Symptoms of altitude sickness include all except
Shivering during cold is beneficial for mammals. It helps to
(1) Decrease body temperature.
(2) Increase body temperature.
(3) Decrease rate of metabolism.
(4) None of these
Many adaptations of an organism have evolved over a long duration of evolution and are______
(1) Genetically unstable.
(2) Genetically fixed.
(3) Genetically disadvantageous.
(4) Genetically extemporaneous.
The main reason behind building burrows in soil by burrowing animals is
(1) To escape from predators.
(2) To derive nutrition parts of plants from Underground.
(3) The mode of evolution chose burrows as a specific habitat.
(4) To hide and escape from the above ground heat.
Choose the odd one out with respect to structure formed in the different organisms during suspended phase.
(1) Bacteria - Thick-walled spores
(2) Higher plants - Seeds, vegetative propagules
(3) Zooplankton - Diapause state
(4) Ectothermic organism - Torpid state during favourable season.
Smaller animals tend to lose body heat very fast as compared to larger animals because they have
(1) Higher surface to volume ratio.
(2) Lower surface to volume ratio.
(3) Equal values of surface and volume.
(4) Very low BMR (basal metabolic rate).
Select the incorrect statement
(1) Organisms living in oceans, lakes and rivers do not face any water related problems.
(2) Productivity and distribution of plants is dependent on water.
(3) The levels of thermal tolerance of different species determine to a large extent their geographical distribution
(4) Foraging, reproductive and migratory activities of some animals are dependent on seasonal variations in light
Population size in any given habitat is also known as _______
(1) Population cluster
(2) Population explosion
(3) Population abundance
(4) Population density
A striking difference between immigration and emigration is _____
(1) Immigration considers total numbers of species in a given habitat, while emigration refers to a single species.
(2) Emigration considers total numbers of species in a given habitat, while immigration refers to a single species.
(3) Immigration considers total number of dominant species in a given habitat, while emigration refers to a single species.
(4) Immigration considers total number of species in a given habitat, while emigration refers to a dominant single species.
Study the following equation in context of population growth and choose the correct option
dN/dt=(B-D)×N
Substituting r in place of B-D then
dN/dt=rN
Here, r is known as _______
(1) Extrinsic rate of natural decrease.
(2) Intrinsic rate of natural decrease.
(3) Intrinsic rate of natural increase.
(4) Extrinsic rate of natural increase.
What four factors define population growth?
(1) Births, deaths, immigration, emigration
(2) Survivorship, age specific, mortality, fecundity, death rate
(3) Mark recapture, census, quadrate, sampling transects
(4) Age specific birth rate, metapopulation structure, quardrate, ecotone
Which of the following is true concerning exponential growth?
(1) No population can grow exponentially for long.
(2) Exponential growth slows down as the population reaches near its maximal size.
(3) Bacterial colonies have been observed to maintain exponential growth for over a month.
(4) Exponential growth is commonly observed in large, slow growing species such as humans and elephants.
Read the following statements and select the correct option with respect to population attributes.
a. Population density is necessarily measured in numbers.
b. Tiger census is often based on pug marks and fecal pellets.
c. Biomass is not a meaningful measure to known population size.
d. Size of a population for any species is not a static parameter.
1. a. and b. 2. a. and d.
3. c. and d. 4. b. and d.
Select the incorrect statement regarding different population attributes.
(1) Natural selection operates at population level.
(2) Size of the population tells a lot about its status in the habitat.
(3) In age pyramids, age distribution of male and females are shown in separate diagrams.
(4) Population is a group of individuals multiplying by sexual and asexual reproduction
A species whose distribution is restricted to a small geographical area because of the presence of a competitive superior species is found to expand its distributional range dramatically when the competing species is experimentally removed. This phenomenon is known as
(1) Competitive exclusion.
(2) Competitive release.
(3) Interference competition.
(4) Resource partitioning.
Find out the false statements
(1) Parasite that feeds on the external surface of the host organism are called ectoparasite.
(2) The life cycle of endoparasite are more complex because of their extreme specialisation
(3) Parasitism has evolved in so many taxonomic groups from plants to higher vertebrates.
(4) Endoparasites have greatly complex morphological and anatomical features along with high reproductive potential.
Find out the incorrect statement
(1) Under a particular set of selection pressure, organisms evolve towards the most efficient reproductive strategy.
(2) In ammensalisms, one species is harmed whereas other is not benefitted.
(3) Some species of insects and frogs are cryptically coloured (camouflaged) to avoid being detected easily by the predator.
(4) Biological control methods adopted in agricultural pest control are based on the ability of the prey to regulate predator population.
Through resource partitioning
(1) Two species can compete for the same prey
(2) Slight variation in niche allow closely related species to co-exist in the same habitat.
(3) Competitive exclusion results in the success of the superior species.
(4) Two species undergo character displacement that allows them to compete.
The association between clown fish and sea anemone is the same as between
(1) Egret and grazing cattle
(2) Cuckoo and crow
(3) Fig and wasp
(4) Cuscuta and hedge plant
For the defense against predators, butterflies become highly distasteful due to having certain chemical in their bodies. What is the source of the chemical?
(1) The butterflies have genes for synthesis of this chemical in its each and every cell.
