During which stage of sewage treatment microbes are used?
1. Primary treatment
2. Secondary treatment
3. Tertiary treatment
4. All of these
Mendel’s work remained unrecognised till 1900. Which of the following was not a reason for this?
| 1. | His work was widely publicised and it brought bad name to Mendel |
| 2. | His concept of factors as stable and discrete units that did not ‘blend’ was not accepted. |
| 3. | His approach of using mathematics to explain biological phenomena was unacceptable. |
| 4. | He could not provide any physical proof for the existence of factors or say what they were made of. |
It is said that elemental composition of living organisms and that of inanimate objects (like earth’s crust) are similar in the sense that all the major elements are present in both. Then what would be the difference between these two groups? Choose a correct answer from among the following.
| 1. | Living organisms have more gold in them than inanimate objects |
| 2. | Living organisms have more water in their body than inanimate objects |
| 3. | Living organisms have more carbon, oxygen and hydrogen per unit mass than inanimate objects |
| 4. | Living organisms have more calcium in them than inanimate objects |
'Terror of Bengal' was introduced in India for its beautiful flowers and shape of leaves. It can grow vegetatively at a phenomenal rate through:
1. Offset
2. Stolon
3. Sucker
4. Rhizome
Read the following statements
| a. | Free nuclear division in developing embryo sac. |
| b. | Free nuclear division in PEN. |
| c. | Formation of heterogametes in Fucus. |
| d. | Chemotactic movement of sperms in Marchantia. |
| e. | Germination of seed within fruit when fruit is still attached to mother plant. |
How many of the above statements represent post-fertilization development?
| 1. | Two | 2. | Three |
| 3. | Four | 4. | One |
Parthenocarpic fruits lack:
1. Endocarp
2. Epicarp
3. Mesocarp
4. seed
Untranslated regions (UTRs) are present:
| 1. | before and after the stop codon |
| 2. | after the start codon and before the stop codon |
| 3. | before and after the start codon |
| 4. | before the start codon and after the stop codon |
a. Part marked as (B) has five types of histone proteins.
b. Part marked as (A) is rich in basic amino acid residues.
c. Part marked as (C) is made up of 400 nucleotides.
Which of the above statement(s) is/are incorrect?
1. a, c
2. a only
3. c only
4. a, b
Placed below is a karyotype of a human being.
On the basis of this karyotype, which of the following conclusions can be drawn?
1. Normal human female
2. Person is suffering from colour blindness
3. Affected individual is a female with Down’s syndrome
4. Affected individual is a female with Turner’s syndrome
Select the incorrect match:
| 1. | Universe | 13.8 billion years old |
| 2. | Earth | 4.5 million years back |
| 3. | UV rays | Broke up water into H2 and O2 |
| 4. | Louis Pasteur | Demonstrated that life comes only from pre-existing life |
It takes very long time for pineapple plants to produce flowers. Which combination of hormones can be applied to artificially induce flowering in pineapple plants throughout the year to increase yield?
1. Cytokinin and Abscisic acid
2. Auxin and Ethylene
3. Gibberellin and Cytokinin
4. Gibberellin and Abscisic acid
In 2000, maize hybrids had:
| 1. | Twice the amino acids lysine and tryptophan than existing maize hybrids were developed |
| 2. | Thrice the amino acids lysine and tryptophan than existing maize hybrids were developed |
| 3. | Half the amino acids glutamate and tryptophan than existing maize hybrids were developed |
| 4. | Twice the amino acids glutamate and tryptophan than existing maize hybrids were developed |
Pheretima and its close relatives derive nourishment from:
1. Sugarcane roots
2. Decaying fallen leaves and soil organic matter
3. Soil insects
4. Small pieces of fresh fallen leaves of maize
Match each item in Column-I with one item in Column-II regarding various classes of fungi and chose your answer from the codes given below:
| Column-I | Column-II | ||
| I. | Phycomycetes | 1. | Sac fungi |
| II. | Ascomycetes | 2. | Aseptate fungi |
| III. | Basidiomycetes | 3. | Imperfect fungi |
| IV. | Deuteromycetes | 4. | Puffballs |
Codes:
| I | II | III | IV | |
| 1. | 1 | 2 | 3 | 4 |
| 2. | 2 | 1 | 4 | 3 |
| 3. | 2 | 1 | 3 | 4 |
| 4. | 1 | 2 | 4 | 3 |
In nature, ……….. and ……….. provide enough energy to convert Nitrogen to nitrogen oxides (NO, NO2, N2O).
