooth muscle
(2) squamous epithelium
(3) transitional epithelium
(4) simple columnar epithelium
Which of these could appear in the urine from dieting or the utilization of excess lipids?
(1) urea
(2) uric acid
(3) glycine
(4) Ketone
The targets of angiotensin II are blood vessels and:
(1) nerves
(2) adrenal cortex
(3) adrenal medulla
(4) kidney nephron
Which muscle metabolism waste product is eliminated by the kidneys?
(1) urea
(2) uric acid
(3) creatine
(4) Creatinine
Which area actually secretes renin into the blood?
1. macula densa
2. juxtaglomerular apparatus
3. juxtaglomerular cells
4. cortical nephron
Which section of the nephron follows the ascending limb of the loop of Henle?
1. descending limb of the loop
2. proximal convoluted tubule
3. distal convoluted tubule
4. collecting duct
How much of the cardiac output passes through the kidneys?
(1) 10%
(2) 20%
(3) 50%
(4) 65%
The kidney secretes _____ for the purpose of stimulating bone marrow activity.
(1) renin
(2) aldosterone
(3) erythropoietin
(4) Somatomedin
The kidneys are located in the _____ space.
(1) pelvic cavity
(2) peritoneal cavity
(3) abdominal
(4) retroperitoneal
The two organs most responsible for acid-base balance are the
(1) heart and kidneys
(2) liver and lungs
(3) kidneys and lungs
(4) lungs and heart
Which statement about aldosterone is false?
1. It is a major steroid (mineralocorticoid) hormone secreted by the kidney.
2. It promotes the reabsorption of Na+ in the late distal tubule and the cortical region of the collecting duct.
3. It stimulates the secretion of K+ from the peritubular blood into the distal tubule.
4. Without it, no K+ is ever excreted in the urine.
The concentrations of which plasma electrolyte is not regulated mainly by the kidneys?
1. hydrogen
2. potassium
3. iron
4. Bicarbonate
Which substance is filtered, reabsorbed, and secreted by different regions of the nephron tubules?
(1) potassium ion (K+)
(2) inulin
(3) urea
(4) glucose
Which substance is filtered and then completely (100%) reabsorbed by the nephron?
(1) protein
(2) inulin
(3) urea
(4) glucose
The disease diabetes insipidus
(1) is associated with the inadequate secretion or action of antidiuretic hormone (ADH).
(2) results from overeating and hypersecretion of hormones by the pancreas.
(3) is characterized by an abnormal increase in glucose concentrations in the blood and urine.
(4) can be cured by injections of the hormone, insulin.
Which statement about the glomerular filtration rate (GFR) is false?
| 1. | The GFR averages approximately 180 L per day. |
| 2. | The GFR will increase during fight-or-flight situations due to the vasodilation of afferent arterioles. |
| 3. | A decrease in GFR results in a decrease in the total urine output. |
| 4. | Renal autoregulation maintains the GFR at a relatively constant rate despite fluctuations in the mean arterial blood pressure. |
The net filtration pressure at the glomerulus that results in the formation of ultrafiltrate is
(1) about equal to arterial blood pressure at 100 mmHg
(2) partially due to the very low colloid osmotic pressure of plasma
(3) opposed by the high osmotic pressure of ultrafiltrate
(4) estimated to be approximately 10 mm Hg
That portion of the nephron consisting of a single layer of cuboidal cells with microvilli to increase the surface area for reabsorption is the:
(1) glomerular capsule
(2) proximal convoluted tubule
(3) loop of Henle
(4) distal convoluted tubule
The name of the blood vessel delivering blood directly to the glomerulus is the:
(1) renal artery
(2) interlobar artery
(3) arcuate artery
(4) afferent arteriole
Consider the following organic compound
To make it a chiral compound, the attack should be on carbon
1. 1
2. 3
3. 4
4. 7
which of the following will show geometrical isomerism?
1. 1-Butene
2. 1,2-Dibromobutene
3. Propene
4. Isobutylene
The number of isomers for the compound with molecular formula is
1. 3
2. 4
3. 5
4. 6
Which of the following does not show geometrical isomerism?
1. 1,2-dichloro-1-pentene
2. 1,3-dichloro-1-pentene
3. 1,1-dichloro-1-pentene
4. 1,4-dichloro-1-pentene
The geometrical isomerism is shown by
1. Geometrical isomerism
2. Optical isomerism
3. Geometrical isomerism and optical isomerism
4. Neither Geometrical isomerism nor optical isomerism
The absolute configuration of the following compound is
1. 2S, 3R
2. 2S, 3S
3. 2R, 3S
4. 2R, 3R
Which of the following pairs of compounds are enantiomers?