(2) The butterfly acquires this chemical during its caterpillar stage by feeding on a poisonous plant.
(3) The chemical accumulates in the cells of butterfly when it feeds on another insect
(4) The butterfly secretes the chemical
Predation, parasitism and commensalism share a common characteristic, that is
(1) Both the interacting species are benefitted.
(2) Interacting species live closely together.
(3) One of the species is benefitted while other is harmed.
(4) Both the species belong to the same taxonomic group.
Which of the following is not correct?
(1) More recent studies do not support gross generalisations about competition as suggested in Gause’s competitive exclusion principle.
(2) More recent studies do not rule out the occurrence of interspecific competition in nature.
(3) More recent studies point out that species facing competition might evolve mechanisms that promote co-existence rather than exclusion.
(4) Gause’s competitive exclusion principle is effective when resources are in excess.
Secondary productivity means
(1) Rate of formation of organic matter by producer.
(2) Rate of formation of new organic matter by consumers.
(3) Rate of formation of inorganic matter by producer
(4) Rate of formation of inorganic matter by consumer
Match Column-I with Column-II and select the correct option
Column-I Column-II
a. Fragmentation (i) Break down of detritus into smaller particles
b. Leaching (ii) Precipitation of water soluble nutrients as unavailable salt
c. Catabolism (iii) Degradation of detritus by bacterial and fungal enzymes
1. a. — (i) ; b. — (ii) ; c. — (iii)
2. a. — (ii) ; b. — (iii) ; c. — (i)
3. a. — (iii) ; b. — (ii) ; c. — (i)
4. a. — (iii) ; b. — (i) ; c. — (ii)
Humus is degraded by some microbes and release of inorganic nutrients occur by the process known as
(1) Fragmentation
(2) Leaching
(3) Humidification
(4) Mineralisation
Following are the two main structural features of an ecosystem
a. Species composition
b. Energy flow
c. Decomposition
d. Stratification
1. a. and d. 2. b. and c.
3. Only a. 4. b. and d.
Cycling of material in ecosystem is entirely dependent upon
(1) Producers and consumers.
(2) Primary producers and herbivores.
(3) Herbivores and carnivores
(4) Producers and decomposers.
The process of decomposition is in the following order
(1) CatabolismFragmentationLeachingHumificationMineralization
(2) CatabolismFragmentationHumificationLeachingMineralization
(3) FragmentationHumificationCatabolismLeachingMineralization
(4) FragmentationLeachingCatabolismHumificationMineralization
Net primary productivity
(1) For oceans is 60% of biosphere productivity
(2) Depends upon photosynthetic capacity of producers.
(3) Remains constant in temperate area throughout the year
(4) Is limited by light in marine habitats only
Most important climatic factors that regulate the rate of decomposition are
(1) Temperature and soil moisture.
(2) Soil pH and aeration
(3) Aeration and temperature
(4) Moisture and soil Ph
Gross primary productivity is
(1) Rate of which organic molecules are formed in an autotroph.
(2) Rate at which organic molecules are used up by an autotroph
(3) Storage of organic molecules in the body of an autotroph
(4) Rate at which organic molecules are transferred to next higher trophic level.
Which law is obeyed during energy flow in an ecosystem?
(1) First law of thermodynamics
(2) Second law of thermodynamics
(3) Third law of thermodynamics
(4) Both 1. and 2.
Find the incorrect statement
(1) In terrestrial ecosystem larger fraction of energy flow occurs through DFC
(2) In aquatic ecosystem a major conduit for energy flow is GFC
(3) Detritus food chain is not at all connected with grazing food chain at any level.
(4) Natural interconnection of food chain makes it a food web
A graphical representation known as ______is required to express food or energy relationship between organisms at different trophic levels.
(1) Ecological pyramid
(2) Standing state
(3) Ecological quadrilateral
(4) Standing crop
Find the correct statement
(1) Trophic level represents a functional level, not a species as such
(2) A given species never occupies more than one trophic level in the same ecosystem at the same time
(3) In most ecosystem, producers are less in number and biomass than the herbivores
(4) Pyramid of energy can never be upright
A food web
(1) Increases variety of food at each trophic level
(2) Delicately balances the inter-relations among organisms.
(3) Decreases variety of food but increases quantity of food at each trophic level
(4) Increases variety as well as quantity of food at each trophic level
Orderly and sequential changes in communities, parallel with the changes in the physical environment is known as
(1) Ecological succession.
(2) Extinction.
(3) Divergent evolution.
(4) Convergent evolution.
The entire sequence of communities that successively, change in a given area are called
Find out the total number of true statements from the following:
a. Primary succession is a very slow process, taking thousands of years for the climax to be reached.
b. All succession whether taking place in water or land, proceeds to a similar climax community, the mesic.
c. As succession proceeds, the number and types of animals and decomposers also change
d. Saprophytes are not given any place in the ecological pyramids even though they play a vital
role in the ecosystem.
1. One 2. Two
3. Three 4. Four
Amount of nutrients present in soil at any given time is referred as
Which of the following majorly regulates the amount of carbon in the atmosphere?
(1) Respiratory activities of the producers and consumers
(2) Decomposers
(3) Burning of fossil fuel
(4) Oceanic reservoir
What price tag did Robert Constanza put for fundamental ecosystem services?
