Match each item in COLUMN I with one in COLUMN II and select your answer from the codes given:
|
|
COLUMN I |
|
COLUMN II |
|
|
SCIENTIST |
|
CONTRIBUTION |
|
A. |
Francis Crick |
a. |
Breaking the genetic code |
|
B. |
Nirenberg |
b. |
Established Caenorhabditis elegans as a model genetics study organism |
|
C. |
Benzer |
c. |
Central dogma of molecular biology |
|
D. |
Brenner |
d. |
Bacteriophage genetics |
Codes:
A. B. C. D.
1. c a d b
2. c a b d
3. a b c d
4. a c d b
Identify the incorrect statement regarding experiments on Pisum sativum by Gregor Mendel?
| 1. | He conducted hybridization experiments on garden peas for seven years (1856-1863) and proposed the laws of inheritance in living organisms. |
| 2. | It was for the first time that statistical analysis and mathematical logic were applied to problems in biology. |
| 3. | Unfortunately his experiments had a small sampling size, which gave less credibility to the data that he collected. |
| 4. | He investigated characters in the garden pea plant that were manifested as two opposing traits. |
Identify the incorrectly matched pair:
| Organism | Length of DNA |
| 1. Bacteriophage ϕ 174 | 5386 base pairs |
| 2. Bacteriophage Lambda | 48502 base pairs |
| 3. Escherichia coli | 4.6 X 10^6 base pairs |
| 4. Human beings | 3.3 X 10^9 base pairs |
To form a nucleoside, a nitrogenous base is linked to a pentose sugar:
(1) Through a P-Glycosidic linkage at carbon atom number 1
(2) Through a P-Glycosidic linkage at carbon atom number 5
(3) Through a N-Glycosidic linkage at carbon atom number 1
(4) Through a N-Glycosidic linkage at carbon atom number 5
Which of the following would be nucleoside found only in RNA?
1. Thymidine
2. Cytidine
3. Uridine
4. Adenosine
Which one of the following populations would most quickly lead to two groups with few shared traits?
1. a population with disruptive selection
2. a population with directional selection
3. a population with stabilizing selection
4. a population with no selection
Which one of the following would cause the Hardy-Weinberg principle to be inaccurate?
| 1. | The size of the population is very large. |
| 2. | Individuals mate with one another at random. |
| 3. | Natural selection is present. |
| 4. | There is no source of new copies of alleles from outside the population. |
Why is genetic polymorphism important to evolution?
| 1. | individual variability provides the raw material for natural selection to act on |
| 2. | genes cannot mutate unless they are polymorphic |
| 3. | only heterozygous individuals are selected in natural populations |
| 4. | the Hardy-Weinberg equilibrium is less likely to be disturbed in polymorphic populations |
The genetic material must show variation. Which of the following is NOT an example of genetic variation?
1. New mutations can occur during meiosis.
2. Different species have different numbers of chromosomes.
3. Different individuals within a species have different phenotypes.
4. Individuals of different species may have similar phenotypes.
Which of these is an enzymatic function of the ribosome?
1. Polynucleotide kinase.
2. Base pair recognition.
3. Methyl transferase.
4. Peptidyl transferase.
The famous double-helix model of DNA was proposed by Watson and Crick in ____ and they shared Nobel Prize for Physiology or Medicine in ________ for their effort.
1. 1951, 1971
2. 1943, 1963
3. 1953, 1962
4. 1969, 1972
Which of the following was not a character studied by Mendel in garden pea?
1. Flower position
2. Pod shape
3. Flower colour
4. Pod position
An individual affected with Turner’s Syndrome:
I. Is a female
II. Has 45 chromosomes
III. Has rudimentary ovaries
Of the above statements, the correct statements are:
1. Only I and II
2. Only I and III
3. Only II and III
4. I, II and III
Which of the following is a pyrimidine base found in DNA?
1. Adenine
2. Guanine
3. Uracil
4. Cytosine
In a strand of a nucleic acid, two nucleotides are linked together by:
1. 3’ – 5’ phosphodiester bond
2. 5’ – 3’ phosphodiester bond
3. 2’ – 5’ phosphodiester bond
4. 3’ – 1’ phosphodiester bond
The backbone of a polynucleotide chain is made of:
1. Sugar and nitrogenous bases
2. Phosphate and nitrogenous bases
3. Sugar and phosphate
4. Sugar, phosphate, and nitrogenous bases
Histones are:
1. Positively charged and basic amino acids
2. Negatively charged and basic proteins
3. Positively charged and acidic proteins
4. Not found in bacteria
Transcriptionally active chromatin is termed as:
1. Heterochromatin
2. Euchromatin
3. Prechromatin
4. Prochromatin
In Griffith experiment, which of the following bacterial strains were capable of causing pneumonia in mice?
1. Live R
2. Bacteria strain without polysaccharide coat
3. Heat Killed S
4. Live R without capsule and Heat Killed S
The unequivocal proof that DNA is the genetic material was provided by:
1. Avery, Macleod and McCarty
2. Hershey and Chase
3. Meselson and Stahl
4. Watson and Crick
Among the following sets of examples for
divergent evolution, select the incorrect
option.
