Natural ageing of a lake by biological enrichment of its water is called
1. Biomagnification
2. Eutrophication
3. Algal blooms
4. Toxification
Catalytic converter of automobiles converts unburnt hydrocarbons into
1.
2.
3.
4.
Biomagnification of DDT in aquatic food chain leads to high concentration of DDT in fish eating birds. It results in
| 1 | Disturbance in calcium metabolism in birds. |
| 2 | Thinning of egg shell and their premature breaking. |
| 3 | Decline in birds population |
1. (1) and (2) only
2. (2) and (3) only
3. (1) and (3) only
4. All (1), (2) and (3)
Common cold is not cured by antibiotics
because it is
1. caused by a Gram-negaive bacterium
2. not an infectious disease
3. viral disease
4. caused by a Gram-positive bacterium
Which one of the following statements about
mycoplasma is wrong?
1. They are pleomorphic
2. They are sensitive to Penicillin
3. They cause diseases in plants
4. They are called PPLO
In the following diagram A, B, C and D
represent
1. A - Seta, B - Leaves, C- Rhizoids, D - Capsule
2. A - Leaf, B - Rhizoids, C- Scales, D - Root hair
3. A - Capsule, B - Seta, C- Leaves, D - Rhizoids
4. A - Capsule, B - Foot, C- Leaves, D - Rhizoids
The incorrect match for chlorophyll type is
1. Chlorophyll ‘a’ .... Green algae
2. Chlorophyll ‘d’ ... Diatoms
3. Chlorophyll ‘c’ ... Dinoflagellates and Brown algae
4. Chlorophyll ‘d’ ...Red algae
Protonema is a characteristic feature of
1. Fern
2. Marchantia
3. Moss
4. Cycas
Sweet potato is a modified
1. Rhizoid
2. Stem
3. Adventitious Root
4. Leaf
The diagram given below belongs to -
| 1. | Dicot stem having conjoint and collateral vascular bundles |
| 2. | Monocot stem having conjoint and collateral vascular bundles |
| 3. | Dicot root with polyarch condition |
| 4. | Monocot root with radial vascular bundles |
Hypodermis in dicot stem is composed of
1. Parenchyma
2. Collenchyma
3. Sclerenchyma
4. Both 1. and 3.
Manganese is required in
1. Nucleic acid synthesis
2. Plant cell wall formation
3. Photolysis of water during photosynthesis
4. Chlorophyll synthesis
In which of the following option name is written
correctly?
1. APIS INDICA
2. Trypansoma Gambiense
3. Ficus begalensis
4. Mangifera indica
Pneumatophores are not found in
1. Rhizophora
2. Heritiera
3. Bombax
4. Sonneratia
A land snail, a clam, and an octopus all share
1. a mantle
2. calcareous shell
3. a radula
4. distinct cephalization
Which of the following combinations of phylum and description is incorrect?
| 1. | Echinodermata–branch Bilateria, coelom from archenteron |
| 2. | Nematoda–roundworms, pseudocoelomate |
| 3. | Cnidaria–radial symmetry, polyp and medusa body forms |
| 4. | Porifera - gastrovascular cavity, mouth from blastopore |
Which of the following structures or substances is
incorrectly paired with a tissue ?
1. Haversian system—bone
2. platelets—blood
3. fibroblasts—skeletal muscle
4. chondroitin sulfate—cartilage
The majority of water and salt filtered into Bowman’s capsule is reabsorbed by:
1. the brush border of the transport epithelia of the proximal tubule
2. diffusion from the descending limb of the loop of Henle into the hypertonic interstitial fluid of the medulla
3. active transport across the transport epithelium of the thick upper segment of the ascending limb of the loop of Henle
4. selective secretion and diffusion across the distal tubule
The depolarization of the presynaptic membrane of
an axon directly causes
1. voltage-gated calcium channels in the membrane
to open
2. synaptic vesicles to fuse with the membrane
3. an action potential in the postsynaptic cell
4. the opening of chemically sensitive gates that
allow neurotransmitter to spill into the synaptic
cleft
Which of the following structures or regions is
incorrectly paired with its function?
1. limbic system—screening of information between
the spinal cord and the brain; regulates arousal
and sleep
2. medulla oblongata—homeostatic control center
3. cerebellum—unconscious coordination of
movement and balance
4. corpus callosum—band of fibers connecting left
and right cerebral hemispheres
Which of the following is a true statement about
cardiac muscle cells?
1. They lack an orderly arrangement of actin and
myosin filaments
2. They have less extensive sacroplasmic reticulum
and thus contract more slowly than smooth
muscle cells
3. They are connected by intercalated discs,
through which action potentials spread to all cells
in the heart
4. They have a resting potential more positive than
an action potential threshold
Which of the following reaction is not catalysed by
brush border enzyme?
1.
2.
3.
4.
Which of the following is not a part of female external
genitalia?
1. Birth canal
2. Hymen
3. Clitoris
4. Labia minora
Which of the following structure can not be seen in ovary during post ovolutionary phase?
| (i) | Primary follicle. |
| (ii) | Tertiary follicle. |
| (iii) | Corpus luteum. |
| (iv) | Primary polar body. |
1. (i) and (ii) only
2. (ii) and (iv) only
3. (ii) only
4. (iii) and (iv) only
Which of the following takes maximum duration for its
completion?