Out of the total cost of various ecosystem services, the soil account for __________%, recreation and nutrient cycling constitute less than ___________% and climate regulation and habitat for wildlife account for about __________% each.
The two major differences between carbon and phosphorus cycles are
a. Atmospheric inputs of phosphorus through rainfall are much smaller than carbon inputs
b. Atmospheric inputs of phosphorus through rainfall are much greater than carbon inputs
c. Gaseous exchange of phosphorus between organism and environment is not negligible
d. Gaseous exchange of phosphorus between organism and environment is negligible
1. a. and b. 2. b. and c.
3. a. and d. 4. a. and c.
Read the given statements and choose the right option
a. The amount of living matter (biomass) present at every tropical level is called standing crop
b. Saprophytes do not form part of ecological pyramids
c. Ecological pyramid does not account the same species belonging to two or more trophic levels
d. Humus is reservoir of nutrients
1. All are correct
2. All are incorrect
3. Only d. is correct
4. a, d and c are correct
A major advantage of using YAC as a cloning vector over the plasmids is that :
1. it can replicate independently
2. it can be selected easily
3. it can accommodate larger inserts
4. it has multiple cloning sites
What is the problem in providing a bone marrow transplant to a patient of SCID as a treatment?
1. rejection of the graft by the recipient
2. graft-versus-host disease
3. cardiovascular failure
4. anaphylaxis
Genetic engineering of nitrogen fixation is made difficult by the fact that it :
| 1. | is a complex process involving many enzymes |
| 2. | is a process that is partly anaerobic |
| 3. | involves about 15 genes (the Nif Genes) |
| 4. | All of these |
Which of the following genetically modified product is used to dissolve blood clots in patients of coronary Thrombotic stroke?
(1) streptokinase
(2) urokinase
(3) tPA
(4) heparin
The genetically engineered vaccine for the prevention of hepatitis B is made with the help of the microbe :
| 1. | E.coli | 2. | Agrobacterium |
| 3. | Saccharomyces | 4. | Pseudomonas |
The advantage of using DNA polymerases from thermophilic organisms in PCR is that:
| 1. | the DNA polymerases of these bacteria are much faster than those from other organisms |
| 2. | the DNA polymerases of these bacteria can withstand the high temperatures needed to denature the DNA strands |
| 3. | the DNA polymerases of these bacteria never make mistakes while replicating DNA |
| 4. | all of the above |
First discovered, Type II restriction endonuclease was
(1) Hind I
(2) Eco K
(3) Hind II
(4) EcoRI
Which of the following genetic disease would be amenable to genetic engineering?
| 1. | Down's syndrome | 2. | Muscular dystrophy |
| 3. | Cystic fibrosis | 4. | Cri du Chat |
Using recombinant DNA technology, genes from a donor cell can be implanted into a bacterium for DNA replication and protein synthesis. The kind of cell(s) that can be used as gene donors in this technology is/are:
1. Bacteria only
2. Either yeast or bacteria only
3. Eukaryotic cells only
4. Any kind of cell
A gene carried by recombinant DNA is cloned when:
1. Its host bacterium divides by binary fission
2. It is transcribed
3. It is fragmented by restriction enzymes
4. It is hybridized
A piece of nucleic acid used to find a gene, by forming a hybrid with it, is called a
1. Probe
2. Vector
3. Restriction sequence
4. Retrovirus
In situ hybridization is used to :
| 1. | Remove mutations from a chromosome |
| 2. | Remove plasmids from a cell |
| 3. | Clone a gene |
| 4. | Locate a gene on a chromosome |
Restriction enzymes are synthesized by
(1) Bacteria only
(2) Either yeast or bacteria only
(3) Eukaryotic cells, only
(4) All kind of cell
A bacterium adds methyl group to its DNA, by a process known as modification, in order to:
| 1. | Clone its DNA |
| 2. | Turn its genes on |
| 3. | Transcribe many genes simultaneously |
| 4. | Protect DNA from its own restriction enzymes |
A fragment of DNA, cut by a restriction enzyme, forms bonds with other DNA molecules that have
1. Been fragmented by the same restriction enzyme
2. Sticky ends
3. Plasmid component
4. Attached methyl group
Hybridization of mRNA with DNA can reveal which segment of the DNA are introns and which segment are exons. When the magnification is high enough to see the molecule, the introns appear as
1. Single stranded loops in a double stranded molecule
2. Separate fragments
3. Triple stranded nucleic acid molecules
4. Barr bodies
Which of the following primers would allow copying of the single-stranded DNA sequence 5' ATGCCTAGGTC?
| 1. | 5' TACGG | 2. | 5' CTGGA |
| 3. | 5' GACCT | 4. | 5' GGCAT |
The "Southern" technique involves:
| 1. | the detection of DNA fragments on membranes by specific radioactive antibodies. |
| 2. | the detection of proteins on membranes using a radioactive DNA probe. |
| 3. | the detection of DNA fragments on membranes by a radioactive DNA probe. |
| 4. | the detection of proteins on membranes using specific radioactive antibodies. |
Which of the following tools of recombinant DNA technology is INCORRECTLY paired with one of its uses?