The most stable carbocation among the following is
Consider the following carbocations
1. II < I < III < IV
2. II < III < I < IV
3. III < I < II < IV
4. IV < III < I < II
Which one of the following is most reactive towards electrophilic attack?
Pick out the correct order of stability for the following species:
1. II > IV > I > III
2. II > II > III > IV
3. II > I > IV > III
4. I > III > II > IV
Arrange the following halides in the decreasing order of reactivity
(I)
(II)
(III)
1. I > II > III
2. II > I > III
3. II > III > I
4. III > II > I
Butanonitrile may be prepared by heating
1. Propyl alcohol with KCN
2. Butyl chloride with KCN
3. Butyl alcohol with KCN
4. Propyl chloride with KCN
Identify X
1.
2. Ethyl alcohol
3. Methyl cyanide
4. tert-Butyl alcohol
Which of the following chloro derivatives of benzene would undergo hydrolysis most readily with aqueous KOH to give the corresponding hydroxy derivative?
The correct order of solvolysis in the following alkyl halide is
Identify the set of reagent and reaction conditions 'X' and 'Y' in the following set of transformations,
1. X = Dilute aqueous NaOH, ;
Y = HBr/acetic acid,
2. X = Concentrated alcoholic NaOH, ;
Y = HBr/acetic acid,
3. X = Dilute aqueous NaOH, ;
Y =
4. X = Concentrated alcoholic NaOH, ;
Y =
Compound [X] having molecular formula on hydrolysis gives a ketone. Therefore [X] will be
1.
2.
3.
4.
The intermediate during the additional of HCl to propene in the presence of peroxide is
1.
2.
3.
4.
The products of reaction of alcoholic silver nitrite with ethyl bromide are :
1. Ethane
2. Ethene
3. Ethyl alcohol
4. Nitroethane
Identify C in the following series:
1.
2.
3.
4.
Which one of the following is most reactive towards nucleophilic substitution reaction?
1.
2.
3.
4.
Bottles containing and lost their orginal lables. They were lablled A and B for testing. A and B were separately taken in test tube and boiled with NaOH solution. The end solution in each tube was made acidic with and some solution added. Solution B gave a yellow ppt. which one of the following statements is true for the experiment ?
1. Addition of was necessary
2. A was
3. A was
4. B was
On monochlorination of 2-methylbutane, the total number of chiral compounds is
1. 2
2. 4
3. 6
4. 8
Phenylmagnesium bromide reacts with methanol to give
1. A mixture of toluene and Mg(OH)Br
2. A mixture of phenol and Mg(Me)Br
3. A mixture of anisol and Mg(OH)Br
4. A mixture of benzene and Mg(Me)Br
Tertiary alcohol can be prepared from
1. Ketone and
2. Aldehyde and RMgX
3. and RMgX
4. and RMgX
Consider the following reaction :
In this reaction what will take place:
1. Inversion
2. Retention
3. Racemisation
4. Epimerisation
Phenol is least reactive for aromatic nucleophilic substitution because
1. Carbon-oxygen has some double bond character due to resonance
2. Oxygen is present on -hybrid carbon which makes carbon-oxygen bond stronger
3. Oxygen is highly electronegative which decreases bond length between
4. All are correct
4. All of these
Which of the following phenols is soluble in ?
1. 2, 4, 6- Trinitrophenol
2. 4- Nitrophenol
3. 2- Nitrophenol
4. All of these
A compound X with molecular formula can be oxidised to a compound Y with the molecular formula , X is most likely to be
1. Primary alcohol 2. Secondary alcohol
3. Aldehyde 4. Ketone
Reaction of t-butyl bromide with sodium methoxide produces
1. Isobutane 2. Isobutylene
3. Sodium t-butoxide 4. t-butyl methyl ether
Identify X in the following sequence of reactions
1.
2.
3.
4.
Phenolphthalein is produced on heating phthalic anhydride and conc. sulphuric acid with
1. Salicyclic acid 2. Phenol
3. Phenacetin 4. Phenanthrene
The compound Y in the above sequence is
1. 2-Methyl-2-phenyl-1-propanol
2. 2-Phenyl-2-propanol
3. Acetophenone
4. 2-Methyl-1-Phenyl-2-propanol
An organic compound of molecular formular does not react with sodium. With excess of HI, it gives only two types of alkyl halide. The compound is
1. Ethoxyethane
2. 2- Methylpropane-2-ol
3. 1-Methyoxypropane
4. 1-Butanol
Methanol and ethanol can be distinguished by the following:
1. By reaction with metallic sodium
2. By reaction with caustic soda
3. By heating with iodine and washing soda
4. By heating with zinc and inorgnaic mineral acid
Glycerol on treatment with oxalic acid at 110 forms:
1. Formic acid 2.
3. Allyl alcohol 4. Glycol
1-propanol and 2-propanol can be best distinguished by:
1. Oxidation with alkaline followed by reaction with Fehling solution.
2. Oxidation with acidic dichromate followed by reaction with Fehling solution.
3. Oxidation by heating with copper followed by reaction with Fehling solution
4. Oxidation with conc. followed by reaction with Fehling solution
Identify (X) in the sequence:
1.