1. Meiosis I in primary oocyte.
2. Formation of haploid spermatozoa.
3. Formation of ootid from secondary oocyte.
4. Physiological maturity of spermatozoa.
Ovulation can be delayed by
1. Declining the level of progesterone.
2. Preventing LH surge.
3. Increasing the level of estrogen.
4. Declining the level of FsH.
Odd one out in given barrier method of contraception
| 1. | Nirodh | 2. | Diaphragm |
| 3. | Vaults | 4. | Cervical cap |
Match the Column I with Column II –
|
|
Column-I |
|
Column-II |
|
1. |
Terminalization of chiasmata |
A. |
Zygotene |
|
2. |
Synapsis |
B. |
Diplotene |
|
3. |
Crossing over |
C. |
Metaphase I |
|
4. |
Dissolution of synaptonemal complex |
D. |
Diakinesis |
|
5. |
Best stage for the study of chiasmata |
E. |
Pachytene |
|
6. |
Nuclear membrane and nucleolus disappear |
|
|
|
7. |
Tetrads are arranged on equatorial line |
|
|
1. A – 2, B – 4,5, C – 7, D – 1,6, E – 3
2. A – 2, B – 3, C – 7, D – 1, 4, 6, E – 5
3. A – 2, B – 7, C – 3, D – 1, 4, 5, E – 6
4. A – 2, B – 1, C – 4, D – 5, 3, E – 6
The events shown below occur during different phases –
| A. | Centromere splits, chromatids separate and move to opposite poles. Chromatids are now called chromosome. |
| B. | Chromosomes cluster at opposite poles, decondensation of chromosome & reappearance of NM, GB, ER and nucleolus. |
| C. | Chromosomal replication. |
| D. | Kinetochores attach to spindle fibres and chromosome are arranged at equatorial plate. |
| E. | Spiration of chromosomes/condensation of chromosomal materials. |
Which of the following correctly identifies each of the phases described –
| Interphase | Prophase | Metaphase | Anaphase | Telophase | |
| 1. | C | E | D | A | B |
| 2. | C | D | E | A | B |
| 3. | C | E | D | B | A |
| 4. | C | A | D | E | B |
Choose the correct statements –
| I: | Mitochondria and Chloroplast transfer energy. |
| II: | Mitochondrion is a power-house of cell as it produces most of the cellular ATP. |
| III: | Mitochondria and chloroplast are found in all eukaryotic cells. |
| IV: | Mitochondria are the sites of anaerobic respiration. |
| V: | The matrix of mitochondria posses a single linear DNA, many RNA molecules, 80S ribosomes. |
1. IV and V
2. I, II
3. II, IV and V
4. III and V
Which of the following is the correct sequence / route of the secretory product?
| 1. | ER Vesicles Cis region of GB Trans region of GB Vesicle Plasma membrane. |
| 2. | RER GB Lysosome Nuclear membrane Plasma membrane. |
| 3. | ER Vesicles Trans region of GB Cis region of GB Vesicles Plasma membrane. |
| 4. | Lysosome ER GB Vesicles Cell membrane. |
The following diagram refers to the process of transcription in Eukaryotes. Identify A, B, C and D –
| 1. | A – RNA polymerase II, B – Exon, C – Intron, D – Poly A tail |
| 2. | A – DNA polymerase II, B – Intron, C – Exon, D – Poly A tall |
| 3. | A – RNA polymerase II, B – Intron, C – Exon, D – Poly A tail |
| 4. | A – RNA polymerase II, B – Intron, C – Exon, D – Poly G tail |
Genetic code translates the language of
1. RNA into that of protein
2. RNA into that of DNA
3. amino acids into that of RNA
4. protein into that of DNA.
Wild type Escherichia coli growing on medium having glucose is transferred to lactose containing medium. Which one of the following change will occur?
1. The bacterium stops dividing
2. All operons are induced
3. Lac operon is suppressed
4. Lac operon is induced.
Choose the correct statements –
I. Binding of mRNA to the ribosome is loose and reversible.
II. The ribosome, which is composed of tRNA and proteins, consists of a small and large subunit.
III. Each of the 20 amino acids has a minimum of one specific tRNA molecule.
IV. tRNAMET binds to the first codon (AUG) – this is called the START codon.
V. The second tRNA binds to the second codon so that their attached monosaccharides are next to each other.
VI. The newly formed polypeptide may undergo post-transcriptional modification.
1. I, III and IV
2. II, IV, VI
3. II, V and VI
4. I, V, VI.
A test cross enables one to
1. Determine the viability of cross.
2. Distinguish between homozygous dominant and heterozygous dominant.
3. Determine whether two species can interbreed.
4. Determine the similarities in the DNA of two species.
The experiment shown in the figure has been carried out by Morgan to show the phenomenon of linkage and recombination. If in Cross I, genes are tightly linked and in Cross II, genes are loosely linked then what will be the percentage of recombinants produced in Cross I and Cross II respectively?
1. 98.7% and 62.8%
2. 1.3% and 37.2%
3. 37.2% and 1.3%
4. 62.8% and 98.7%
A. Genetic maps are extensively used as starting point in the sequencing of whole genome as was done in case of the Human genome sequencing project.
B. UV-rays can cause mutation in organisms.
C. Deletion and insertions of base pairs of DNA cause frame-shift mutations.
D. Mendelian disorders may be dominant or recessive.
1. All are correct
2. All are incorrect
3. a, b & d are correct
4. b, c & d are correct.
Which of the following is a false statement?
1. All organisms have evolved similar mechanism to multiply and produce offsprings.
2. Asexual reproduction is uniparental.
3. Sexual reproduction is biparental.
4. In asexual reproduction no fertilization occurs.
Which of the following is false about xenogamy?
1. It is the transfer of pollen grains from anther to stigma of another plant of the same species.
2. It produces genetic variation
3. It is genetically and ecologically (= functionally) cross pollination.
4. It occurs in cleistogamous flowers
Which of the following statements is false?
| I: | Vallisneria and Hydrilla are freshwater plants while sea-grasses (e.g., Zostera) are marine plants. |
| II: | Pollination in water lily / Lotus (Nymphea) and Eichhornia (water hyacinth) takes place by insects. |
| III: | In majority of aquatic plants, flowers emerge above the level of water and are pollinated by insects or wind. |
| IV: | In most of the water-pollinated species, pollen grains are protected from wetting due to presence of a mucilaginous covering. |
| V: | In hydrophilous plants, pollen grains are spherical. |
| 1. | All | 2. | None |
| 3. | V | 4. | IV |
Identify structures A, B, C and phenomena – P1, P2, P3.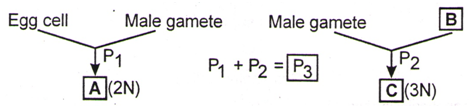
Match the Column A with Column B –
|
|
Column A |
|
Column B |
|
I. |
Many people have deficiencies as they cannot buy fruits & vegetables |
A. |
Single cell proteins |
|
II. |
Crops with higher vitamins, proteins and fats are bred |
B. |
Micropropagation |
|
III. |
Growing microbes as the alternative source of proteins |
C. |
Somaclones |
|
IV. |
Capacity to generate plant from single cell or explant |
D. |
Hidden hunger |
|
V. |
Production of thousand plants through tissue culture |
E. |
Biofortification |
|
VI. |
Genetically identical plants |
F. |
Totipotency. |
1. I – D, II – E, III – F, IV – A, V – B, VI – C
2. I – D, II – E, III – F, IV – A, V – C, VI – B
3. I – D, II – E, III – A, IV – F, V – B, VI – C
4. I – F, II – E, III – A, IV – D, V – B, VI – C.
A. DNA polymorphism plays important role in evolution and speciation
B. 99.9% nucleotides bases are exactly same in all people
C. Chromosome 1 has the fewest genes (231) and Y-chromosomes has most genes (2968)
D. BAC and YAC have been used in Human Genome Project
1. All correct
2. All incorrect
3. A, B, D are correct
4. B, C, D are correct
Similarity between protochordata and hemichordata is
1. Presence of notochord.
2. Exclusively marine.
3. Dorsal hollow and single central nervous system.
4. Exclusively parasitic.
Nucleotide is made up of
1. Heterocyclic compound, Monosaccharide,
Phosphoric acid.