| 1. | restriction endonuclease - production of DNA fragments for gene cloning. |
| 2. | DNA polymerase - copies DNA sequences to create primers in the polymerase chain reaction. |
| 3. | reverse transcriptase - production of cDNA from mRNA. |
| 4. | electrophoresis - RLFP analysis. |
DNA from a eukaryotic organism is digested with a restriction endonuclease and the resulting fragments are cloned into a plasmid vector. Bacteria transformed by these plasmids collectively contain all of the genes of the organism. This culture of bacteria is referred to as :
| 1. | restriction map | 2. | RFLP profile |
| 3. | F' factor | 4. | library |
Which of the following is not part of the normal process of cloning recombinant DNA in bacteria?
| 1. | restriction endonuclease digestion of cellular and plasmid DNAs. |
| 2. | production of recombinant DNA using DNA ligase and a mixture of digested cellular and plasmid DNAs. |
| 3. | separation of recombinant DNAs by electrophoresis using the Southern technique to determine where the desired recombinant migrates. |
| 4. | transformation of bacteria by the recombinant DNA plasmids and selection using ampicillin. |
A key feature of insertional mutagenesis for the identification of plasmids containing recombinant DNA is:
| 1. | the production of nutritional auxotrophs. |
| 2. | the DNA sequencing of recombinant plasmids. |
| 3. | the production of restriction endonuclease maps of recombinant plasmids. |
| 4. | the disruption of a gene on the plasmid by the inserted recombinant DNA. |
The first drug produced using recombinant DNA technology is used to treat:
| 1. | haemophilia | 2. | dwarfism |
| 3. | immunodeficiency | 4. | diabetes |
Which gene transfer technique involves the use of a fatty bubble to carry a gene into a somatic cell?
1. electroporation
2. liposome transfer
3. microinjection
4. particle bombardment
What type of probe would you use for a Western blot experiment?
| 1. | A known DNA sequence. | 2. | An RNA molecule. |
| 3. | A purified protein. | 4. | An antibody. |
Prior to the production of recombinant human insulin, diabetic patients could use insulin isolated from cattle. Which of the listed items would not be a drawback to this procedure?
| 1. | Insulin from cattle is slightly different from human insulin, potentially leading to allergic reactions. |
| 2. | Members of some religions consider cattle sacred and thus are unable to use insulin isolated from cattle. |
| 3. | Infectious diseases may be transferred in the isolated insulin. |
| 4. | Despite the relatively stable supply of cattle pancreases from slaughterhouses, insulin isolated from cattle is still relatively expensive and is in, somewhat, limited supply. |
Bacteria that are unable to synthesize chemicals that allow the formation of ice crystals have been engineered. Commercial fruit farmers can spray this bacterial strain on their fruit plants, with the idea that they will compete with unmodified bacteria and reduce frost damage. This is an example of:
| 1. | Bioremediation | 2. | Biological control |
| 3. | Biodegradation | 4. | Bioeradication |
In gene knockout technology:
| 1. | A gene is inserted by nonhomologous recombination. |
| 2. | A mutant gene is replaced by a functional copy. |
| 3. | A functional gene is replaced by a mutant copy. |
| 4. | A functional gene is inserted in addition to the mutant copy. |
Which of the following explains why production of transgenic plants is easier than production of transgenic animals?
| 1. | Plant cells can grow in cell culture. |
| 2. | Plant cells have a lower number of potentially lethal genes. |
| 3. | Plant cells are totipotent. |
| 4. | Production of mutant plants poses less ethical dilemmas than production of mutant animals. |
What process causes shortening of telomeres during cell division?
1. DNA synthesis.
2. Exonuclease removal of primers.
3. Ligation of DNA fragments.
4. Telomerase activity.
Which of these uses of transgenic technology is not directly relevant to agricultural applications?
| 1. | Production of a pharmaceutical protein in goat's milk. |
| 2. | Production of salmon that grow all year round instead of just in warm weather. |
| 3. | Production of soybean plants that are resistant to herbicides. |
| 4. | Production of "super mice" that carry the human growth hormone gene. |
Which of these is an accurate description of gene therapy?
| 1. | A form of site directed mutagenesis. |
| 2. | Introduction of cloned genes into living cells to cure disease. |
| 3. | Cloning of a disease gene in order to understand the disease. |
| 4. | All of these. |
In the first gene cloning experiment:
| 1. | Researchers successfully identified a human gene responsible for the disease. |
| 2. | Researchers successfully inserted a gene for kanamycin resistance into a plasmid vector. |
| 3. | Researchers demonstrated that many different DNA fragments could insert into a plasmid vector |
| 4. | Researchers produced a strain of bacteriophage with an increased ability to infect E. coli. |
An important limitation to the use of Agrobacterium tumefaciens is that it can not:
1. infect dicots
2. be genetically modified
3. be cultured on a nutrient medium
4. infect crop plants such as wheat and corn
A danger of genetic engineering is that the genetically modified bacterium can escape the lab and infect humans. This possibility can best be prevented by:
| 1. | selecting a mutant form of the bacteria |
| 2. | selecting a live attenuated bacterium |
| 3. | application of stringent asepsis in the lab |
| 4. | prophylactically immunising the persons working in the lab |
If you are using a vector with lacZ as a screenable marker, which of the following bacterial strains would you want to use as host cells?
1. lacO-
2. lacI-
3. lacZ-
4. Any of these would work.
Which of the enzymes whose recognition sites are shown here would produce sticky ends compatible with BstAUI (5'-G|GTACC-3')?