1. Brain of bat, man and cheetah
2. Heart of bat, man and cheetah
3. Forelimbs of man, bat and cheetah
4. Eyes of Octopus, bat and man
The process by which organisms with different evolutionary history evolve similar phenotypic adaptations in response to a common environmental challenge, is called
1. natural selection
2. convergent evolution
3. non- random evolution
4. adaptive radiation
Darwin’s finches are a good example of
1. industrial melanism
2. connecting link
3. adaptive radiation
4. convergent evolution
In a population of 1000 individuals, 360
belongs to genotype AA, 480 to Aa and the
remaining 160 to aa. Based on this data, the
frequency of allele A in the population is
1. 0.4 2. 0.5
3. 0.6 4. 0.7
The chronological order of human evolution
from early to the recent is
1. Australopithecus Ramapithecus Homo
habilis Homo erectus
2. Ramapithecus Australopithecus Homo
habilis Homo erectus
3. Ramapithecus Homo habilis
Australopithecus Homo erectus
4. Australopithecus Homo habilis
Ramapithecus Homo erectus
Which of the following is a correct
statement ?
1. IUDs one inserted need not to be replaced
2. IUDs are generally inserted by the user herself
3. IUDs increase phagocytosis of sperms in the
uterus
4. IUDs suppress gametogenesis
In context of amniocentesis, which of the
following statements is incorrect?
1. It is used for prenatal sex- determination
2. It can be used for detection of Down syndrome
3. It can be used for detection of cleft palate
4. It is usually done when a woman is between
14-16 weeks pregnant
Which one of the following is the most widely
accepted method of contraception in India, at
present?
1. Tubectomy
2. Diaphragm
3. IUDs (Intrauterine Devices)
4. Cervical caps
Which one is not applicable with respect to control experiment of Miller ?
1. Ratio of is 1:1:2
2. Presence of water vapour
3. Created electric discharge in closed flask 800C
4. Both 1 and 3
Which of the following is not an example of analogous structure ?
1. Eye of Octopus and mammal
2. Flippers of penguins and dolphin
3. Sweet potato and potato
4. Mouth parts of cockroach and mosquito
Mark the correct statement with respect to industrial melanism:
| 1. | Lichens can be used as an industrial pollution indicator. |
| 2. | Low count of melanic moths are found in rural areas where industrialisation did not occur. |
| 3. | Neither grey nor the dark variety of moths has been completely wiped out. |
| 4. | All of these |
Which of the following is not a natural method for birth control ?
| 1. | Periodic abstinence |
| 2. | Coitus interruptus |
| 3. | Lactational amenorrhoea |
| 4. | Vaults |
In a human female, the menstrual cycle is of 24 days. What will be the length of follicular phase and luteal phase if the menstrual phase is for four days?
Follicular Phase Luteal Phase
1. 14 days 6 days
2. 6 days 14 days
3. 10 days 10 days
4. 10 days 14 days
Identify the STI (Sexually Transmitted Infection) caused due to a protozoan.
1. Trichomoniasis
2. Gonorrhoea
3. Chancroid
4. Syphilis
During oogenesis, meiosis I in a female is completed in ___________ within __________ respectively.
Select the option which fill the blanks correctly.
| 1. | Secondary oocyte, tertiary follicle |
| 2. | Primary oocyte, tertiary follicle |
| 3. | Primary oocyte, graafian follicle |
| 4. | Secondary oocyte, graafian follicle |
Opinion of how many registered medical practitioners is required for MTP, if the pregnancy has lasted more than 12 weeks, but fewer than 24 weeks?
1. One
2. Two
3. Three
4. Four
The shedding of endometrium during menstruation occurs due to decline in concentration of
1. Estrogen
2. Progesterone
3. Oxytocin
4. FSH
Graphical representation of ovarian and pituitary hormones are given below. Identify the hormones A, B, C and D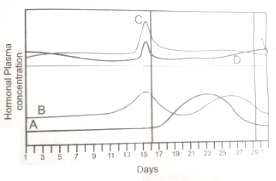
1. A - estrogen, B - progesterone, C - LH, D - Fish
2. A - FSH, B - LH, C - estrogen, D - Progesterone
3. A - progesterone, B - estrogen, C - LH, D - FSH
4. A - Progesterone, B - estrogen, C - FSH, D - LH
The first movement of foetus and appearance of hair on the head are usually observed during
1. month
2. month
3. month respectively
4. month respectively
Which of the following is a correct match?
| 1. | Treponema Pallidum - Causative agent of STI Syphilis |
| 2. | Castration - Emergency contraceptive methods |
| 3. | GIFT - in-vitro fertilization in surrogate mother |
| 4. | Saheli - Oral contraceptive, steroid pill developed at CDRI |
Arrange the following process starting from the germ
cells of seminiferous tubule?
(i) Meiotic division in secondary spermatocytes.
(ii) Spermiation.
(iii) Spermiogenesis.
(iv) Capacitation.
1. (i), (ii), (iii), (iv)
2. (i), (iii), (ii), (iv)
3. (ii), (i), (iv), (iii)
4. (i), (ii), (iv), (iii)
Mark the incorrect match that is not related?
1. Sertoli cell - Spermiation
2. Spermatid - Spermiogenesis
3. Secondary spermatocyte - Mitotic division
4. Spermatozoa - Capacitation
Mark the incorrect match
| (i) | All spermatocytes have haploid structure. |
| (ii) | All of the spermatogonia changes into primary spermatocyte. |
| (iii) | FSH is required for the process of spermiogenesis. |
| (iv) | Seminal plasma is essential for maturation and motility of sperms. |
1. (i), (ii), (iii) and (iv)
2. (iii) and (iv) only
3. (i) and (ii) only
4. Only (iii)
Female accessory ducts related to reproductive system is
(i) Oviducts
(ii) Uterus
(iii) Vagina
1. (i), (ii), (iii)
2. (i) only
3. (i) and (ii) only
4. (iii) only
Which of the following duct is present outside the
testis?