1. Lightning, Forest fires
2. Industrial combustions, UV radiation
3. Forest fire, Lightning
4. Lightning, UV radiation
Find the correct match with respect to gynoecium and placentation in the respective family:
| Column-I | Column-II | ||
| a. | \(G_{(3)}\) , Axile | i. | Brassicaceae |
| b. | \(G_{(2)}\) , Basal | ii. | Liliaceae |
| c. | \(G_{(2)}\) , Parietal | iii. | Solanaceae |
| d. | \(G_{(2)}\) , Axile | iv. | Asteraceae |
| 1. | a(i), b(ii), c(iv), d(iii) |
| 2. | a(ii), b(iv), c(i), d(iii) |
| 3. | a(ii), b(iv), c(iii), d(i) |
| 4. | a(iv), b(ii), c(i), d(iii) |
Chloroplast differs from mitochondria in:
| 1. | Having circular DNA and 70S ribosomes |
| 2. | The phase of division or duplication during cell cycle |
| 3. | Having porins in the outer membrane |
| 4. | Having enzymes for carbohydrates synthesis in the stroma |
Which of the following characteristics of living beings is not said to be true for worker bees?
1. Anabolism
2. Reproduction
3. Consciousness
4. Growth
Which one of the following is a case of wrong matching?
1. Root pressure - observable at night and early morning
2. Pressure potential - usually negative
3. Symplast - plasmodesmata
4. Loading of sugars at source - active transport
Small protein attached to the outer surface of inner mitochondrial membrane and acting as a mobile carrier is:
1. Cytochrome
2. Cytochrome
3. Cytochrome a
4. Cytochrome c
Photosynthetic active radiation (PAR) has which of the following range of wavelength?
1. 400-700 nm
2. 450-920 nm
3. 340-450 nm
4. 500-600 nm
Phloem in gymnosperms lacks:
| 1. | Both sieve tubes and companion cells |
| 2. | Albuminous cells and sieve cells |
| 3. | Sieve tubes only |
| 4. | Companion cells only |
Gemmae are present in:
| 1. | Some Gymnosperms | 2. | Some Liverworts |
| 3. | Mosses | 4. | Pteridophytes |
Which of the following is not correct?
| 1. | As the seed matures, its water content is reduced and seeds become relatively dry(10-15% moisture by mass) |
| 2. | The seed dormancy is the internal or innate inhibition of generation of normal or viable seeds |
| 3. | Embryo in dormant seed shows higher rate of general metabolic rate |
| 4. | Because of dormancy seeds remain viable for longer period and can be stored |
Which of the following is wrong about the diversity of Amazon Rain forest?
1. Mammals- 427
2. Fishes-3000
3. Birds-1300
4. Reptiles-278
The formulation with suitable preservatives in the case of drugs has to undergo:
1. Thorough clinical trials
2. Thorough strict separation techniques
3. Thorough harsh heat shock control
4. Thorough long restriction digestion
Which of the following organism is common in root ecosystems and acts as an effective biocontrol agent of several plant pathogens?
1. Nucleopolyhedrovirus
2. Lady bird beetle
3. Cactoblastis
4. Trichoderma
Find the set of incorrect statements:
| a. | RNA is labile, easily degradable, and reactive as compared to DNA |
| b. | Presence of thymine at the place of uracil confers additional stability to DNA |
| c. | Both DNA and RNA are able to mutate, viruses having DNA genome mutate and evolve faster |
| d. | DNA can easily express the characters directly |
| 1. | b & d | 2. | c & d |
| 3. | b & c | 4. | a & c |
In the equation GPP - R = NPP
R represents:
| 1. | Environment factor | 2. | Respiration losses |
| 3. | Radiant energy | 4. | Retardation factor |
Which of the following statements is correct?
1. Lichens do not grow in polluted areas.
2. Algal component of lichens is called mycobiont
3. Fungal component of lichens is called phycobiont
4. Lichens are not good pollution indicators.
Match the placental types (Column-I) with their examples (Column-II)
| Column-I | Column-II | ||
| (a) | Basal | (i) | Mustard |
| (b) | Axile | (ii) | China rose |
| (c) | Parietal | (iii) | Dianthus |
| (d) | Free central | (iv) | Sunflower |
Choose the correct answer from the following options:
| 1. | (a)-(ii), (b)-(iii),(c)-(iv), (d)-(i) |
| 2. | (a)-(i), (b)-(ii), (c)-(iii), (d)-(iv) |
| 3. | (a)-(iv), (b)-(ii), (c)-(i), (d)-(iii) |
| 4. | (a)-(iii), (b)-(iv), (c)-(i), (d)-(ii) |
Between which among the following, the relationship is not an example of commensalism?