2.
3.
4.
Dehydration of alcohols
will be in order:
1. I< II< III< IV
2. I> II> III> IV
3. III< II < I< IV
4. II < III < IV < I
In the following sequence of reactions,
The compound C formed will be
1. Butanol-1
2. Butanol-2
3. 2-Methylpropanol-1
4. 2-Methylpropan-2-ol
The reaction products of
1. 2.
3. 4.
are the conductances of three conductors. When they are joined in series, their equivalent conductance will be
4. Given by some other relation
The resistance of a 10m long wire is 10. Its length is increased by 25% by stretching the wire uniformly. The resistance of the wire will change to (approximately)
1. 12.5
2. 14.5
3. 15.6
4. 16.6
In the circuit shown in figure, the point F is grounded. Which of the following is a wrong statement?
1. D is at 5V
2. E is at zero potential
3. The current in the circuit will be 0.5A
4. The potential at E is same whether or not F is grounded
To get a maximum current through a resistance of 2.5 , one can use m rows of cells having n cells. The internal resistance of each cell is 0.5 . What are the values of m and n, if the total no. of cells is 20?
1. m = 2, n = 10
2. m = 4, n = 5
3. m = 5, n = 4
4. n = 2, m = 10
A battery of internal resistance 4 is connected to the network of resistances as shown. In order to give the maximum power to the network, the value of R (in ) should be
1. 4/9
2. 8/9
3. 2
4. 18
Seven resistances each of 20 are connected with 2 volt battery as shown in figure. The reading of the ammeter will be
1. (1/10)A
2. (3/10)A
3. (4/10)A
4. (4/10)A
Equivalent resistance between A and B is
1.
2. 4R
3.
4. None of these
If I be the safe current for the fuse wire of radius r, then:
Figure shows three similar lamps connected across a power supply. If the lamp L3 fuses, How will the light emitted by L1 and L2 change?
1. No change
2. Brilliance of L1 decreases and that of L2 increases
3. Brilliance of both L1 and L2 increases
4. Brilliance of both L1 and L2 decreases
An electric kettle has two coils in parallel. When one of these is switched on, the water boils in 6 minutes. When the other coil is switched on, the water boils in 3 minutes. If both of them are switched on simultaneously, how much time will be taken to boil the water?
1. 2 minutes
2. 1.5 minute
3. 1 minute
4. 0.5 minute
A copper wire of length l and radius r is nickel plated till its final radius is R. If the specific resistance of copper and nickel be and , then the conductance of the wire will now
1.
2.
3.
4.
Magnetic field B on the axis of a circular coil and far away distance x from the centre of the circular coil are related as:
1.
2.
3.
4. None of these
An infinite current carrying conductor is bent into a circle as shown in the figure. If the radius of the circle is R, the magnetic field at the centre of the coil is:
1. Infinite
2. Zero
3.
4.
A metallic pipe carries direct current. Which of the following statements about the existance of the magnetic field is true?
1. It exists outside only
2. It exists inside only
3. It exists both inside as well as outside
4. It exists neither inside nor outside
An infinite long straight wire is bent into a semicircle of radius R, as shown in the figure.
A current I is bent through the conductor. The magnetic field at the centre of the semicircle is:
1. Infinite
2. Zero
3.
4.
What is the magnetic field at the point of intersection of diagonals of a current carrying square loop of each side L. The current through the loop is I.
1.
2.
3.
4. None of these
A current I amperes flows in the loop having circular arc of radius r metres subtending an angle radian as shown in figure. The magnitude of magnetic field at the centre O is:
1.
2.
3.
4.
A currernt passing through a circular coil of two turns produces a magnetic field B at its centre. The coil is then removed so as to have four turns and the same current is passed through it. Now magnetic field at the centre of the coil is
1. 2B
2. 4B
3. B/2
4. B/4
A cell is connected between points A and C of a circular conductor ABCD of centre O with as shown in the figure. If are the magnitudes of magnetic fields at O due to the currents in ABC and ADC respectively, the ratio is
1. 0.2
2. 6
3. 5
4. 1
A straight conductor carrying direct current I is split into a circular loop as shown in figure. Then the magnetic induction at the centre of the circular loop of radius r is.