(i) Rete testis
(ii) Vasa efferentia
(iii) Vas deferens
(iv) Epididymis
1. (i), (ii), (iii), (iv)
2. (iii) and (iv) only
3. (ii), (iii), (iv)
4. (iii) only
Mark the incorrect match
| 1. | Fimbriae - Collection of ovum after ovulation. |
| 2. | Endometrium - Undergoes cyclic changes during menstrual cycle. |
| 3. | Labia majora - Fleshy folds of tissues surrounds the vaginal opening. |
| 4. | Ovarian stroma - It is the inner medulla of ovary which contains all the stages of growing ovarian follicle. |
Gamete formation always occurs in
(i) Sexual reproduction.
(ii) Asexual reproduction.
(iii) Parthenogenesis.
1. (i) only
2. (i), (ii), (iii)
3. (i) and (iii) only
4. (i) and (ii) only
Which of the following is not an application of IUDs?
| 1. | Create a sterile inflammatory response by producing a tissue injury of a minor degree. |
| 2. | Changing the intra-uterine environment and making it spermicidal. |
| 3. | Make the cervix hostile to sperms. |
| 4. | Hormonal changes prevent ovulation. |
All are the characteristics of an individual from trisomy
of chromosome 21, except
1. Short stature
2. Gynaecomastia
3. Palm crease
4. Furrowed tongue
The F1 generation resembles both the parents
in case of
1. Complete Dominance.
2. Incomplete Dominance.
3. Codominance.
4. Both (2) and (3)
What is the ploidy of female gametophyte and PEN
respectively in typical angiosperms?
1. n, 3n
2. 2n, 3n
3. 2n, 2n
4. 3n, 3n
Identify the mismatched pair.
1. Anemophily — Maize.
2. Entomophily Vallisneria.
3. Hydrophily — Zostera.
4. Ornithophily—Bombax.
Identify the incorrect statement.
1. The two alleles of a gene pair are located on
homologous sites of homologous chromosomes.
2. Dominance is not an autonomous feature of a gene.
3. Recessive trait is seen due to functional enzyme
or enzyme produced in abundance .
4. Alleles are forms of a gene which code for a pair of
contrasting traits.
The large holes in 'Swiss cheese' are due to production
of CO2 by
1. Saccharomyces cerevisiae.
2. Lactobacillus acidophilus.
3. Propionibacterium sharmanii.
4. Penicillium roqueforti.
In mature pollen grain, vegetative cell differs from
generative cell as the former
1. Floats in the cytoplasm of generative cell.
2. Is smaller in size.
3. Is spindle shaped with dense cytoplasm.
4. Have abundant food reserve and large nucleus
Statement A - In seed plants, both male and female
gametes are non-motile.
Statement B - In spermatophytes, pollen grains are
the carrier of male gametes.
1. Only statement A is correct.
2. Only statement B is correct.
3. Both statements A and B are incorrect.
4. Both statements A and B are correct
Which among the following traits of Pisum sativum is
only visible when both the alleles are identical?
1. Violet flower.
2. Inflated pod.
3. Axial flower.
4. Green seeds.
A plant breeder employ technique of emasculation
1. To cover the flower with a bag.
2. To prevent self-pollination.
3. To prevent stamen from being contaminated.
4. To produce female plant.
Look at the diagram given below and answer
appropriately
(i) Since ___A___ is associated with fruit, it is called
as ___(i)___ .
(ii) Since ___B___ is present, fruit cannot be called
as ___(ii)___ .
1. A - Pericarp, (i) - True fruits
2. B - Seed, (ii) - Ex-albuminous
3. A - Thalamus, (i) - True fruit
4. B - Seed, (ii) - Parthenocarpic
A typical anther shows
i. Two lobes with two theca each
ii. Each lobe as dithecous
iii. All cells in a given microsporangium as potential PMC
iv. Thousands of microspores per microsporangium
Out of these statements :
1. ii is incorrect.
2. i and ii are correct.
3. iii and iv are incorrect.
4. i, ii, iii and iv are correct.
Find correct match
|
|
Column-I |
|
Column-II |
|
a. |
Thick and swollen cotyledons |
(i) |
Castor. |
|
b. |
Unused endosperm in mature seed |
(ii) |
Legumes |
|
c. |
Unused nucellus in seed |
(iii) |
Cashew nut. |
|
d. |
Thalamus contributes in fruit formation |
(iv) |
Beet. |
1. a(iii), b(iv), c(ii), d(i)
2. a(i), b(iii), c(iv), d(ii)
3. a(ii), b(i), c(iii), d(iv)
4. a(ii), b(i), c(iv), d(iii)
A: All organisms have to reach a certain stage of growth
and maturity in their life, before they can reproduce
sexually.
B: Male and female gametes must be physically
brought together to facilitate syngamy :
1. A - correct B – incorrect.
2. Both A and B are correct.
3. Both A and B are incorrect.
4. A - incorrect B - correct.
In mycorrhiza association the fungal symbiont
helpful in :
1. Phosphorus nutrition.
2. Resistance to root borne pathogen.
3. Tolerance to salinity and drought.
4. All the above.
In monoecious plants like castor and maize
1. Autogamy and allogamy are not prevented
2. Geitonogamy is prevented
3. Autogamy is not prevented
4. Geitonogamy is not prevented
Read the following statements-
I. Each cell of sporogenous tissue in anther is capable of giving rise to microspore tetrad.
II. The pollen grain represent male gametophyte
III. Pollen grains are usually triangular and 10-15 um in diameter.
IV. Sporopollenin is one of the most resistant organic materials which can be destroyed only by strong acids and alkali.
1. I, II are incorrect but III, IV are correct
2. III, IV are incorrect but I, II are correct
3. I, III are incorrect but II, IV are correct
4. II, IV are correct but I, III are incorrect
Which one of the following statements is correct?