1. Bcl I (5'-T|GATCA-3')
2. Sun I (5'-C|GTACG-3')
3. Hind III (5'-A|AGCTT-3')
4. Kpn I (5'-GGTAC|C-3')
Which of the following would NOT be a useful selectable marker?
| 1. | A gene encoding a protein that degrades the antibiotic ampicillin. |
| 2. | A gene encoding a protein that allows the cell to synthesize histidine. |
| 3. | A gene encoding a protein that is an essential structural component of the cell. |
| 4. | All of these are useful selectable markers. |
Diseases amenable to treatment by gene therapy:
| 1. | Must be relatively mild. |
| 2. | Must be caused by a defect in cells that are clinically accessible. |
| 3. | Must have other types of treatments available in case gene therapy fails. |
| 4. | Must lower the humoral and cell mediated immune responses |
One possible use of transgenic plants is in the production of human proteins, such as vaccines. Which of the following is a possible hindrance that must be overcome?
| 1. | prevention of transmission of plant allergens to the vaccine recipients |
| 2. | prevention of vaccine-containing plants being consumed by insects |
| 3. | use of plant cells to translate non-plant derived mRNA |
| 4. | inability of the human digestive system to accept plant-derived protein |
Which of the following is one of the technical reasons why gene therapy is problematic?
| 1. | Most cells with an engineered gene do not produce gene products. |
| 2. | Most cells with engineered genes overwhelm other cells in a tissue. |
| 3. | Cells with transferred genes are unlikely to replicate. |
| 4. | Transferred genes may not have appropriately controlled activity. |
The DNA profiles below represent four different individuals. Which of the following statements is consistent with the results?
1. B is the child of A and C.
2. C is the child of A and B.
3. D is the child of B and C.
4. A is the child of B and C.
Dideoxyribonucleotide chain-termination is a method of
1. cloning DNA.
2. sequencing DNA.
3. digesting DNA.
4. synthesizing DNA.
What is the most logical sequence of steps for splicing foreign DNA into a plasmid and inserting the plasmid into a bacterium?
| I. | Transform bacteria with recombinant DNA molecule. |
| II. | Cut the plasmid DNA using restriction enzymes. |
| III. | Extract plasmid DNA from bacterial cells. |
| IV. | Hydrogen-bond the plasmid DNA to non-plasmid DNA fragments. |
| V. | Use ligase to seal plasmid DNA to non-plasmid DNA. |
1. I, II, IV, III, V
2. II, III, V, IV, I
3. III, II, IV, V, I
4. III, IV, V, I, II
How many formula units are there in the unit cell of Sodium Chloride?
1. 2
2. 4
3. 6
4. 8
The most unsymmetrical and symmetrical system are respectively
1. Tetragonal, Cubic
2. Triclinic, Cubic
3. Rhombohedral, Hexagonal
4. Orthorhombic, Cubic
In a compound atoms of element Y form CCP lattice and those of element X occupy 2/3rd of tetrahedral voids. The formula of compound will be
1. X3Y4
2. X2Y3
3. X2Y
4. X4Y3
The edge of unit cell of fcc of Xe crystal is 620 pm. The radius of Xe atom is
1. 219.23 pm
2. 235.16 pm
3. 189.37 pm
4. 319.23 pm
Schottky defect occurs mainly in electrovalent compound where
1. Positive ions have larger size
2. Negative ions have small size
3. Positive and negative ions are of similar size
4. Positive ions are big and negative ions are small
The appearance of colour in solid alkali metal halides is generally due to
1. Frenkel defect
2. Interstitial defect
3. F - Centres
4. Schottky Defect
The edge length of fcc unit cell of NaCl is 508 pm. If the radius of cation is 110 pm then radius of anion is
1. 114 pm
2. 249 pm
3. 288 pm
4. 398 pm
An element (Atomic mass = 50 g/mol) having fcc stucture has unit cell edge length 400 pm. The density of element is
1. 5.188 g/cc
2. 10.376 g/cc
3. 2.56 g/cc
4. 1.2 g/cc
In a crystalline solid, anion B are arranged in cubic close packing and cation A are equally distributed between octahedral and tetrahedral voids. If all the octahedral voids are occupied, the formula for the solid is
1. AB
2. AB2
3. A2B
4. A2B3
In a FCC lattice, atom A occupies the corner positions and atom B occupies the face centred positions. If one atom of B is missing from one of the face centred points, the formula of the compound is
1. A2B
2. AB2
3. AB
4. A2B5
In HCP arrangement, the co-ordination number is
1. 6
2. 12
3. 8
4. 10
A binary solid has a structure with B- ion constituting the lattice and A+ ion occupying 25% tetrahedral holes. The formula of solid is
1. AB
2. A2B
3. AB2
4. AB4
A solid has a bcc structure. If the distance of closest approach between the two atoms 1.73 . The edge length of the cell is
1. 200 pm
2.