2. Nitrogenous base, hexose sugar, phosphate.
3. Hetrocylic compound and pentose sugar only.
4. Hetrocyclic compound and nucleoside.
NAD and NADP are consider as
| 1. | Apoenzyme and cofactor respectively. |
| 2. | Coenzyme and co-factor respectively. |
| 3. | Both as co-enzyme. |
| 4. | Apoenzyme and holoenzyme respectively |
Common bile duct opens into:
1. Gall bladder
2. Jejunum
3. Hepato-pancreatic duct
4. Stomach
‘Erythroblastosis Foetalis’ occurs when
I. Mother is Rh+
and foetus is Rh–
II. Mother is Rh–
and foetus is Rh+
III. Mother and foetus both are Rh+.
IV. Mother and foetus both are Rh–
1. I & II
2. Only I
3. Only II
4. II, III & IV
In ECG, ventricular contraction occurs
1. Just after P wave and before Q wave
2. Just after Q wave but before T wave
3. In between S-T segment
4. After the end of T wave
Blood present in afferent arteriole and efferent arteriole will differ in
I. Amount of protein.
II. Number of blood cells.
III. Amount of glucose.
IV. Amount of urea.
1. I, II, III & IV
2. III & IV only
3. IV only
4. III only
Which of the following is very effective sedative and
pain killer?
1. Cocaine.
2. Heroin.
3. Morphine.
4. Cannabis extract
Cellular oncogenes can be best explained as
| 1. | Mutated gene. |
| 2. | Present in normal cells. |
| 3. | Present only in the cells of benign tumour. |
| 4. | Genes due to which cells loose the property of contact inhibition. |
There are two opposing views about the origin of modern
man. According to one view, Homo erectus in Asia
were the ancestors of modern man. A study of variation
in DNA, however, suggested African origin of modern
man. What kind of observation of DNA variation could
suggest this?
1. Greater variation in Asia than in Africa.
2. Greater variation in Africa than in Asia.
3. Similar variation in Africa and Asia.
4. Variation only in Asia and no variation in Africa
How does a bacterial cell protect its own DNA from restriction enzymes?
| 1. | By adding methyl groups to adenine and cytosines. |
| 2. | By reinforcing bacterial DNA structure with covalent phosphodiester bonds. |
| 3. | By adding histones to protect the double-stranded DNA. |
| 4. | By forming “sticky ends” of bacterial DNA to prevent the enzyme from attaching |
DNA fragments separated by gel electrophoresis are shown. Mark the correct statement :
| 1. | Band 3 contains more positively charged DNA molecules than band 1. |
| 2. | Band 3 indicates more charge density than bands 1 and 2. |
| 3. | Band 1 has longer DNA fragment than bands 2 and 3. |
| 4. | All bands have equal lengths and charges but differ in base composition. |
Why photorespiration does not take place in C4 plants?
1. Do not contain RuBisCo.
2. Have a mechanism that increases the concentration of CO2 at the enzyme site.
3. Cells do not allow oxygen to accumulate in them.
4. Cells are impermeable to oxygen.
RQ of tripalmitin is
1.
2.
3.
4.
In photosynthesis, fixation of 1 CO2 in maize (A) and wheat (B) plant requires
| 1. | (A) 3 ATP and 2 NADPH + H+ (B) 3 ATP and 2 NADPH + H+ |
| 2. | (A) 5 ATP and 2 NADPH + H+ (B) 3 ATP and 2 NADPH + H+ |
| 3. | (A) 2 ATP and 2 NADPH + H+ (B) 3 ATP and 2 NADPH + H+ |
| 4. | (A) 30 ATP and 12 NADPH + H+ (B) 18 ATP and 12 NADPH + H+ |
The phytohormone which promotes rapid internode elongation in deep water rice plant is
| 1. | Gibberellins | 2. | Ethylene |
| 3. | Auxin | 4. | Cytokinin |
A person with Down syndrome will show all the given symptoms except
1. Many loops on finger tips.
2. Flat back of head
3. Big and wrinkled tongue
4. Narrow round face
Decomposition is controlled by several factors. Find out the incorrect option with respect to it.
1. Slower if detritus is rich in lignin and chitin.
2. Quicker if detritus is rich in nitrogen.
3. Cool and moist environment favour decomposition
4. Low temperature and anaerobiosis inhibit decomposition.
Trichoderma species are free-living fungi that are very common in the
1. Root ecosystem
2. Stem ecosystem
3. Leaf ecosystem
4. Both 2 and 3
A cross was made between two Antirrrhinium species with a genotype Rr. Of the total 500 progenies obtained individual with parental phenotype would be
1. 250
2. 125
3. 370
4. 400
Find the odd one out with respect to functional component of ecosystem
1. Productivity
2. Stratification
3. Decomposition
4. Nutrient cycling
Which is not a characteristic of imbibition ?
1. There is generation of heat.
2. It is a irreversible phenomena.
3. It involves both adsorption and capillarity.
4. It is also diffusion of water.
Oxygen haemoglobin dissociation curves are represented as A and B. Select the incorrect interpretation
| 1. | Curve B has increased ability to unload O2 in tissues, with respect to curve A. |
| 2. | The shift in a curve from B to A is associated with decreased P50 value. |
| 3. | At elevated temperature the curve will shift from B to A. |
| 4. | Shift in a curve from A to B is associated with decreased O2 carrying capacity of haemoglobin. |
The plasma calcium levels is very effectively
maintained by a balance between the activities of
1. Thyroxine and parathormone.
2. Parathormone and calcitonin.
3. Aldosterone and vasopressin.
4. Insulin and glucagon.
Which of the following is correct about probe?
| A: | It is ssDNA or ssRNA. |
| B: | Used to detect gene of interest from gene library. |
| C: | Used to detect mutations in genes in suspected cancer patients. |
| D: | Used to detect HIV in suspected AIDS patients. |
1. A only
2. A and B
3. A, B and C
4. A,B,C and D
Which of the following is not present in ATP?