1. Sporogenous tissue is haploid
2. Endothecium produces the microspores.
3. Tapetum nourishes the developing pollen
4. Hard outer layer of the pollen is called intine.
The wider part of the fallopian tube is
1. Infundibulum
2. Isthmus
3. Ampulla
4. Cervix
The hilum is a scar on the
1. Seed, where micropyle was present
2. Seed, where funicle was attached
3. Fruit, where it was attached to pedicel
4. Fruit, where style was present
Mark the incorrect statement
| 1. | Outer three layers of anther wall are protective in function |
| 2. | Sporogenous tissue occupies the centre of each microsporangium |
| 3. | Cells of tapetum and endothecium show increase in DNA contents by endomitosis and polyteny |
| 4. | Ploidy level of microspore tetrad is haploid |
Genes which are tightly linked on chromosome show :
1. Very low recombination
2. High recombination
3. Very low parental combination
4. Clear cut independent assortment
In large number of insects the mechanism of sex determination is of XO type, then which of the
following statements is true for XO type determination :
1. All eggs bear an additional X-chromosome besides the autosome
2. All sperms bear only autosome
3. All eggs bear only autosomes
4. All sperms bear only X-chromosomes
In Morgan's experiment, the F2 ratio deviated very significantly from 9:3:3:1, This is due to :
1. Independent assortment
2. Segregation
3. Linkage
4. Dominance
Three region's in DNA - a promoter, structural gene and terminator are collectively known as :
1. Replication unit
2. Transcription unit
3. Translation unit
4. Operon
Which of the following is not correct with respect to salient features of the double helix structure of DNA?
1. The plane of one base pair stacks over the other
2. The backbone is constituted by sugar-phosphate and the bases project outside
3. The two chains have anti-parallel polarity
4. The pitch of the helix is 3.4 nm
In pea plant, the intermediate size of starch grains is due to
1. Dominant epistasis
2. Codominance
3. Incomplete dominance
4. Recessive epitasis
Read the following four statements (A-D)
A. The characters never blend in heterozygous condition
B. Change in a single base pair of DNA does not cause mutation
C. Cancer cells commonly show chromosomal aberrations
D. In chicken, sex chromosomes in male are ZW and in females are ZZ
How many of the above statement is/are right?
1. Two
2. Three
3. Four
4. One
In capping of RNA
1. Unusual nucleotide (methyl guanosine triphosphate) is added to the 5'-end of hnRNA
2. Adenylate residues (200-300) are added at 3'-end
3. Adenylate residues is added to the 5'-end of hnRNA
4. Unusual nucleotide (methyl guanosine triphosphate) is added at 3'-end
Consider the statements regarding Mendel’s work. Find out the incorrect option.
1. Mendel selected 14 true-breeding pea plant varieties, as pairs which were similar except for one character with contrasting traits.
2. To determine the genotype of a tall plant at F2, Mendel crossed the tall plant from F2 with a dwarf plant. This he called a test cross.
3. Mendel proposed that something was being stably passed down, unchanged, from parent to offspring through the gametes, over successive generations. He called these things as ‘factors’.
4. Based on his observations on monohybrid crosses Mendel proposed three general rules to consolidate his understanding of inheritance in monohybrid crosses.
Drosophila was used by Morgan for his experiments as
1. It completes its life cycle in two months.
2. Large size male can be easily grown on sucrose medium.
3. Hereditary variations can be seen with low power microscope.
4. It is a saprophyte.
Consider the following statements:
i. The most interesting molecule with autocalytic and heterocatalytic functions in the living system.
ii. Most abundant genetic material.
iii. Functions as adaptor or structural molecule.
iv. A long polymer of deoxyribonucleotides.
Which of these are correct for DNA?
1. (i), (ii) and (iv)
2. (i), (iii) and (iv)
3. (ii), (iii) and (iv)
4. (i), (ii), (iii) and (iv)
Which one is incorrect (with respect to mutation)?
1. It results in alteration of DNA sequence
2. Usually mutation change gene in next generation
3. Point mutation arise due to change in single base pair
4. Mutation is also a phenomenon that lead to variation in DNA
Seed-set is assured even in absence of pollinators, when:
1. Both flowers ( and
) of a plant mature at the same time
2. The numbers of flowers clustered into an inflorescence to make them conspicuous, colorless and rich in nectar
3. The flowers are cleistogamous
4. The flowers are functionally cross-pollinating but genetically self-pollinating
These are two statements related to sexual reproduction :
(I) In both plants and animals, hormones are responsible for the transitions between the three phases.
(II) Interaction between hormones and certain environmental factors regulate the reproductive processes and the associated behavioural expressions of organisms.
Options :
1. Both (I) and (II) is correct
2. Only (I) is correct
3. Only (II) is correct
4. Neither (I) nor (II) is correct
Different varieties of cheese are known by their characteristic texture, flavour and taste that is due to?
1. Flavouring agent used
2. Living agent used
3. It is inherent quality
4. Arise due to molecular interaction between chemicals
During biological treatment of sewage water the term activated sludge refers to?
1. Activated charcoal which is used for purification
2. Masses of aerobic bacteria and fungal filament which is sedimented
3. Different biochemical’s that comes with polluted water
4. It is digested solid waste which can be used as active fertilizer
Find out which one of the following statement is not correct with respect to gobar-gas plant?