3. 142.2 pm
4. 400 pm
A metal crystallises as body centred cubic lattice with the edge length of unit cell equal to 0.304 nm. If the molar mass of the metal is 50.3 g mol-1, its density is
1. 5.945 g cm-3
2. 2.9725 g cm-3
3. 8.9175 g cm-3
4. 4.458 g cm-3
The edge length of the unit cell of NaCl crystal lattice is 5.623 , density is 2.16 g cm-3 and the molar mass of NaCl is 58.5 g mol-1. The number of formula units per unit cell is
1. 4
2. 3
3. 1
4. 2
Analysis shows that an oxide ore of nickel has formula The percentage of nickel as ions is nearly
1. 2
2. 96
3. 4
4. 98
Perovskite is mineral containing Calcium, Oxygen and Titanium, in which oxygen atoms are at the face centres, calcium atoms are at the corners and titanium atoms are at the centre of the cube. Oxidation number of titanium in the mineral is
1. +2
2. +3
3. +4
4. +3/4
If the distance between Na+ and Cl- ions in sodium chloride crystal is X pm, the length of the edge of the unit cell is
1. 4X pm
2. X/4 pm
3. X/2 pm
4. 2X pm
In a crystal A atoms are present at all the corners and face centres while B atoms at all the edge centres and body centre of cubic unit cell. If the atoms from one of the body diagonals are removed then what will be the simplest composition of unit cell?
The number of cationic vacancies per mole in NaCl crystal, If it is doped with 10-3 mole % GaCl3 (ionic salt), are
Which has highest freezing point?
1. 0.01 M NaCl
2. 0.05 M Urea
3. 0.01 M MgCl2
4. 0.02 M NaCl
for is 1.86 k/m. Therefore, the freezing point of 0.1 M NaCl solution in water is expected to be
When mercuric iodide is added to the aqueous solution of potassium iodide is
1. Freezing point is raised
2. Feezing point is lowered
3. Freezing point does not change
4. Boiling point does not change
A 5% solution of cane sugar (molar mass = 342) is isotonic with 1% of a solution of an unknown solute. The molar mass of unknown solute is
1. 136.2
2. 171.2
3. 68.4
4. 34.2
What is the freezing point of a solution contains 8.1 gm HBr in 100 gm water assuming the acid to be 90% ionized? (Kf for H2O = 1.86 k/m)
Which of the following solution has the highest equivalent conductance?
1. 0.01 M NaCl
2. 0.05 M NaCl
3. 0.005 M NaCl
4. 0.02 M NaCl
The resistance of 0.01 N solution of an electrolyte AB at 328K is 100 ohm. The specific conductance of solution is (cell constant = 1 cm-1)
1. 100 ohm
2. 10-2 ohm
3. 10-2 ohm-1cm-1
4. 102 ohm-cm
The increase in the molar conductivity of HCl with dilution is due to
1. Increase in the self ionisation of water
2. Hydrolysis of HCl
3. Decrease in the self ionisation of water
4. Decrease in the interionic forces
A solution of in water is electrolysed using pt electrodes. The products at the cathode and anode are respectively
Given that
The most reactive metal which displaces other metals from their salts in solution is
1. Ag
2. Cu
3. Co
4. Zn
The emf of the cell is 0.83V. The emf of this cell could be increased by
1. increasing the concentration of Tl+ ions
2. increasing the concentration of Cu2+ ions
3. increasing the concentration of both
4. None of the above
For a reaction has been found to be . The is
1. 0.354 V
2. 0.708 V
3. 0.0098 V
4. 1.36 V
The solution of nickel sulphate in which nickel rod dipped is diluted to 10 times. The potential of nickel
1. Decreases by 60 mV
2. Increases by 30 mV
3. Decreases by 30 mV
4. Decreases by 60 mV
Two electrodes are fitted in conductance cell 1.5 cm apart while the area of cross section of each electrode is 0.75 cm2. The cell constant is
1. 1.0125 cm
2. 0.5 cm
3. 2.0 cm-1
4. 0.2 cm-1
Pick out the incorrect statement
1. Equivalent conductance increases with dilution
2. Molar conductance increases with dilution
3. Specific conductance increases with dilution
4. Specific resistance increases with dilution
Aluminium phosphate is 100% ionized in 0.01m aqueous solution. Hence, is
1. 0.01 2. 0.015
3. 0.0175 4. 0.02
60 gm of urea is dissolved in 1100 gm solution. To keep as 1 mol/kg, amount of water separated in the form of ice will be
1. 40 gm 2. 60 gm
3. 100 gm 4. 200 gm
Which solution has the highest osmotic pressure?
1. 0.1 M NaCl 2. 0.5 M Urea
3. 0.1 M MgCl2 4. 0.2 M NaCl
An ideal solution is made by mixing 2 moles of benzene (Po = 266 mm) and 3 moles of another liquid (Po = 236 mm). The total vapour pressure of the solution at the same temperature would be
1. 502 mm 2. 248 mm
3. 600 mm 4. 258 mm
A binary solution of ethanol and n-heptane is an example of
1. Ideal solution
2. Non-ideal solution with +ve deviation
3. Non-ideal solution with -ve deviation
4. Unpredictable behaviour
0.5 M solution of urea is isotonic with
1. 0.5 M NaCl solution
2. 0.5 M sugar solution
3. 0.5 M BaCl2 solution
4. 0.5 M solution benzoic acid in benzene
The value of for NH4Cl , NaOH and NaCl are respectively 149.74, 248.1 and
126.4 ohm-1cm2equiv-1. The value of for NH4OH is
1. 317.44
2. 271.44
3. 71.44
4. It cannot be calculated from the data given
In passing 3 faraday of electricity through the three electrolytic cells connected in series containing Ag+, Cu2+ and Al3+ respectively. The molar ratio in which the three metal ions are liberated at the electrode is
1. 1:2:3 2. 3:2:1
3. 6:3:2 4. 3:4:2
Given that
Predict which of the following is true
1. I- ions will be able to reduce bromine
2. Br- ions will be able to reduce iodine
3. Iodine will be able to reduce bromide ions
4. Bromine will be able to reduce iodide ions
What is the EMF of the cell?