1. Orchid and the tree on which it grows
2. Cattle Egret and grazing cattle
3. Sea Anemone and Clown fish
4. Female wasp and fig species
In angiosperms, microsporogenesis and megasporogenesis:
| 1. | form gametes without further divisions |
| 2. | Involve meiosis |
| 3. | occur in ovule |
| 4. | occur in anther |
Incomplete dominance is present in:
1. Snapdragon flower
2. Dog flower
3. Antirrhinum flower
4. All of the above
What will be the sequence of mRNA produced by the following stretch of DNA?
3' ATGCATGCATGCATG 5' TEMPLATE STRAND
5' TACGTACGTACGTAC 3' CODING STRAND
| 1. | 3' AUGCAUGCAUGCAUG 5' |
| 2. | 5' UACGUACGUACGUAC 3' |
| 3. | 3' UACGUACGUACGUAC 5' |
| 4. | 5' AUGCAUGCAUGCAUG 3' |
Choose the odd one with respect to composition of the cell wall of algae:
1. Cellulose, galactans
2. Mannans, minerals
3. Hemicellulose, pectin
4. Cellulose, minerals
Which of the following occurs naturally in plants?
1. Kinetin
2. NAA
3. IBA
4. 2,4 D
Flowers of aquatic plants like water hyacinth and water lily:
1. Are not pollinated by water
2. Emerge above the level of water for epihydrophily
3. Reach the surface of the water for hypohydrophily
4. Have exine in their pollen grains
Which of the following is true for the reproduction of bacteria?
1. Reproduce mainly by fission.
2. Under unfavourable conditions, they produce spores.
3. Also reproduce by sort of sexual reproduction.
4. All of these
Which of the following statement is correct about Leaf?
| 1. | It is attached to the stem by the leaf base |
| 2. | It always bears two lateral small leaf-like structures called stipules |
| 3. | All monocots have a small sheath-like leaf base that covers the stem wholly |
| 4. | All leguminous plants have swollen leaf base |
Read the following statements (with respect to angiosperms):
| (a) | Embryo sac formation is preceded by meiosis |
| (b) | Pollen grains germinate on the ovule and the resulting pollen tube grows through the tissues of stigma and style |
| (c) | A large group of plants occurring in a wide range of habitats |
| (d) | Synergids, antipodals and PEN degenerate after fertilization |
How many of the above statement(s) is/are wrong?
| 1. | Two | 2. | Three |
| 3. | Four | 4. | One |
Mark the correct statement (with respect to lateral meristem):
| 1. | Secondary meristem producing primary permanent tissues |
| 2. | Intercalary meristem producing secondary tissues |
| 3. | Cylindrical meristem producing the secondary tissues |
| 4. | Promeristem producing secondary permanent tissues |
Which of the following is an incorrect statement?
| 1. | Closely packed imbibant will imbibe more water than loosely packed one |
| 2. | During plasmolysis water is first lost from cytoplasm and then from vacuole |
| 3. | ψ s is always less than zero in solutions |
| 4. | Transpiration maintains the shape and structure of the plants by keeping cells turgid |
How many statements are correct with respect to ETS?
| (1) | ETS is helpful restoration of NAD+ & F A D+ |
| (2) | O2 is terminal e‐ acceptor in electron transport system |
| (3) | Ubiquinone & FMN are hydrogen carriers and helpful in the movement of H+ from matrix to space |
| (4) | Complex lV is also known as cytochrome C oxidase |
| (5) | Movement of e‐ in ETS is due to variable valency of metal ions present in carrier complexes |
1. 3
2. 4
3. 5
4. 2
Which of the following statements is incorrect?
| 1. | RuBisCO is a bifunctional enzyme |
| 2. | In C4 plants, the site of RuBisCO activity is mesophyll cell |
| 3. | The substrate molecule for RuBisCO activity is a 5-carbon compound |
| 4. | RuBisCO action requires ATP and NADPH |
Mycorrhiza promotes plant growth by:
1. Serving as a plant growth regulator
2. Absorbing inorganic ions from the soil
3. Helping the plant in utilizing atmospheric nitrogen
4. Protecting the plant from infection
Why is it necessary to remove sulfur from petroleum products?
| 1. | To reduce the emission of sulfur dioxide in exhaust fumes |
| 2. | To increase the efficiency of automobiles engines |
| 3. | To use sulfur removal from petroleum for commercial purposes |
| 4. | To increase the life span of engine silencers |
Match the following:
| (a) | Aquaporin | (i) | Amide |
| (b) | Asparagine | (ii) | Polysaccharide |
| (c) | Abscisic acid | (iii) | Polypeptide |
| (d) | Chitin | (iv) | Carotenoids |
Select the correct option:
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (ii) | (iii) | (iv) | (i) |
| 3. | (ii) | (i) | (iv) | (iii) |
| 4. | (iii) | (i) | (ii) | (iv) |
Which one of the following diseases is not due to contamination of water?