(1) Pentose sugar
(2) Deoxyribose sugar
(3) Purine base
(4) Phospho-ester linkage
(5) Phospho-diester linkage
(6) Hydrogen bond
1. (2),(4), (5),(6)
2. (1), (6), (2), (5)
3. (2), (5), (6)
4. (2), (6) only
Which of the following statements about Bacillus thuringiensis are correct?
| A. | One of the toxins produced by the bacteria is thurioside, which is active against different groups of insect larvae. |
| B. | The toxin accumulates in the bacteria during the sporulation stage. |
| C. | Upon ingestion by susceptible insects they are converted into active form and kill them by inhibition of ion transport in the midgut. |
| D. | The proteins encoded by the gene cry II Ab control corn borer, a lepidopteran. |
1. A and B
2. B and C
3. A, B and C
4. A, B, C and D
In terms of algebraic symbols used in the Hardy Weinberg equation (p and q) the most likely effect of assortative mating (highly selective mating) on the frequencies of allele and genotype for a gene locus would be
| 1. | A decrease in p2 compare to q2 |
| 2. | A trend toward zero for q2 |
| 3. | Convergence of p2 and q2 towards equal values. |
| 4. | A decrease in 2pq below the value expected by Hardy-weinberg theoram. |
Statins have been commercialised as __A__ and is produced by a __B__ .
Select the correct option to fill in the blanks A and B.
1. A – Blood-cholesterol lowering agents
B – Fungus
2. A – Clot busters
B – Fungus
3. A – Clot busters
B – Bacterium
4. A – Immunosuppressive agents
B – Bacterium
Select the wrongly matched pair
1. Predator - Herbivore.
2. Brood parasitism - Cuckoo.
3. Commensalism - Barnacles on the whale.
4. Mutualism - Sea anemone on a hermit crab.
Which of the following statement is/are correct?
| a. | As we move on Earth from low to high latitude, the biodiversity increases. |
| b. | In rivet popper hypothesis given by Paul Ehrlich, rivets on the wings are considered as key species. |
| c. | India possesses 8.1% species diversity of the world. |
1. b and c only
2. b only
3. All a, b and c
4. a and c only
Out of the following, which group of animals has least
number of species in Amazonian rain forest?
1. Mammals
2. Birds
3. Amphibians
4. Reptiles
In 1987, the international treaty to control the emission
of ODS was signed in
1. Copenhagen
2. Canada
3. Paris
4. Morocco
Select the mismatch
|
1. |
Scapula |
- |
Situated in between 2nd to 7th ribs |
|
2. |
Ball and socket joint |
- |
Humerus and pectoral girdle |
|
3. |
Rib cage |
- |
Formed by vertebrae, ribs and sternum |
|
4. |
Pelvic girdle |
- |
Articulates with thigh bone at pubic symphysis. |
Mark the correct set for appendicular skeleton?
1. Coxal bone, Clavicle, Hyoid bone.
2. Patella, Coxal bone, Sacrum.
3. Coxal bone, Scapula, Patella.
4. Ulna, Hyoid bone, Zygonatic bone
Which of the following symptom can not be expected, if there is tumour like growth in parathyroid glands?
| 1. | Tetany of muscle. |
| 2. | Increase in Ca++ level deposition of bones. |
| 3. | More tubular reabsorption of Ca++ from the nephric tubule. |
| 4. | Both 1 and 2. |
Which of the following set of hormones can easily pass through the cell membrane of a target cell and bind to specific intracellular receptors?
a. Placental progesterone
b. Aldosterone
c. Estrogen
d. Thyroxine
Mark the correct set:
1. b&c
2. a,b&c
3. a &c
4. a,b,c & d
In cattles homozygosity results in
1. Improvement of breed.
2. Inbreeding depression.
3. Cross-breed formation.
4. All of these.
The organism found as free living in rhizosphere and is an effective biocontrol agent of several plant pathogens is
1. Cercospora
2. Fusarium
3. Trichoderma
4. Bacillus
Which of the following statement is incorrect about menstrual cycle?
| 1. | LH Surge causes ovulation at the mid of the menstrual cycle |
| 2. | Corpus luteum is formed by graafian follicle on the next day of ovulation under the influence of progesterone |
| 3. | Abrupt fall in progesterone is the instant cause of menstruation |
| 4. | Corpus luteum remains active about 10-12 days in a normal menstruating female |
One of the following is haploid cell
1. Primordial germ cell
2. Oogonium
3. Primary oocyte
4. Secondary oocyte
How many of the following belong to family Solanaceae ?
Chilli, Tulip, Petunia, Colchicine, Belladonna, Lupin, Tobacco, Indigifera,Tomato, Gloriosa,
Dolichos, Citrus.
1. 4
2. 6
3. 5
4. 3
The swollen base of semicircular canals containing projecting ridge is called–
1. Macula
2. Jacobson’s organ
3. Crista ampullaris
4. Organs of Corti
The figure given below illustrates the changes taking place during the human menstruation cycle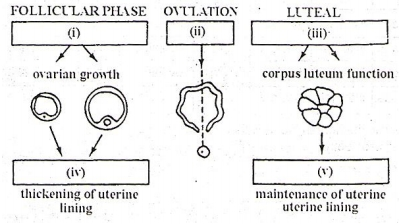
In each of the boxes shown in the figure write the name of the hormone, or hormones controlling the stage in the human menstrual cycle.
| 1. | (i) FSH, (ii) LH, (iii) LH, (iv) Estrogen, (v) Progesterone |
| 2. | (i) LH, (ii) FSH, (iii) LH, (iv) Estrogen, (v) Progesterone |
| 3. | (i) FSH, (ii) LH, (iii) FSH, (iv) Estrogen, (v) Progesterone |
| 4. | (i) FSH, (ii) LH, (iii) LH, (iv) Progesterone, (v) Estrogen |
In Galapagos island, Darwin found different varieties of finches, originated from a common ancestor. The original variety of these finches was:-
1. Insectivorous
2. Vegetarian
3. Seed-eating
4. Cactus eating
Match the following columns of groups and examples?