1. It has a floating cover which keeps on rising as gas is produced
2. It is developed by IARI and KVIC
3. Main gas produced is butane, isobutene & propane
4. Spent slurry may be used as fertilizer
In the STPs, biological process of microbial degradation of organic matter involves
1 Utilisation of activated sludge as inoculant produced in the physical process
2 Mainly involves anaerobic breakdown of organic matter
3 Masses of unicellular bacteria entangled in filamentous bacterial forms represent flocs
4 BOD is greatly reduced by the microbial activity
Read the following four statements (a-d), find out the incorrect option with respect to lactic acid bacteria
| a. | In our stomach, they play beneficial role by checking disease causing microbes. |
| b. | LAB produce acids that coagulate and completely digest the milk proteins. |
| c. | Improves the nutritional quality by increasing the amount of riboflavin. |
| d. | Require suitable temperature for their multiplication. |
1. a & b
2. a & c
3. b & c
4. b & d
Alexander Fleming accidently discovered antibiotic penicillin from a mould, but he was working on
1. Streptococci
2. Streptobacilli
3. Staphylobacilli
4. Staphylococci
Find out which one of the following statement is not correct with respect to biogas plant?
1. It has a floating cover which keeps on rising as gas is produced
2. It consists of a 10-15 feet deep tank in which bio-wastes are collected and a slurry of dung is fed
3. Main gases produced are methane, isobutane and propane
4. Spent slurry may be used as fertiliser
The mass of a unit cell of CsCl corresponds to:-
1. 8Cs+ and Cl-
2. 1Cs+ and 6Cl-
3. 1Cs+ and 1Cl-
4. 4Cs+ and Cl-
A hypothetical reaction, A2 +B2 2AB mechanism as given below;
A2 A+ A ............(Fast)
A+B2 → AB + B ............(Slow)
A+ B → AB ............(Fast)
The order of the overall reaction is:
1. 2
2. 1
3. 1.5
4. zero
The rate of a chemical reaction doubles for every 10°C rise of temperature. If the temperature is raised by 50°C, the rate of the reaction increases by about :
1. 10 times
2. 24 times
3. 32 times
4. 64 times
From the stability constant (hypothetical values)given below, predict which is the strongest ligand?
1. Cu2++4NH3[Cu(NH3)4]2+;(k=4.5x1011)
2. Cu2++4CN[Cu(CN)4]2-;(K=2.0x1027)
3. Cu2++2en[Cu(en)2]2+;(K=3.0x1015)
4. Cu2++4H2O[Cu(H2O)4]2+;(K=9.5x108)
The correct IUPAC name for Mn3(CO)12 is:
1. Dodecacarbonylmanganate(0)
2. Dodecacarbonylmanganate(II)
3. Dodecacarbonyltrimanganese(0)
4. Manganicdodecacarbonyl0)
[Cr(H2O)6]Cl3 (At. no. of Cr = 24) has a magnetic moment of 3.83 BM.
What is the correct distribution of 3d electrons in the chromium of the complex:
1. 3d1xy, 3d1yz, 3d1z2
2. 3d(x2-y2), 3d1z2, 3d1xz
3. 3dxy, 3d(x2-y2), 3d1yz
4. 3d1xy, 3d1yz, 3d1zx
If the activation energy is 65 kJ then the reaction at 25°C is .......times fast as compared to 0°C :-
1. 2 times
2. 5 times
3. 11 times
4. 16 times
EAN of Fe in K3[Fe(CN)6] is
1. 34
2. 35
3. 36
4. 47
A solution containing 6.8 g of a non-ionic solute in 100 g of water was found to freeze at −0.93 oC. The freezing point depression constant of water is 1.86. The molecular weight of the solute is-
| 1. | 13.6 m | 2. | 34 m |
| 3. | 68 m | 4. | 136 m |
Which of the following products are formed when potassium bromide reacts with potassium permanganate in alkaline pH?
1.
2.
3.
4.
Zinc dissolve in an excess of NaOH because of the formation of:
| 1. | ZnO | 2. | Zn(OH)2 |
| 3. | NaZn(OH)3 | 4. | Na2ZnO2 |
The degree of dissociation (α ) of a weak
electrolyte, is related to van’t Hoff
factor (i) by the expression:
1.
2.
3.
4.
Ionic radii of Mg2+ and O2- ions are 66 pm and 140 pm respectively. The type of interstitial void and coordination number of Mg2+ ion respectively are
1. Tetrahedral, 12
2. Octahedral, 6
3. Tetrahedral, 6
4. Octahedral, 8
The half cell reduction potential of a hydrogen electrode at pH = 10 will be :
1. 0.59 V
2. – 0.59 V
3. 0.059 V
4. – 0.059 V
The activation energies of two reactions are and with > .
f the temperature of the reacting system is increased from T1 to T2 .
The correct relation is:
1.
2.
3.
4.
The correct name of 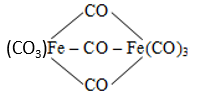 is
is
1. Tri--carbonyl bis (tricarbonyl iron (0)
2. Hexacarbonyl iron (III) -tricarbonyl ferrate (0)
3. Tricarbonyl iron (0) -tricarbonyl iron (0) tricarbonyl
4. Nonacarbonyl iron
Increasing order of 0 of the following complex is –
1. Cr(NH3)63+ < Cr(H2O)63+ < Cr(NO2)63–
2. Cr(H2O)63+ > Cr(NH3)63+ > Cr(NO2)63–
3. Cr(H2O)63+ < Cr(NH3)63+ < Cr(NO2)63–
4. Cr(NH3)63+ < Cr(NO2)63– < Cr(H2O)63+
Calculate the molal depression constant of a solvent which has freezing point 16.60 and latent heat of fusion 43 calories.