1. Hepatitis-B
2. Jaundice
3. Cholera
4. Typhoid
Select the correct statement about G1 phase:
| 1. | Cell is metabolically inactive |
| 2. | DNA in the cell does not replicate |
| 3. | It is not a phase of synthesis of macromolecules |
| 4. | Cell stops growing |
The special system of blood vessel giving blood to heart is known as:
1. coronary system
2. systemic system
3. pulmonary system
4. fibrous system
Which of the following is not an inactive enzyme?
1. Amylases
2. Nucleases
3. Lipases
4. Enterokinase
The wall of the alimentary canal from oesophagus to rectum possesses four layers namely serosa, mucosa, muscularis, sub mucosa. What is the correct sequence of layers from inside to outside?
| 1. | Mucosa, Sub-mucosa, Inner circular and Outer longitudinal muscles in Muscularis, Serosa |
| 2. | Muucosa, Sub-mucosa, inner longitudinal, Outer circular muscle i Muscularis, Serosa |
| 3. | Serosa, Outer longitudinal and Inner circular muscles in Muscularis, Submucosa and Mucosa |
| 4. | Serosa, outer circular and Inner longitudinal muscles in Muscularis, Submucosa and Mucosa |
The cytoskeleton is made up of:
1. calcium carbonate granules
2. callose deposits
3. cellulosic microfibrils
4. proteinaceous filaments
The sequence of amino acids forms the _________ structure of the protein. The left end amino acid is ___ terminal amino acid and the right end amino acid is ____ terminal amino acid.
1. Primary, N, C
2. Primary, C, N
3. Tertiary, N, C
4. Tertiary, C, N
Match List-I with List-II
| List-I | List-II | ||
| (a) | Metamerism | (i) | Coelenterata |
| (b) | Canal system | (ii) | Ctenophora |
| (c) | Comb Plates | (iii) | Annelida |
| (d) | Cnidoblasts | (iv) | Porifera |
Choose the correct answer from the options given below.
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (ii) | (i) |
| 2. | (iv) | (i) | (ii) | (iii) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (iii) | (iv) | (i) | (ii) |
Down's syndrome and Klinefelter's syndrome both can be included in:
1. Autosomal trisomy
2. Monosomy
3. Trisomy
4. Allosomal trisomy
| 1. | Chlorophyte ancestors \(\rightarrow\) Rhynia-type plants \(\rightarrow\) Seed ferns \(\rightarrow\) Progymnosperms |
| 2. | Progymnosperms \(\rightarrow\) Seed ferns \(\rightarrow\) Cycads \(\rightarrow\) Angiosperms |
| 3. | Sauropsids \(\rightarrow\) Thecodonts \(\rightarrow\) Therapsids \(\rightarrow\) Mammals |
| 4. | Psilophyton \(\rightarrow\) Progymnosperms \(\rightarrow\) Seed ferns \(\rightarrow\) Angisperms |
Which one of the following is the correct statement for respiration in humans?
| 1. | Cigarette smoking may not lead to inflammation of bronchi |
| 2. | Neural signals from pneumotoxic centre in pons region of brain can increase the duration of inspiration |
| 3. | Workers in grinding and stone breaking industries may suffer, from lung fibrosis |
| 4. | About 90% of carbon dioxide () is carried by haemoglobin as carbamino haemoglobin |
For a detailed evaluation of the heart’s function, multiple leads are attached to:
1. Wrist and arms
2. Abdomen
3. Chest region
4. All the fingers
Fill in the blanks in different columns of the table given below with respect to regulation of kidney function.
| Hormone | Factor stimulating its release | Function | |
| (i) | A | An excessive loss of fluid from the body | Prevents diuresis and caused increase in blood pressure |
| (ii) | Renin | B | Converts angiotensinogen in blood to angiotensin I |
| (iii) | C | An increase in blood flow to atria | Vasodilation decrease in blood pressure |
| (iv) | Angiotensin II | Fall in B.P. and blood volume | D |
The correct option for all the four blanks is
| A | B | C | D | |
| 1. | A.N.F. | Increase in blood volume | ADH | Stimulates the release of aldosterone |
| 2. | Vasopressin | Increase in blood pressure | ANF | Vasoconstrictor |
| 3. | ADH | Fall in glomerular В.Р. | ANF | Vasoconstrictor |
| 4. | Vasopressin | Increase in blood volume and blood pressure | ANF | Vasodilator |
Match the following columns and select the correct option:
| Column-I | Column -II | ||
| (a) | Rods and cones | (i) | Absence of Cones photoreceptor cells |
| (b) | Blind Spot | (ii) | Cones are densely packed |
| (c) | Fovea | (iii) | Photoreceptor cells |
| (d) | Iris | (iv) | Visible coloured portion of the eye |
| 1. | (a)-(iii), (b)-(i), (c)-(ii), (d)-(iv) |
| 2. | (a)-(ii), (b)-(iii), (c)-(i), (d)-(iv) |
| 3. | (a)-(iii), (b)-(iv), (c)-(ii), (d)-(i) |
| 4. | (a)-(ii), (b)-(iv), (c)-(iii), (d)-(i) |
Microbes found to be very useful in genetic engineering are:
1. Escherichia coli and Agrobacterium tumefaciens
2. Vibrio cholerae and a tailed bacteriophage
3. Diplococcus sp. and Pseudomonas sp.
4. Crown gall bacterium and Caenorhabditis elegans
Inbreeding:
1. Refers to the mating of distantly related individuals
2. Refers to the mating of more closely related individuals
3. Reduces homozygosity
4. Reduces inbreeding depression
The Green revolution increased the food supply to:
1. Triple fold
2. Double fold
3. Four fold
4. One and half fold
| 1. | Precipitation | 2. | Elution |
| 3. | Spooling | 4. | Fragmentation |
Which of the following is seen in Gout?
| 1. | Inflammation of joints due to accumulation of uric acid crystals |
| 2. | Inflammation of bones due to accumulation of uric acid crystals |
| 3. | Inflammation of joints due to accumulation of calcium |
| 4. | Inflammation of joints due to accumulation of estrogens |
Consider the following four conditions (I-IV) and select the correct pair of them as adaption to environment in desert lizards.
The conditions:
| I. | Burrowing in soil to escape high temperature |
| II. | Losing heat rapidly from the body during high temperature |
| III. | Bask in sun when temperature is low |
| IV. | Insulating body due to thick fatty dermis |
| 1. | (I) and (III) |
| 2. | (II) and (IV) |
| 3. | (I) and (II) |
| 4. | (III) and (IV) |
Menstrual bleeding is the result of:
1. low levels of ovarian hormones
2. high levels of ovarian hormones
3. high levels of LH
4. high levels of FSH
According to NCERT data how many recombinant therapeutics have been approved for human use and how many are marketed in India?
1. 12, 6
2. 32, 16
3. 30, 12
4. 12, 10
The base of canal is swollen and is called ampulla which contains a ……….. called ……..ampullaris which has …..
1. Projecting ridge, Crista, Hair cells
2. Projecting Furrows, macula, nerves
3. Projecting fissures, Crista, hair cell
4. Convolution, Crista, nerves
The detailed structure of the cell membrane was studied only after the advent of the:
1. compound microscope
2. cryo-electron microscopy
3. X-ray crystallography
4. electron microscope
Given below are four statements pertaining to the separation of DNA fragments using Gel electrophoresis. Identify the incorrect statements:
| (a) | DNA is a negatively charged molecule and so it is loaded on gel towards the Anode terminal. |
| (b) | DNA fragments travel along the surface of the gel whose concentration does not affect the movement of DNA. |
| (c) | Smaller the size of the DNA fragment, the larger is the distance it travels through it. |
| (d) | Pure DNA can be visualized directly by exposing to UV radiation. |
| 1. | (a), (c) and (d) |
| 2. | (a), (b) and (c) |
| 3. | (b), (c) and (d) |
| 4. | (a), (b) and (d) |
Which of the following contraceptive methods do involve a role of hormone?
| 1. | Pills, Emergency contraceptives, barrier methods |
| 2. | Lactational amenorrhea, Pills, Emergency contraceptives |
| 3. | Barrier method, Lactational amenorrhea, pills |
| 4. | CuT, Pills, Emergency contraceptives |
Tidal Volume and Expiratory Reserve Volume of an athlete are 500 mL and 1000 mL respectively. What will be his Expiratory Capacity if the Residual Volume is 1200 mL?
1. 2700 mL
2. 1500 mL
3. 1700 mL
4. 2200 mL
Match the following hormones with the respective disease:
| (a) | Insulin | (i) | Addison's disease |
| (b) | Thyroxin | (ii) | Diabetes insipidus |
| (c) | Corticoids | (iii) | Acromegaly |
| (d) | Growth Hormone | (iv) | Goitre |
| (v) | Diabetes mellitus |
Select the correct option:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iv) | (i) | (iii) |
| 2. | (v) | (i) | (ii) | (iii) |
| 3. | (ii) | (iv) | (iii) | (i) |
| 4. | (v) | (iv) | (i) | (iii) |
Match the following columns and select the correct option:
| Column I | Column II | ||
| (a) | 6 - 15 pairs of gill slits | (i) | Trygon |
| (b) | Heterocercal caudal fin | (ii) | Cyclostomes |
| (c) | Air Bladder | (iii) | Chondrichthyes |
| (d) | Poison sting | (iv) | Osteichthyes |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (i) | (ii) |
| 2. | (iv) | (ii) | (iii) | (i) |
| 3. | (i) | (iv) | (iii) | (ii) |
| 4. | (ii) | (iii) | (iv) | (i) |
The yellowish fluid "colostrum" secreted by mammary glands of the mother during the initial days of lactation has abundant antibodies (IgA) to protect the infant. This type of immunity is called as:
| 1. | Passive immunity | 2. | Active immunity |
| 3. | Acquired immunity | 4. | Autoimmunity |
Which of the following is associated with a decrease in cardiac output?