| Group | Example | ||
| (i) | Sphinopsida | (a) | Adiantum |
| (ii) | Bryopsida | (b) | Polysiphonia |
| (iii) | Pteropsida | (c) | Equisetum |
| (iv) | Liver Wort | (d) | Funaria |
| (v) | Rhodophyceae | (e) | Marchantia |
| 1. | i-a, ii-b, iii-c, iv-d, v-e |
| 2. | i-c, ii-a, iii-e, iv-b, v-d |
| 3. | i-c, ii-d, iii-a, iv-e, v-b |
| 4. | i-d, ii-a, iii-c, iv-e, v-b |
The optically active compound among the following is-
1. Isobutyric acid
2. beta-Chloropropionic acid
3. Propionic acid
4. alpha-Chloropropionic acid
Which among the given molecules can exhibit tautomerisrn?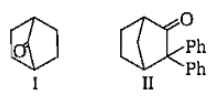
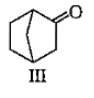
1. III Only
2. Both I and III
3. Both Iand II
4. Both II and III
In the Kjeldahl's method for estimation of nitrogen present in a soil sample, ammonia evolved from 0.75 g of sample neutralised 10 mL of 1 M H2SO4. The percentage of nitrogen in the soil is
1. 37.33 2. 45.33
3. 35.33 4. 43.33
The osmotic pressure of 5 % (mass-volume) solution of cane sugar at 150 °C (mol. mass of sugar = 342 g/mole) is:
| 1. | 4 atm | 2. | 5.07 atm |
| 3. | 3.55 atm | 4. | 2.45 atm |
A solution containing 6.8 g of a non-ionic solute in 100 g of water was found to freeze at −0.93 oC. The freezing point depression constant of water is 1.86. The molecular weight of the solute is-
| 1. | 13.6 m | 2. | 34 m |
| 3. | 68 m | 4. | 136 m |
The rate constant for a reaction of zero-order in A is 0.0030 mol L-1 s-1. How long will it take for the initial concentration of A to fall from 0.10 M to 0.075 M?
1. 8.3 sec
2. 0.83 sec
3. 83 sec
4. 10.3 sec
The process requiring the absorption of energy is :
1.
2.
3.
4.
In a reaction, the rate = k[A]1[B]-2/3 the order of the reaction is-
1. 1/3
2. 2
3. -1/3
4. Zero
The correct order of covalent bond character among
LiCl, BeCl2, BCl3 and CCl4, is:
1. LiCl < BeCl2 > BCl3 > CCl4
2. LiCl > BeCl2 < BCl3 < CCl4
3. LiCl < BeCl2 < BCl3 < CCl4
4. LiCl > BeCl2 > BCl3 > CCl4
The density of a gaseous substance at 1 atm pressure and 773 K is 0.4 g/L. If the molecular weight of the substance is 30, the dominant force existing among gas molecules is/are-
| 1. | Repulsive | 2. | Attractive |
| 3. | Both (1) & (2) |
4. | None of these |
Van der Waals equation at low pressure is -
1. PV = RT - Pb
2. PV = RT -
3. PV = RT +
4. PV = RT + Pb
A 0.1 molar solution of weak base BOH is 1% dissociated. If 0.2 moles of BCl is added in 1 litre solution of BOH, the degree of dissociation of BOH will become
1.
2.
3. 0.05
4. 0.02
The product of acetone with is
1.
2.
3.
4.
The final product Y in the below mentioned sequence is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Consider the following reaction
The structure of B is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The products A and B in the below mentioned reaction are, respectively:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
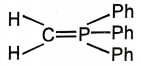
This conversion may be carried by using reagent A.
A maybe
1. 
2.
3.
4.
The numbers of lone pairs and bond pairs in hydrazine are, respectively
1. 2 and 4
2. 2 and 6
3. 2 and 5
4. 1 and 5
Oxidation states of C in Toluene is/are
1. -3
2. -1
3. 0
4. All of these
As an isolated box, equally partitioned, contains two ideal gasses A and B as shown:
When the partition is removed, the gases mix. The changes in enthalpy and entropy in the process, respectively, are
1. Zero, positive
2. Zero, negative
3. Positive, zero
4. Negative, zero
The for vaporization of a liquid is \(20 \mathrm{~kJ} / \mathrm{mol}.\) Assuming ideal behaviour, the change in internal energy for the vaporization of \(1 \mathrm{~mol}\) of the liquid at \(60^{\circ} \mathrm{C}\) and 1 bar is close to:
| 1. | \(13.2 \mathrm{~kJ} / \mathrm{mol} \) | 2. | \(17.2 \mathrm{~kJ} / \mathrm{mol} \) |
| 3. | \(19.5 \mathrm{~kJ} / \mathrm{mol} \) | 4. | \(20.0 \mathrm{~kJ} / \mathrm{mol}\) |
The amount of required to completely reduce 100 mL of 0.25 N iodine solution, is
1. 6.20 g
2. 9.30 g
3. 3.10 g
4. 7.75 g
A metal Oxide can be reduced by hydrogen to give free metal and water. O. 1596 gm of metal Oxide requires 6 mg Of hydrogen for complete reduction. The atomic weight of the metal is:
1. 27.9
2. 159.6
3. 79.8
4. 55.8
| Molar mass | |
| 461 |
The compound that does not reduce Benedict's solution is:
| 1. | Glucose | 2. | Fructose |
| 3. | Sucrose | 4. | Aldehyde |
A photon with an initial frequency of \(10^{11}~\mathrm{Hz}\) scatters off an electron at rest. Its final frequency is \(0.9 \times10^{11}~\mathrm{Hz}.\) The speed of scattered electron is close to:
| 1. | \(3 \times10^{2}~\mathrm{ms}^{-1}\) | 2. | \(3.8 \times10^{3}~\mathrm{ms}^{-1}\) |
| 3. | \(2 \times10^{6}~\mathrm{ms}^{-1}\) | 4. | \(30~\mathrm{ms}^{-1}\) |
The wavelength of a certain line in the Balmer series is observed to be 4341 Ao for hydrogen atoms.
The electronic transition among the following may be:
1. 3 → 2
2. 4 → 1
3. 5 → 2
4. 5 → 3
Zieglar-Natta catalyst is
1.
2.
3.
4.