1. 3.3
2. 3.86
3. 2.9
4. 38.6
The van’t Hoff factor for a very dilute
solution of is
1. 9
2. 5
3. 2
4. 6
CsBr crystallizes in a body-centered cubic lattice. The unit cell length is 436.6pm. Given that the atomic mass of Cs u =133 and that of Br = 80 u and Avogadro number is , the density of CsBr is
1. 42.5 g/
2. 4.25 g/
3. 8.25 g/
4. 82.5 g/
According to Freundlich adsorption isotherm, at high pressure, the value of is -
1. Directly proportional to the pressure.
2. Inversely proportional to the pressure.
3. Directly proportional to the square of the pressure.
4. Independent of the pressure.
Ammonia is not a product in the
1. Hydrolysis of nitrolim
2. Hydrolysis of Aluminium nitride
3. Decomposition of Ammonium nitrite
4. Hydrolysis of urea
Phosphine can not be produced by the following reaction:
1. White P is heated with NaOH.
2. Red P is heated with NaOH.
3. Ca3P2 is heated with water.
4. Phosphorus trioxide is boiled with water.
If NaCl is doped with mol% of , the number of cation vacancies will be -
1.
2.
3.
4. 2
A compound that shows Schottky as well as Frenkel defect among the following is -
1. ZnS
2. AgBr
3. NaCl
4. AgCl
What will happen to the elevation in boiling point of a solution, if the weight of the solute dissolved is doubled but the weight of the solvent taken is halved?
1. Elevation in boiling point will become four times
2. Elevation in boiling point will become double
3. No change in elevation in boiling point
4. Elevation in boiling point will become half.
The free energy change for the decomposition reaction
The minimum potential difference needed to reduce will be
1. 1.874 V
2. -2.487
3. 960 V
4. 2.487 V
If hydrogen electrodes dipped in two solutions of pH = 4 and pH = 6 are connected by a salt bridge, the emf of the resulting cell is -
1. 0.177 V
2. 0.3 V
3. 0.118 V
4. 0.104 V
If equivalent conductance of 1 M is 12.8 and if the conductance of acetate ion and ion at infinite dilution are 42 and 288.42 respectively, its degree of dissociation is
1. 39%
2. 3.9%
3. 0.35%
4. 0.039%
If electrode is diluted 100 times, then the change in emf is
1. Increase of 59 mV
2. Decrease of 59 mV
3. Increase of 29.5 mV
4. Decrease of 29.5 mV
Among the following options, the gas that will be more readily adsorbed on the surface of charcoal is:
1.
2.
3.
4.
A current of 9.65 ampere flowing for 10 minutes deposits 3.0 g of the metal which is trivalent. The atomic mass of the metal is
1. 30
2. 150
3. 90
4. 289.5
The incorrect statement among the following is -
1. is more stable than
2. is less stable than but is more stable than
3. is less stable than
4. Pb(II) is a good reducing agent while Sn(II) is not
Which of the following statements is incorrect?
1. Nitric oxide becomes brown when released in air
2. solid phosphorous pentachloride exhibits some ionic character
3. Ammonia is a good complexing agent
4. gets hydrolysed readily
which of the following halides is most acidic?
1.
2. Sb
3. Bi
4.
The correct statement among the following is:
| 1. | If ∆0 < P, low spin state is more stable. |
| 2. | CO is a very weak ligand. |
| 3. | The colour of a complex depends only on the nature of metal ion. |
| 4. | CO is a weak base but strong ligand. |
The complex that does not have a tetrahedral shape is:
1.
2.
3.
4.
The complex that contains only one unpaired electron is:
1. [CoF6]3-
2. [Co(C2O4)3]3-
3. [Fe(CN)6]3-
4. [MnCl6]3-
sol can be most easily coagulated by
1.
2.
3.
4.
Which metal can be purified by zone refining?
1. Ni
2. Zr
3. In
4. Fe
The amount of ice that will separate out on cooling
a solution containing 50 g of ethylene glycol in 200
g of water to –9.3ºC will be -
[Kf for water = 1.86 K kg mol–1]
1. 18.9 g
2. 30.3 g
3. 21.9 g
4. 38.7 g
Which one of the following statements about unit is correct?
| 1. | unit has one peroxy linkage. |
| 2. | has two tetrahedral units joined through Cr—O— Cr. |
| 3. | The Cr — O — Cr bond angle is equal to 109°28′. |
| 4. | All of the above. |
Which one of the following is a diamagnetic ion?
| 1. | La3+ | 2. | Lu3+ |
| 3. | Ce4+ | 4. | All of the above. |
Iodide ion on oxidation in the basic medium by KMnO4
will give:
1.
2.
3.
4.
Liquation can be used for
1. Fe
2. Pb
3. Mn
4. Ag
Two-point charges each of charge +q are fixed at (+a, 0) and (-a, 0). Another positive point charge q placed at the origin is free to move along X-axis. The charge q at origin in equilibrium will have
1. maximum force and minimum potential energy.
2. minimum force and maximum potential energy.
3. maximum force and maximum potential energy.
4. minimum force and minimum potential energy.
A simple pendulum consists of a small sphere of mass m suspended by a thread of length l. The sphere carries a positive charge q. The pendulum is placed in a uniform electric field of strength E directed vertically upwards. With what period will the pendulum oscillate if the electrostatic force acting on the sphere is less than the gravitational force? Assume the oscillations to be small.
1.
2.
3.
4.