1. Sympathetic nerves
2. Parasympathetic neural signals
3. Pneumotaxic center
4. Adrenal medullary hormones
Select the correct statement:
| 1. | Atrial Natriuretic Factor increases the blood pressure. |
| 2. | Angiotensin II is a powerful vasodilator. |
| 3. | Counter current pattern of blood flow is not observed in vasa recta. |
| 4. | Reduction in Glomerular Filtration Rate activates JG cells to release renin. |
Match List-I with List-II:
| List-I | List-II |
| (a) Adaptive radiation | (i) Selection of resistant varieties due to excessive use of herbicides and pesticides |
| (b) Convergent evolution | (ii) Bones of forelimbs in Man and Whale |
| (c) Divergent evolution | (iii) Wings of Butterfly and Bird |
| (d) Evolution by anthropogenic action | (iv) Darwin Finches |
Choose the correct answer from the options given below.
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iv) | (iii) |
| 2. | (i) | (iv) | (iii) | (ii) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (iii) | (ii) | (i) | (iv) |
| Column I | Column II | |
| 1. | Ovulation | LH and FSH attain peak level and sharp fall in the secretion of progesterone |
| 2. | Proliferative phase | Rapid regeneration of myometrium and maturation of Graafian follicle |
| 3. | Development of corpus luteum | Secretory phase and increased secretion of progesterone |
| 4. | Menstruation | Breakdown of myometrium and ovum not fertilized |
Which of the following is correct?
| 1. | The chemical or metabolic conversion refers to a reaction. |
| 2. | The chemical which is converted into a product is called a substrate. |
| 3. | Proteins with three dimensional structures including an active site is called enzyme. |
| 4. | All of these |
The mechanism of breathing depends on the animal’s:
1. habitat
2. level of organisation
3. Both 1 and 2
4. Body segmentation
What percentage of ventricular filling is achieved by atrial contraction?
| 1. | 30 | 2. | 50 |
| 3. | 70 | 4. | 90 |
The patient of diabetes mellitus is characterized by:
1. Ketonuria
2. Glycosuria
3. Haematuria
4. Both 1 & 2
Consider the following statements:
| I. | Proximal radioulnar joint is a type of ellipsoidal joint. |
| II. | The first carpo-metacarpal joint is a saddle joint. |
| III. | Gleno-humeral joint is a ball and socket joint. |
Which of the above statements are true?
| 1. | I and II only |
| 2. | I and III only |
| 3. | II and III only |
| 4. | I, II and III |
Which of the following is incorrect about hormones?
1. Highly specific in nature
2. released into blood
3. nutrient chemicals
4. primary messenger
Mark the incorrect statement:
| 1. | Bipolar neurons (with one axon and one dendrite) are found in the retina of the eye |
| 2. | The gaps between two adjacent internodes are called nodes of Ranvier |
| 3. | Unmyelinated nerve fibres do not have myelin sheath around the axon |
| 4. | Nissl's granules are found in cyton and are absent in dendrites |
Which of the following is wrong?
| 1. | Spermatogonia and leydig cells are present in seminiferous tubules |
| 2. | Small blood vessels are present along with Leydig cells |
| 3. | Leydig cells secrete Androgens |
| 4. | Sertoli cell provide nutrition to the germ cell. |
Ovulation in the human female normally takes place during the menstrual cycle:
| 1. | At the mid secretory phase |
| 2. | At the end of proliferative phase |
| 3. | Just before the end of secretory phase |
| 4. | At the beginning of the proliferative phase |
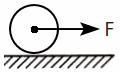
Select the correct statement with respect to figure given below:
| 1. | It is a sterilization method of contraception |
| 2. | Mode of action is similar to that of contraceptive pills and their effective period is much longer |
| 3. | Irreversible method of contraception |
| 4. | One of the most widely accepted methods of contraception in India |
Consider the following features:
(a) Organ system level of organisation
(b) Bilateral symmetry
(c) True coelomates with segmentation of the body
Select the correct option of animal groups that possess all the above characteristics:
1. Annelida, Mollusca and Chordata
2. Annelida, Arthropoda and Chordata
3. Annelida, Arthropoda and Mollusca
4. Arthropoda, Mollusca and Chordata
| a. | Spindle fibers attach to kinetochores of chromosomes during metaphase. |
| b. | Cell growth results in disturbing the ratio between the nucleus and the cytoplasm. |
| c. | Pachytene stage is relatively short-lived compared to the leptotene. |
| d. | Interkinesis is a short-lived stage characterized by duplication of DNA. |
| 1. | a, b & c are correct |
| 2. | Only c is incorrect |
| 3. | b & d are correct |
| 4. | a & b are correct |
How sickle cell anaemia is different from Thalassemia?