Zn(s) | Zn+2 (0.1M) || Sn+2 (0.001M) | Sn(s)
Given
1. 0.62V 2. 0.56V
3. 1.12V 4. .31V
At what altitude, will the acceleration due to gravity be 25% of that at the earth's surface (given radius of the earth is R)?
1. R/4
2. R
3. 3R/8
4. R/2
At what distance from the centre of the moon is the point at which strength of the resultant of earth's and moon's gravitational field is equal to zero. The earth's mass is 81 times that of the moon and the distance between centres of the earth and that of the moon is 60R where R is the radius of the earth.
1. 6 R
2. 4 R
3. 3 R
4. 5 R
Two masses m1 and m2 are initially at rest and are separated by a very large distance. If the masses approach each other subsequently, due to gravitational attraction between them, their relative velocity of approach at a separation distance of d is :
1.
2.
3.
4.
A spherical hole of radius R/2 is excavated from the asteroid of mass M as shown in fig. The gravitational acceleration at a point on the surface of the asteroid just above the excavation is :
1. GM/R2
2. GM/2R2
3. GM/8R2
4. 7GM/8R2
If the radius of the earth be increased by a factor of 5, by what factor its density be changed to keep the value of g the same?
1. 1/25
2. 1/5
3.
4. 5
Two springs of force constant k and 2k are connected to a mass as shown below. The frequency of oscillation of the mass is :
An infinite number of masses, each of one kg are placed on the +ve X-axis at 1m, 2m, 4m............from the origin. The magnitude of the gravitational field at origin due to this distribution of masses is:
1. 2G
2.
3.
4.
A satellite revolves in the geostationary orbit but in a direction east to west. The time interval between its successive passing about a point on the equator is :
1. 48 hrs
2. 24 hrs
3. 12 hrs
4. never
A satellite of the earth is revolving in a circular orbit with a uniform velocity v. If the gravitational force suddenly disappears, the satellite will
1. Continue to move the same velocity in the same orbit.
2. move tangentially to the original orbit with velocity v.
3. fall down with increasing velocity.
4. come to a stop somewhere in its original orbit.
At what height above the earth's surface does the acceleration due to gravity fall to 1% of its value at the earth's surface?
1. 9R
2. 10R
3. 99R
4. 100R
Two concentric shells of uniform density of mass M1 and M2 are situated as shown in the figure. The forces experienced by a particle of mass m when placed at position A, B, C respectively are (given OA = p, OB = q and OC = r)
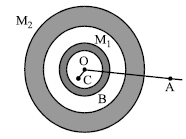
1.
2.
3.
4.
A newly discovered planet has a density eight times the density of the earth and a radius twice the radius of the earth. The time taken by 2 kg mass to fall freely through a distance S near the surface of the earth is 1 second. Then the time taken for a 4 kg mass to fall freely through the same distance S near the surface of the new planet is :
1. 0.25 sec.
2. 0.5 sec.
3. 1 sec.
4. 4 sec.
The radius of a planet is R. A satellite revolves around it in a circle of radius r with angular velocity . The acceleration due to the gravity on the planet's surface is :
1.
2.
3.
4.
The mass and diameter of a planet are twice those of earth. What will be the period of oscillation of pendulum on this planet if it is a seconds pendulum on earth?
1.
2.
3.
4.
A simple pendulum performs simple harmonic motion about x = 0 with an amplitude a, and time period T. The speed of pendulum at x = a/2 will be :
A particle starts from rest at a distance R from the centre and along the axis of a fixed ring of radius R & mass M. Its velocity at the centre of ring is :
1.
2.
3.
4.
A spherical uniform planet is rotating about its axis. The velocity of a point on its equator is V. Due to the rotation of the planet about its axis, the acceleration due to gravity g at equator is 1/2 of g at poles. The escape velocity of a particle on the planet in terms of V.
1. Ve = 2V
2. Ve = V
3. Ve = V/2
4. Ve = V
Two point masses of mass 4m and m respectively separated by d distance are revolving under mutual force of attraction. Ratio of their kinetic energies will be :
1. 1 : 4
2. 1 : 5
3. 1 : 1
4. 1 : 2
Two planets A and B have the same material density. If the radius of A is twice that of B, then the ratio of the escape velocity is -
1. 2
2.
3.
4.