1. Zero
2. Infinite
3.
4.
What is the time rate of work done by the magnetic field on the test charge, when it follows a helical path?
1.
2.
3.
4. Zero
In which direction, no change in momentum of test charge occurs, if magnetic field is along the z-axis and velocity of charge is makig angle with magnetic field in x-z plane.
1. Along Z-axis
2. Along Y-axis
3. Along X-axis
4. At angle with X axis
A current carrying loop of radius r carries a current I. It is placed in a magnetic field such that the plane of the coil is perpendicular to the magnetic field. What is the net force on the coil ?
1.
2.
3.
4. Zero
A rectangular loop carries a current I0. It is placed near a long straight conductor carrying current I as shown in figure. What will be the nature of the force experienced by the coil?
1. No force
2. Repulsion
3. Attraction
4. It will experience a torque
The magnetic field in case of the moving coil galvanometer should be radial so that current I through the coil and its deflection are related to each other as:
In the circuit shown, each resistance is 2. The potential as indicated in the circuit, equal to
1. 11 V
2. -11 V
3. 9 V
4. -9 V
In the circuit shown, the value of R in ohm that will result in no current in 10 resistor.
1. 10
2. 25
3. 30
4. 40
A cell of emf E having an internal resistance 'r' is connected to an external resistance R. The potential difference 'V' across the resistance R varies with R as shown by the curve:
1. A
2. B
3. C
4. D
Circuit for the measurement of resistance by potentiometer is shown. The galvanometer is first connected at point A and zero deflection is observed at length PJ = 10 cm. In the second case, it is connected at point C and zero deflection is observed at length 30 cm from the P. Then the unknown resistance X is
1. 2R
2.
3.
4. 3R
For the circuit shown in the figure, determine the charge of capacitor in steady state:
1.
2.
3.
4. Zero
In the following figure, the reading of an ideal voltmeter V is zero. Then, the relation between R, and :
1.
2.
3.
4.
In a meter bridge experiment, resistances are connected as shown in figure. The balancing length l1 is 55 cm. Now an unknown resistance x is connected in series with P and the new balancing length is found to be 75 cm. The value of x is:
1.
2.
3.
4.
n identical cells are joined in series with two cells A and B with reversed polarities. The Emf of each cell is E and internal resistance r. The potential difference across cell A or B is (n > 4)
1.
2.
3.
4.
In a potentiometer experiment, the balancing length with a cell is 560 cm. When an external resistance of 10 is connected in parallel to the cell, the balancing length changes by 412 cm. The internal resistance of the cell (in ohm) is:
1. 3.6
2. 2.4
3. 1.2
4. 0.6
In the shown circuit the resistance R can be varied :
The variation of current through R against R is correctly plotted as :
In the circuit, the galvanometer G shows zero deflection. If the batteries A and B have negligible internal resistance, the value of the resistor R will be :
1.
2.
3.
4.
At a certain place, the horizontal component of earth's magnetic field is times the vertical component. The angle of dip at that place is
The deflection is related to the electric current I in a galvanometer by the relation
Two magnets are held together in a vibration magnetometer and are allowed to a oscillate in the earth's magnetic field with like poles together and 12 oscillations per minute are made but for unlike poles together, only 4 oscillations per minute are executed. The ratio of their magnetic moment is
1. 5 : 4
2. 3 : 1
3. 3 : 5
4. 1 : 3
A magnet oscillates in the earth's magnetic field with time period T. If the mass is quadrupled, then a new time period will be:
1. 2T, motion remaining SHM
2. 4T, motion remaining SHM
3. T/2, motion remaining SHM
4. Unaffected but motion is not SHM
Two bar magnets of the same mass, same length and breadth having magnetic moments M and 2M are joined together pole for pole and suspended by a string. The time period of the assembly in a magnetic field of strength H is 3 seconds. If now the polarity of one of the magnets is reversed and the combination is again made to oscillate, their time period will be,
1. 3 sec
2.
3.
4. 6 sec
The magnetic moment of a circular loop carrying current I and of length L is
1.
2.
3. IL
4. None of these
Vertical component of earth's magnetic field is zero at a place where angle of dip is
1.
2.
3.
4.
When a magnet is suspended horizontally at the neutral point, its frequency of oscillations is
1. Infinity
2. Zero
3. Neither 1. nor 2.
4. Data incomplete
For perfect coupling of two coils of inductance and , their mutual inductance M should be given by
1.
2.
3.
4.