The initial reaction in the formation of bakelite is an example of:
1. Aromatic electrophilic substitution
2. Aromatic nucleophilic substitution
3. Free radical reaction
4. Aldol reaction
A metal ion has configuration. In octahedral complex entity of low spin, the number of electrons present in set of orbitals will be
| 1. | 6 | 2. | 4 |
| 3. | 0 | 4. | 2 |
A compound is made by mixing cobalt (III) nitrite and potassium nitrite solution in a ratio of 1 : 3. The aqueous solution of the compound showed 4 particles per molecule whereas molar conductivity reveals the presence of six electrical charges. The formula of the compound is:
1. Co(NO2)3 · 2KNO2
2. Co(NO2)3 · 3 KNO2
3. K3[Co(NO2)6]
4. K[Co(NO2)4]
A substance that gives a brick red flame and breaks down
on heating to give oxygen and a brown gas is:
1. Calcium carbonate
2. Magnesium carbonate
3. Calcium nitrate
4. None of the above
Gypsum on heating can change to :
1. Orthorhombic form.
2. Plaster of paris.
3. Dead plaster.
4. All of the above.
Out of the following which is a hydride?
1. Hydrolith
2. Oxidane
3. Azane
4. All of these
Identify the metal M, which has the following extraction principle.
1. Mg
2. Pb
3. Sn
4. Fe
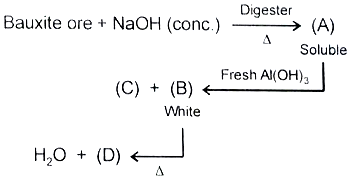
Compound (D) is
1.
2.
3.
4.
NaCN is a depressant that is used:
| 1. | To Separate PbS from ZnS by forming a complex with PbS. |
| 2. | To Separate PbS from ZnS by forming a complex with ZnS. |
| 3. | To Form froth. |
| 4. | As a collector. |
Why is roasting important in the iron metallurgy process?
| 1. | FeO gets oxidized to Fe2O3 which does not combine with acidic SiO2 |
| 2. | SiO2 is an acidic oxide which can combine with metallic oxides i.e. FeO, and Fe2O3 |
| 3. | FeO when roasted get oxidized to more basic Fe2O3 |
| 4. | None of the above. |
The correct statement is
1. Green vitriol and blue vitriol are isomorphus
2. KMnO4 and K2Cr2O7 are coloured due to d-d transitions
3. Cu2Cl2 and Ag2S are coloured
4. Upon strong heating paramagnetic gases are evolved by CuSO4 and AgNO3
While Fe3+ is stable, Mn3+ is not stable in acidic solution because
1. O2 oxidises Mn2+ to Mn3+
2. O2 oxidises both Mn+2 to Mn+3 and Fe2+ to Fe3+
3. Fe3+ oxidises H2O to O2
4. Mn3+ oxidised H2O to O2
Choose the correct statement regarding bonding in FeCl3 at high temperature?
(I) It contains 2c-2e- bond
(II) It contains 3c-2e- bond
(III) It contains a co-ordinate bond
1. (I), (II)
2. (I), (III)
3. (II), (III)
4. (I), (II) & (III)
The lanthanide compound which is used as a most powerful liquid laser after dissolving in selenium oxychloride is
1. Cerium oxide
2. Neodymium oxide
3. Promethium sulfate
4. Cerium sulfate
For which of these electrodes will the reduction potential vary with pH?
l.
ll.
lll.
1. l only
2. ll only
3. l and ll only
4. l, ll and lll only
Equivalent conductance of saturated \(\mathrm {BaSO}_4\) solution is \(400 \mathrm { ~ohm}^{-1}\) \(\mathrm {cm}^2\) \(\mathrm { ~equivalent}^{-1}\) and it's specific conductance is \(8 \times 10^{-5} \text { ohm}^{-1} \text {cm}^{-1}\) ; hence solubility product \(K_{sp}\) of \(\mathrm {BaSO}_4\) is :
1. \(4 \times 10^{-8} \text {M}^2\)
2. \(1 \times 10^{-8} \text {M}^2\)
3. \(2 \times 10^{-4} \text {M}^2\)
4. \(1 \times 10^{-4} \text {M}^2\)
The number of stereoisomers obtained by bromination of trans-2-butene is:
1. 1
2. 2
3. 3
4. 4
Instantaneous displacement current 1 A in the space between the parallel plates of 1 F capacitor can be established by changing the potential difference at the rate of:
1. 0.1 V/s
2. 1 V/s
3.
4.
Some equipotential surfaces are shown in the figure. The electric field at points \(A\), \(B\) and \(C\) are respectively:

| 1. | \(1~\text{V/cm}, \frac{1}{2} ~\text{V/cm}, 2~\text{V/cm} \text { (all along +ve X-axis) }\) |
| 2. | \(1~\text{V/cm}, \frac{1}{2} ~\text{V/cm}, 2 ~\text{V/cm} \text { (all along -ve X-axis) }\) |
| 3. | \(\frac{1}{2} ~\text{V/cm}, 1~\text{V/cm}, 2 ~\text{V/cm} \text { (all along +ve X-axis) }\) |
| 4. | \(\frac{1}{2}~\text{V/cm}, 1~\text{V/cm}, 2 ~\text{V/cm} \text { (all along -ve X-axis) }\) |
If \(\overrightarrow{A} \times \overrightarrow{B} = \overrightarrow{C} + \overrightarrow{D}\), then which of the following statement is correct?
| 1. | \(\overrightarrow B\) must be perpendicular to \(\overrightarrow C\) |
| 2. | \(\overrightarrow A\) must be perpendicular to \(\overrightarrow C\) |
| 3. | Component of \(\overrightarrow C\) along \(\overrightarrow A\) = Component of \(\overrightarrow D\) along \(\overrightarrow A\) |
| 4. | Component of \(\overrightarrow C\) along \(\overrightarrow A\) = - (Component of \(\overrightarrow D\) along \(\overrightarrow A\)) |
The position of a particle is given by , momentum . The angular momentum is perpendicular to :-
1. X-axis
2. Y-axis
3. Z-axis
4. Along the line at equal angles to all the three axes
| 1. | plane parallel to \(yz\) plane |
| 2. | plane parallel to the \(x\text{-}\)axis |
| 3. | concentric circle centered at the origin |
| 4. | coaxial cylinder with axis parallel to the \(y\text-\)axis |
The graph below shows position as a function of time for two trains running on parallel tracks.
Which of the following statements is true?
| 1. | At time \(t_B \) both the trains have the same velocity |
| 2. | Both the trains have the same velocity at some time after \(t_B \) |
| 3. | Both the trains have the same velocity at some time before \(t_B \) |
| 4. | Both the trains have the same acceleration |
To the captain of a ship \(A\) travelling with velocity \(\overrightarrow{v_{A}} = \left( 3 \hat{i} - 4 \hat{j} \right)\) km/h, a second ship \(B\) appears to have a velocity \(\overrightarrow{v_{B}} = \left(5 \hat{i} +12 \hat{j} \right)\) km/h. What is the true velocity of the ship \(B\)?