The charge flowing across the cell on closing the key K is equal to-
1. CV
2. CV/2
3. 2CV
4. zero
A point charge q moves from point P to S along the path PQRS in a uniform electric field pointing parallel to the positive direction of the x-axis. The coordinate of the point P, Q, R and S are (a, b, 0), (2a, 0, 0), (a, -b, 0) and (0, 0, 0) respectively. The work done by the field in the above process is given by the expression
A charge Q is distributed over two concentric hollow spheres of radii r and R (R >r) such that the surface densities are equal. Find the potential at the common centre.
1.
2.
3.
4.
The equivalent capacitance between A and B is (each of the capacitors obtained is of capacitance equal to C)
1.
2.
3.
4.
An isolated sphere of radius \(R\) contains a uniform volume distribution of positive charge. Which of the curve on the graph below correctly illustrates the dependence of the magnitude of the electric field of the sphere as a function of the distance \(r\) from its centre?

1. A
2. B
3. C
4. D
The current in a wire varies with time according to the equation \(I=(4+2t),\) where \(I\) is in ampere and \(t\) is in seconds. The quantity of charge which has passed through a cross-section of the wire during the time \(t=2\) s to \(t=6\) s will be:
| 1. | \(60\) C | 2. | \(24\) C |
| 3. | \(48\) C | 4. | \(30\) C |
A copper wire is stretched to make it 0.1 % longer. The percentage change in its resistance is
1. 0.2 % increase
2. 0.2% decrease
3. 0.1 % increase
4. 0.1 % decrease
The potential of point O in the steady state circuit shown is :
1. 11/12V
2. 18/11V
3. 16/9V
4. none of the above
An electrical cable of copper has just one wire of radius 9 mm. Its resistance is 5 . This single wire of cable is replaced by 6 different well-insulated copper wires each of radius 3 mm. The total resistance of the cable will now be equal to?
1. 7.5
2. 45
3. 90
4. 270
The Wheatstone bridge shown in the figure below is balanced when the uniform slide wire \(AB\) is divided as shown. Value of the resistance \(X\) is:

1. \(3~\Omega\)
2. \(4~\Omega\)
3. \(2~\Omega\)
4. \(7~\Omega\)
What is total resistance across terminals \(A\) and \(B\) in the following network?
| 1. | \(R\) | 2. | \(2R\) |
| 3. | \(\dfrac{3R}{5}\) | 4. | \(\dfrac{2R}{3}\) |
There are two points A and B on the extended axis of a 2 cm long bar magnet. Their distances from the centre of the magnet are x and 2x respectively. The ratio of magnetic fields at points A and B will be-
1. 8 : 1 (approximately)
2. 4 : 1 (approximately)
3. 4 : 1
4. 8 : 1
A magnet is parallel to a uniform magnetic field. If it is rotated by \(60^{\circ}\), the work done is \(0.8\) J. How much work is done in moving it \(30^{\circ}\) further?
1. \(0.8\times 10^{7}~\text{ergs}\)
2. \(0.4~\text{J}\)
3. \(8~\text{J}\)
4. \(0.8~\text{ergs}\)
A wire of length L carrying current I is bent into a circle of one turn. The field at the center of the coil is B1. A similar wire of length L carrying current I is bent into a square of one turn. The field at its center is B2. Then
1. B1 > B2
2. B1 < B2
3. B1 = B2
4. Nothing can be predicted
An electric current is flowing in a very long pin
as shown in the figure. The value of magnetic
flux density at point O will be-
1.
2.
3.
4.
A 6.28m long wire is turned into a coil of diameter
0.2m and a current of 1 amp. is passed in it. The
magnetic induction at its centre will be-
1. 6.28 × 10-5 Tesla
2. 0
3. 6.28 Tesla
4. 6.28 × 10-3 Tesla
An particle is moving in a magnetic field of
( ) tesla with a velocity of 5×105 m/s. The
magnetic force acting on the particle will be-
1. 3.2 × 10–13 dyne
2. 3.2 × 1013 N
3. 0
4. 3.2 × 10–13 N
A potential difference of 600 volts is applied across the plates of a parallel plate condenser placed in a magnetic field. The separation between the plates is 3 mm. An electron projected vertically upward parallel to the plates with a velocity of 2 × 106 m/s moves undeflected between the plates. The magnitude and direction of the magnetic field in the region between the condenser plates will be (in Wb/) (Given charge of electron = –1.6 × 10–19 coulomb).
1. 0.1, into the plane.
2. 0.2, into the plane.
3. 0.3, out of the plane.
4. 0.4, out of the plane.
An ammeter and a voltmeter are connected in series to a battery with an emf E = 6 volt . When a certain resistance is connected in parallel with voltmeter, the reading of latter decreases two times, where as the reading of the ammeter increasing the same number of times. What is ratio of resistance of voltmeter to resistance of ammeter?