A simple harmonic motion having an amplitude A and time period T is represented by the equation:
Then the values of A (in m ) and T (in sec ) are:
1. A = 5; T = 2
2. A = 10; T = 1
3. A = 5; T = 1
4. A = 10; T = 2
The displacement of a body executing SHM is given by x= A sin (2t + /3). The first time from t = 0 when the velocity is maximum is :
1. 0.33 sec
2. 0.16 sec
3. 0.25 sec
4. 0.5 sec
The maximum acceleration of a particle in SHM is made two times keeping the maximum speed to be constant. It is possible when :
1. amplitude of oscillation is double while the frequency remains constant
2. amplitude is doubled while frequency is halved
3. frequency is doubled while the amplitude is halved
4. frequency is doubled while amplitude remains constant
The potential energy of a simple harmonic oscillator of mass 2 kg in its mean position is 5J. If its total energy is 9J and its amplitude is 0.01 m, its time period would be :
1.
2.
3.
4.
A plank with a small block on top of it is undergoing vertical SHM. Its period is 2 sec. The minimum amplitude at which the block will separate from plank is :
1.
2.
3.
4.
The time period of a particle executing SHM is 8 sec. At t = 0 it is at the mean position. The ratio of the distance covered by the particle in the 1st second to the 2nd second is :
1.
2.
3.
4.
The angular velocity of motion whose equation is is : (y = displacement and t = time) -
1.
2.
3.
4.
Two particles are in SHM in a straight line. Amplitude A and time period T of both the particles are equal. At time t = 0, one particle is at displacement Y1 = + A and the other at Y2 = -A/2, and they are approaching towards each other. After what time they cross each other?
1. T/3
2. T/4
3. 5T/6
4. T/6
Two particles execute SHM of the same amplitude of \(20\) cm with the same time period along the same line about the same equilibrium position. The maximum distance between the two is \(20\) cm. Their phase difference in radians is:
1. \(\frac{2\pi}{3}\)
2. \(\frac{\pi}{2}\)
3. \(\frac{\pi}{3}\)
4. \(\frac{\pi}{4}\)
Two particles P and Q describe simple harmonic motions of the same period, the same amplitude, along the same line about the same equilibrium position O. When P and Q are on opposite sides of O at the same distance from O, they have the same speed of 1.2 m/s in the same direction. When their displacements are the same, they have the same speed of 1.6 m/s in opposite directions. The maximum velocity in m/s of either particle is
1. 2.8
2. 2.5
3. 2.4
4. 2
A particle performs SHM with a period T and amplitude a. The mean velocity of the particle over the time interval during which it travels a distance a/2 from the extreme position is :
1. a/T
2. 2a/T
3. 3a/T
4. a/2T
If a spring has a time period T and is cut into n equal parts, then the time period of each part will be :
A graph of the square of the velocity against the square of the acceleration of a given simple harmonic motion is -
A simple pendulum is made of a body which is a hollow sphere containing mercury suspended by means of a wire. If a little mercury is drained off, the period of pendulum will :
1. Remain unchanged
2. Increase
3. Decrease
4. Become erratic
If a hole is bored along the diameter of the earth and a stone is dropped into the hole :
1. The stone reaches the centre of the earth and stops there
2. The stone reaches the other side of the earth and stops there
3. The stone executes simple harmonic motion about the centre of the earth
4. The stone reaches the other side of the earth and escapes into space.
Choose the incorrect answer.
When the point of suspension of the pendulum is moved its period of oscillation -
1. Decreases when it moves vertically upwards with acceleration a
2. Decreases when it moves vertically downwards with acceleation less than g
3. Increases when it moves horizontally with acceleration a
4. Decreases when it moves horizontally with acceleration a
A pendulum suspended from the ceiling of a train has period T when the train is at rest. When the train is accelerating with a uniform acceleration a, the period of oscillation will
1. Increase
2. Decrease
3. Remained unaffected
4. Become infinite
The work done by a string of simple pendulum during one complete oscillation is equal to :
1. Total energy of pendulum
2. Kinetic energy of pendulum
3. Potential energy of pendulum
4. Zero
A particle of mass m is hanging vertically by an ideal spring of force constant K. If the mass is made to oscillate vertically, its total energy is :
1. Maximum at extreme position
2. Maximum at mean position
3. Minimum at mean position
4. Same at all position
Length of the second's pendulum is decreased by 1%, then the gain or loss in time per day will be :
1. 432 sec
2. 4.40 sec
3. 44 sec
4. 0.44 sec
A body of mass m is suspended from a rubber cord with force constant K. The maximum distance over which the body can be pulled down for the body's oscillation to remain harmonic is :
1.
2.
3.
4.
A particle executes simple harmonic motion along a straight line with an amplitude A. The potential energy is maximum when the displacement is
1.
2. Zero
3.
4.
The earth moves around the sun in an elliptical orbit as shown in the figure. The ratio . The ratio of the speeds of the earth at B and at A is :
A body is released from a point at a distance r from the centre of the earth. If R is the radius of earth and r > R, then the velocity of the body at the time of striking the earth will be:
A straight rod of length L extends from x = a to x = L+a. The gravitational force, it exerts on a point mass m at x = 0, if mass per unit length is is :
(a)
(b)
(c)
(d)
A body performs SHM. Its kinetic energy K varies with time T as indicated in the graph :
Tobacco plant has been made resistant to the tobacco mosaic virus by :
| 1. | introducing T-DNA of Agrobacterium |
| 2. | introducing a gene for virus coat protein |
| 3. | introducing meristematic activity in somatic cells |
| 4. | applying newer generation pesticides to the crops |