1. \(2 \hat{i} + 16 \hat{j}\) km/h
2. \(13 \hat{i} + 8 \hat{j}\) km/h
3. \(- 2 \hat{i} - 16 \hat{j}\) km/h
4. none of these
A varying horizontal force F = at acts on a block of mass m kept on a smooth horizontal surface. An identical block is kept on the first block. The coefficient of friction between the blocks is µ. The time after which the relative sliding between the blocks prevails is:-
1.
2.
3.
4.
A chain of length L is placed on a horizontal surface as shown in figure. At any instant x is the length of chain on rough surface and the remaining portion lies on smooth surface. Initially x = 0. A horizontal force P is applied to the chain (as shown in figure). In the duration x changes from x = 0 to x = L, for chain to move with constant speed-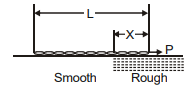
1. the magnitude of P should increase with time
2. the magnitude of P should decrease with time
3. the magnitude of P should increase first and then decrease with time
4. the magnitude of P should decrease first and then increase with time
1. \(u^{2} \sin^{2}\alpha\)
2. \(\dfrac{m u^{2} \cos^{2} \alpha}{2}\)
3. \(\dfrac{m u^{2}\sin^{2} \alpha}{2}\)
4. \(- \dfrac{m u^{2}\sin^{2} \alpha}{2}\)
A square loop of side 'a' hangs from an insulating hanger of spring balance. The magnetic field of strength B occurs only at the lower edge of loop which carries a current I. Find the change in the reading of the spring balance if the direction of current is reversed :-
1. IaB
2. 2IaB
3.
4.
A straight solenoid has \(50\) turns per cm in primary coil and \(200\) turns in the secondary coil. The area of cross-section of the solenoid is \(4\) cm2. Calculate the mutual inductance.
1. \(5.0~\text{H}\)
2. \(5.0\times 10^{-4}~\text{H}\)
3. \(2.5~\text{H}\)
4. \(2.5\times 10^{-4}~\text{H}\)
A man hangs from a rope attached to a hot-air balloon. The man's mass is greater than the mass of the balloon and its contents. The system is stationary in the air. If the man now climbs up to the balloon using the rope, the centre of mass of the "man plus balloon" system will:
| 1. | remain stationary |
| 2. | move up |
| 3. | move down |
| 4. | first moves up and then return to its initial position |
Three rods made of the same material, having the same cross-sectional area but different lengths 10 cm, 20 cm and 30 cm are joined as shown. The temperature of the junction will be:-
1. \(10.8^{\circ}\mathrm{C}\)
2. \(14.6^{\circ}\mathrm{C}\)
3. \(16.4^{\circ}\mathrm{C}\)
4. \(18.2^{\circ}\mathrm{C}\)
If the temperature of the source is increased, then the efficiency of a Carnot engine:-
1. Increases
2. decreases
3. Remains constant
4. First increases and then remains constant
In an astronomical telescope, the focal length of the objective lens is \(100\) cm, and of eyepiece is \(2\) cm. The magnifying power of the telescope for the normal eye is:
1. \(50\)
2. \(10\)
3. \(100\)
4. \(\frac{1}{50}\)
In Young's experiment performed with light of wavelength 6000 Å, when a thin glass plate of refractive index 1.5 is placed in the path of one of the interfering beams, the central bright fringe shifts by 2 mm to a position previously occupied by the seventh bright fringe. Then find thickness of the plate.
1. 10.2
2. 12
3. 6.8
4. 8.4
A container has two immiscible liquids of densites and (> ). A capillary tube of radius r is inserted in the liquid so that its bottom reaches upto the denser liquid. The denser liquid rises in the capillary and attains a height h from the interface of the liquids, which is equal to the column length of the lighter liquid. Assuming angle of contact to be zero, the surface tension of heavier liquid is :-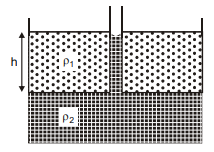
1.
2.
3.
4.
Water flows through a horizontal tube as shown in figure. If the difference in heights of water column in vertical tubes is 80 cm and the area of cross-section at A and B are 5 cm2 and 3 cm2 respectively, then the rate of flow of water at A( in cc/sec) is:-
[ ]
1.
2.
3. 150
4. None of these
A radioactive sample decays by 63% of its initial value in 10 s. It would have decayed by 50% of its initial value in :
1. 7 s
2. 14 s
3. 0.7 s
4. 1.4 s
The kinetic energy of an electron is 5 eV. Calculate the de-Broglie wavelength associated with it (h =6.6 × 10–34 Js, me = 9.1 × 10–31 kg)
1. 5.47
2. 10.9
3. 2.7
4. 12.01
Suppose an electron is attracted towards the origin by a force where 'k' is a constant and 'r' is the distance of the electron from the origin. By applying Bohr model to this system, the radius of the nth orbital of the electron is found to be 'rn' and the kinetic energy of the electron to be 'Tn'. Then which of the following is true:-
1. independent of n,
2.
3.
4.
Logic gates \(X\) and \(Y\) have the truth tables shown below:

| \(X\) | ||
| \(P\) | \(Q\) | \(R\) |
| \(0\) | \(0\) | \(0\) |
| \(1\) | \(0\) | \(0\) |
| \(0\) | \(1\) | \(0\) |
| \(1\) | \(1\) | \(1\) |

| \(Y\) | |
| \(P\) | \(R\) |
| \(0\) | \(1\) |
| \(1\) | \(0\) |
When the output of \(X\) is connected to the input of \(Y\), the resulting combination is equivalent to a single:
1. NOT gate
2. OR gate
3. NAND gate
4. AND gate
When forward bias is applied to a P–N junction, then what happens to the potential barrier VB and the width of depletion region x ?
1. increases, x decreases
2. decreases, x increases
3. increases, x increases
4. decreases, x decreases
Electric field due to an infinite line of charge as shown in figure (a) at a point P at a distance r from the line is E. If the wire is folded at point A so that both parts lie alongside as shown in figure (b), then express electric field at P in vector form.
1.
2.
3.
4.