1. 2 2. 1/2
3. 1/3 4. 3
A circular disc of radius 0.2 meter is placed in a uniform magnetic field of induction in such way that its axis makes an
angle of 60° with . The magnetic flux linked
with the disc is :-
1. 0.08 Wb
2. 0.01 Wb
3. 0.02 Wb
4. 0.06 Wb
A coil of resistance 400 is placed in a
magnetic field. If the magnetic flux (Wb)
linked with the coil varies with time t (sec) as
= 50 t2 + 4. The current in the coil at t = 2 sec is :
1. 2A 2. 1A
3. 0.5A 4. 0.1A
| I: | A small magnet takes a longer time in falling into a hollow metallic tube without touching the wall. |
| II: | There is an opposition to motion due to the production of eddy currents in a metallic tube. |
Choose the correct option for the above statements:
| 1. | Both I and II are True and II is the correct explanation for I. |
| 2. | Both I and II are True and II is not the correct explanation for I. |
| 3. | I is True but II is False. |
| 4. | I is False but II is True. |
A coil is wound of a frame of rectangular cross-section. If the linear dimensions of the frame are doubled and the number of turns per unit length of the coil remains the same, then the self inductance increases by a factor of:
| 1. | \(6\) | 2. | \(12\) |
| 3. | \(8\) | 4. | \(16\) |
An electric dipole with dipole moment \(\vec{p} = \left(3 \hat{i} + 4 \hat{j}\right) \times 10^{- 30}~\text{C-m}\) is placed in an electric field \(\vec{E} = 4000 \hat{i} ~\text{N/C}\). An external agent turns the dipole slowly until its electric dipole moment becomes \(\left(- 4 \hat{i} + 3 \hat{j}\right) \times 10^{- 30}~\text{C-m}\). The work done by the external agent is equal to:
1. \(4\times 10^{-28}~\text{J}\)
2. \(-4\times 10^{-28}~\text{J}\)
3. \(2.8\times 10^{-26}~\text{J}\)
4. \(-2.8\times 10^{-26}~\text{J}\)
The variation of potential with distance \(x\) from a fixed point is shown in the figure. The electric field at \(x=13~\text m\) is:

| 1. | \(7.5~\text{V/m}\) | 2. | \(-7.5~\text{V/m}\) |
| 3. | \(5~\text{V/m}\) | 4. | \(-5~\text{V/m}\) |
In the circuit diagram shown all the capacitors are in \(\mu \text{F} \). The equivalent capacitance between points, \(A\) & \(B\) is (in \(\mu \text{F} \)):

1. \(\frac{14}{5}\)
2. \(7.5\)
3. \(\frac{3}{7}\)
4. None of these
The equivalent resistance between points A
and B is-
1. 32.5
2. 22.5
3. 2.5
4. 42.5
A voltmeter of resistance \(660~\Omega\) reads the voltage of a very old cell to be \(1.32\) V while a potentiometer reads its voltage to be \(1.44\) V. The internal resistance of the cell is:
1. \(30~\Omega\)
2. \(60~\Omega\)
3. \(6~\Omega\)
4. \(0.6~\Omega\)
A rectangular loop carrying a current \(I_1,\) is situated near a long straight wire carrying a steady current \(I_2.\) If the wire is parallel to one of the sides of the loop and is in the plane of the loop as shown in the figure, then the current loop will:

1. move away from the wire.
2. move towards the wire.
3. remain stationary.
4. rotate about an axis parallel to the wire.
The magnetic field due to a straight conductor of a uniform cross-section of radius \(a\) and carrying a steady current is represented by:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| Assertion (A): | Magnetic flux linked with a closed surface is always zero. |
| Reason (R): | Magnetic monopole does not exist. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
A bar magnet is oscillating in the Earth's
magnetic field with a period T. What happens
to its period and motion if its mass is
quadrupled?
1. Motion remains S.H.M with time period =
2. Motion remains S.H.M with time period =2T
3. Motion remains S.H.M with time period =4T
4. Motion remains S.H.M and period remains
nearly constant
The magnetic flux through a circuit of resistance \(R\) changes by an amount \(\Delta \phi\) in time \(\Delta t.\) The total quantity of electric charge which passes during this time through any point of the circuit is given by:
1. \(Q= \frac{\Delta \phi}{\Delta t}\)
2. \(Q= \frac{\Delta \phi}{\Delta t}\times R\)
3. \(Q= -\frac{\Delta \phi}{\Delta t}\times R\)
4. \(Q= \frac{\Delta \phi}{R}\)
A metallic ring connected to a rod oscillates freely like a pendulum. If now a magnetic field is applied in horizontal direction so that the pendulum now swings through the field, the pendulum will
1. Keep oscillating with the old time period
2. Keep oscillating with a smaller time period
3. Keep oscillating with a larger time period
4. Come to rest very soon
In the following four situations, charged particles are at an equal distance from the origin. Arrange the magnitude of the net electric field at origin, starting with the highest.
 |
 |
| (i) | (ii) |
 |
 |
| (iii) | (iv) |
| 1. | (i) > (ii) > (iii) > (iv) | 2. | (ii) > (i) > (iii) > (iv) |
| 3. | (i) > (iii) > (ii) > (iv) | 4. | (iv) > (iii) > (ii) > (i) |
If two conducting spheres are separately charged and then brought in contact
1. The total energy of the two spheres is conserved
2. The total charge on the two spheres is conserved
3. Both the total energy and charge are conserved
4. The final potential is always the mean of the original potentials of the two spheres
A ring of radius 'R' having charge '2q' uniformly distributed over it passes through a sphere of same radius in such a way that centre of sphere lies at circumference of ring and centre of ring lies at surface of sphere. Then flux linked with sphere is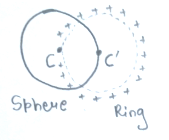
1.
2.
3.
4.
If a soap bubble is given some charge, then its radius:
| 1. | increases. |
| 2. | decreases. |
| 3. | remains unchanged. |
| 4. | may increase or decrease depending upon whether the given charge is positive or negative. |
The vertical component of the earth's magnetic field is zero at:
1. The magnetic poles.
2. The magnetic equator.
3. The geographic poles.
4. The geographic equator.
The variation of the intensity of magnetization (I) with respect to the magnetizing field (H) in a diamagnetic substance is described by which graph in the figure?
1. OD
2. OC
3. OB
4. OA
The number of turns in a coil of wire of fixed radius & length is \(600\) and its self-inductance is \(108\) mH. The self-inductance of a coil of \(500\) turns will be:
1. \(74\) mH
2. \(75\) mH
3. \(76\) mH
4. \(77\) mH