A block of weight \(W\) is supported by two strings inclined at \(60^{\circ}\) and \(30^{\circ}\) to the vertical. The tensions in the strings are \(T_1\) and \(T_2\) as shown. If these tensions are to be determined in terms of \(W\) using the triangle law of forces, which of these triangles should you draw? (block is in equilibrium):

| 1. |  |
2. |  |
| 3. |  |
4. |  |
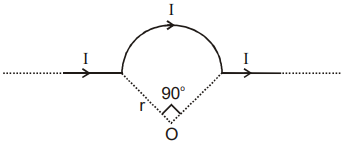
The magnetic field at center O of the arc shown in the figure is:-
1.
2.
3.
4.
When the current in the portion of the circuit shown in the figure is \(2\) A and increases at the rate of \(1\) A/s, the measured potential difference \(V_{ab}=8\) V. However, when the current is \(2\) A and decreases at the rate of \(1\) A/s, the measured potential difference \(V_{ab}= 4\) V. The value of \(R\) and \(L\) is:

| 1. | \(3~\Omega\) and \(2~\text{H}\) respectively |
| 2. | \(3~\Omega\) and \(3~\text{H}\) respectively |
| 3. | \(2~\Omega\) and \(1~\text{H}\) respectively |
| 4. | \(3~\Omega\) and \(1~\text{H}\) respectively |
In a heating arrangement, an alternating current having a peak value of 28 A is used. To produce the same heat energy if the direct current is used, its magnitude must be:-
1. about 14 A
2. about 28 A
3. about 20 A
4. cannot say
When without a change in temperature, a gas is forced in a smaller volume, its pressure increases because its molecules:-
1. strike the unit area of the container wall more often.
2. strike the unit area of the container wall at a higher speed.
3. strike the unit area of the container wall with greater momentum.
4. have more energy.
When a system is taken from state \(i\) to state \(f\) along path \(iaf, \) \(Q=50\) J and \(W=20\) J. If \(W=-13\) J for the curved return path \(fi,\) \(Q\) in this path is:
1. \(33\) J
2. \(23\) J
3. \(-7\) J
4. \(-43\) J
The near point of a person is 50 cm and the far point is 1.5 m. The spectacles required for reading purpose and for seeing distant objects are respectively:-
1.
2.
3.
4.
The Poisson's ratio of a material is \(0.4.\) If a force is applied to a wire of this material, there is a decrease in the cross-section area by \(2\)%. In such a case the percentage increase in its length will be:
1. \(3\%\)
2. \(2.5\%\)
3. \(1\%\)
4. \(0.5\%\)
The potential energy of a satellite having mass \(m\) and rotating at a height of \(6.4\times 10^{6}~\text{m}\) from the Earth's surface is:
1. \(-0.5mg R_e\)
2. \(-mg R_e\)
3. \(-2mg R_e\)
4. \(4mg R_e\)
A free atom of iron emits X-rays of energy 6.4 keV. The recoil kinetic energy of the atom will be- (if given mass of iron atom = 9.3 × kg)
1. 3.9 × eV
2. 4.9 × eV
3. 3.9 × eV
4. 4.9 × eV
An observer starts moving with uniform acceleration towards a stationary sound source of frequency . As the observer approaches the source, the apparent frequency f heard by the observer varies with time t as
| 1. |  |
2. |  |
| 3. |  |
4. |  |
What will be the energy loss per hour in the iron core of a transformer if the area of its hysteresis loop is equivalent to 2500 erg/? The frequency of the alternating current is 50 Hz. The mass of the core is 10 kg and the density of iron is 7.5 gm/ -
1. joule
2. joule
3. joule
4. joule
A student measures the mass of an object five times. The data are 2.46 kg, 2.47 kg, 2.42 kg, 2.41 kg and 2.44 kg. The reported mean mass with error should be:
1. 2.44 0.01 kg
2. 2.44 0.02 kg
3. 2.44 0.03 kg
4. 2.44 0.04 kg
A battery consists of a variable number \(n\) of identical cells having equal internal resistance connected in series. The terminals of the battery are short-circuited and the current \(I\) measured. Which one of the graphs below shows the correct relationship between \(I\) and \(n?\)
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The displacement-time graph of a particle executing SHM is shown in the figure. Its displacement equation is: (Time period = \(2~\text{s}\))

1. \(x=10\sin \left(\pi t +\frac{\pi}{6}\right)\)
2. \(x=10\sin (\pi t )\)
3. \(x=10\cos (\pi t )\)
4. \(x=5\sin \left(\pi t +\frac{\pi}{6}\right)\)
| 1. | due to the decrease in pressure. |
| 2. | due to a decrease in temperature. |
| 3. | as a result of a decrease in both temperature and pressure. |
| 4. | statement is wrong. |
A potential divider is used to give outputs of \(2~\text{V}\) and \(3~\text{V}\) from a \(5~\text{V}\) source, as shown in the figure.
| 1. | \({R}_1=1~\text{k} \Omega, {R}_2=1 ~\text{k} \Omega, {R}_3=2 ~\text{k} \Omega\) |
| 2. | \({R}_1=2 ~\text{k} \Omega, {R}_2=1~\text{k} \Omega, {R}_3=2~\text{k} \Omega\) |
| 3. | \({R}_1=1 ~\text{k} \Omega, {R}_2=2~ \text{k} \Omega, {R}_3=2~ \text{k} \Omega\) |
| 4. | \({R}_1=3~\text{k} \Omega, {R}_2=2~\text{k} \Omega, {R}_3=2~ \text{k} \Omega\) |
A rod is falling down with constant velocity \(V_0\) as shown. It makes contact with hinge A and rotates around it. The angular velocity of the rod just after the moment when it comes in contact with hinge A is:
| 1. | \(2 \mathrm{V}_0 / 3 \mathrm{L} \) | 2. | \(3 \mathrm{V}_0 / 2 \mathrm{L} \) |
| 3. | \(\mathrm{V}_0 / \mathrm{L} \) | 4. | \(2 \mathrm{V}_0 / 5 \mathrm{L}\) |
The coefficient of friction between the block of masses \(m\) and \(2m\) is \(\mu = 2\tan\theta\). There is no friction between the block of mass \(2m\) and inclined plane. The maximum amplitude of the two-block system for which there is no relative motion between both blocks is:

1. \(g\sin\theta \sqrt{\frac{K}{m}}\)
2. \(\dfrac{mg\sin\theta}{K}\)
3. \(\dfrac{3mg\sin\theta}{K}\)
4. \(\dfrac{\mu mg}{K}\)
Find the base resistance in the circuit below, if \(\beta_{DC} =90\) and \(V_{BE}=0.7~V.\)
1. 83
2. 41 k
3. 83 k
4. 41