| 1. | Being autosomal recessive |
| 2. | Being a blood disease |
| 3. | Being a qualitative disorder |
| 4. | Involvement of mutation |
Industrial melanism is an example of:
1. Neo Darwinism
2. Natural Selection
3. Mutation
4. Neo Lamarckism
The IgG antibodies received by the developing fetus from the mother through the placenta will provide it with:
| 1. | Naturally acquired passive immunity |
| 2. | Artificially acquired passive immunity |
| 3. | Naturally acquired active immunity |
| 4. | Artificially acquired active immunity |
There is a restriction endonuclease called EcoRI. What does ‘co’ part in it stand for?
| 1. | Colon | 2. | Coelom |
| 3. | Coenzyme | 4. | coli |
On the basis of the data given below, the gas which shows the least adsorption on a definite amount of charcoal is-
|
Gas |
CO2 |
SO2 |
CH4 |
H2 |
|
Critical temp./K |
304 |
630 |
190 |
33 |
1. CO2
2. SO2
3. CH4
4. H2
Arrange the compounds in increasing order of rate of reaction towards nucleophilic substitution reaction:

1. (i) < (ii) < (iii)
2. (i) < (iii) < (ii)
3. (iii) < (ii) < (i)
4. (ii) < (iii) < (i)
The following reaction is named as:
\(\mathrm{Ar} \mathrm{N}_2^{+} \mathrm{Cl}^{-} \xrightarrow{\mathrm{Cu}/ \mathrm{HCl}} \mathrm{ArCl}+\mathrm{N}_2+\mathrm{CuCl}\)
1. Sandmeyer reaction
2. Gattermann reaction
3. Claisen reaction
4. Carbylamine reaction
The monomer unit present in the given polymer among the following is-
| 1. |  |
2. | |
| 3. |  |
4. |  |
Polyethyleneglycols are used in the preparation of which type of detergents?
1. Cationic detergents
2. Anionic detergents
3. Nonionic detergents
4. Soaps
Which is the correct statement among the following?
| 1. | Starch is a polymer of α-glucose |
| 2. | Amylose is a component of cellulose |
| 3. | Proteins are composed of only one type of amino acid |
| 4. | In the cyclic structure of fructose, there are four carbons and one oxygen atom |
The coordination numbers of Co and Al in [Co(Cl)2(en)2]Cl and K3[Al(C2O4)3], respectively, are :
(en = ethane-1, 2-diamine)
1. 3 and 3
2. 6 and 6
3. 5 and 3
4. 5 and 6
The correct statement among the following is :
| 1. | Leaching of bauxite using concentrated NaOH solution gives sodium aluminate and sodium silicate. |
| 2. | The Hall-Heroult process is used for the production of aluminium and iron. |
| 3. | Pig iron is obtained from cast iron. |
| 4. | The blistered appearance of copper during the metallurgical process is due to the evolution of CO. |
Aqua regia is used for dissolving noble metals (Au, pt, etc.). The gas evolved in this process is:
1. N2O5
2. N2O3
3. N2
4. NO
The increasing order of reactivity of the following compounds in nucleophilic addition reaction is:
Propanal, Benzaldehyde, Propanone, Butanone
1. Butanone < Propanone < Benzaldehyde < Propanal
2. Propanal < Propanone < Butanone < Benzaldehyde
3. Benaldehyde < Propanal < Propanone < Butanone
4. Benzaldehyde < Butanone < Propanone < Propanal
A diatomic molecule X2 has a body-centred cubic (bcc) structure with a cell edge of 300 pm. The density of the molecule is 6.17 g cm–3 . The number of molecules present in 200 g of X2 is-
(Avogadro constant (NA) = 6 × 1023 mol–1)
1. 8 NA
2. 40 NA
3. 4 NA
4. 2 NA
What is the final product (major) 'A' in the given reaction?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Hydrogen peroxide oxidises [Fe(CN)6]4– to [Fe(CN)6]3– in acidic medium but reduces [Fe(CN)6]3– to [Fe(CN)6]4– in alkaline medium. The other products formed are, respectively:
1. (H2O + O2) and H2O
2. (H2O + O2) and (H2O + OH–)
3. H2O and (H2O + O2)
4. H2O and (H2O + OH–)
| Statement I: | An allotrope of oxygen is an important intermediate in the formation of reducing smog. |
| Statement II: | Gases such as oxides of nitrogen and sulphur present in the troposphere contribute to the formation of photochemical smog. |
Choose the correct answer from the options given below:
1. Both Statement I and Statement II are false.
2. Statement I is true but Statement II is false.
3. Both Statement I and Statement II are true.
4. Statement I is false but Statement II is true.
Match the electronic configurations in List-I with their corresponding ionization energies in List-II.
| List-I (Electronic configuration of elements) |
List-II \(\mathbf {({(I.E)}_1 \text { in } \mathbf {kJ} \mathbf{~mol}^{-1})}\) |
||
| a. | 1s22s2 | (i) | 801 |
| b. | 1s22s22p4 | (ii) | 899 |
| c. | 1s22s22p3 | (iii) | 1314 |
| d. | 1s22s22p1 | (iv) | 1402 |
Choose the most appropriate answer from the options given below:
1. (a) (ii), (b) (iii), (c) (iv), (d) (i)
2. (a) (i), (b) (iv), (c) (iii), (d) (ii)
3. (a) (i), (b) (iii), (c) (iv), (d) (ii)
4. (a) (iv), (b) (i), (c) (ii), (d) (iii)
The orbital having two radial as well as two angular nodes is:
| 1. | 3p | 2. | 4f |
| 3. | 4d | 4. | 5d |
3.12 g of oxygen is adsorbed on 1.2 g of platinum metal. The volume of oxygen adsorbed per gram of the
adsorbent at 1 atm and 300 K in L is:
[R = 0.0821 L atm K–1 mol–1]
1. 4 L
2. 3 L
3. 1 L
4. 2 L
Consider separate solutions of 0.500 M C2H5OH(aq), 0.100 M Mg3(PO4)2(aq), 0.250 M KBr(aq) and 0.125 M Na3PO4(aq) at 25ºC. The correct statement about these solutions, assuming all salts to be strong electrolytes is:
| 1. | 0.100 M Mg3(PO4)2(aq) has the highest osmotic pressure |
| 2. | 0.125 M Na3PO4(aq) has the highest osmotic pressure |
| 3. | 0.500 M C2H5OH(aq) has the highest osmotic pressure |
| 4. | They all have the same osmotic pressure |
What is the ratio of the number of molecules of oxygen to nitrogen in a specific gaseous mixture if their mass ratio is 1:4?
| 1. | 7:32 | 2. | 1:8 |
| 3. | 3:16 | 4. | 1:4 |
Consider the following reaction :
\(a\mathrm{Cu}+b\mathrm{HNO}_{3} \rightarrow c\mathrm{Cu}\left(\mathrm{NO}_{3}\right)_{2}+d\mathrm{NO}+e\mathrm{H}_{2} \mathrm{O}\)
The values of a, b, and e in the reaction are, respectively:
1. 3, 8 and 4
2. 5, 2 and 8
3. 5, 2 and 16
4. 2, 5 and 8
The amount of water in litres that must be added to 1 litre of an aqueous solution of HCl with a pH of 1 to create an aqueous solution with pH of 2, is:
| 1. | 2.0 L | 2. | 9.0 L |
| 3. | 0.1 L | 4. | 0.9 L |
| Assertion (A): | Nitrogen and Oxygen are the main components in the atmosphere but these do not react to form oxides of nitrogen. |
| Reason (R): | The reaction between nitrogen and oxygen requires a high temperature. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
What is the major product formed in the following reaction?

| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The products obtained on heating LiNO3 will be:
1.
2.
3.
4.
Consider the reaction, 2A + B → Products.
When concentration of B alone was doubled, the half-life did not change. When the concentration of A alone was doubled, the rate increased by two times. The unit of rate constant for this reaction is:
1. L mol–1 s–1
2. no unit
3. mol L–1s–1
4. s–1
A metal having the strongest metallic bond among the following is:
| 1. | V | 2. | Fe |
| 3. | Cr | 4. | Sc |
In the structure of silicon dioxide:
| 1. | There are double bonds between silicon and oxygen atoms. |
| 2. | Silicon atom is bonded to two oxygen atoms. |
| 3. | Each silicon atom is surrounded by two oxygen atoms and each oxygen atom is bonded to two silicon atoms. |
| 4. | Each silicon atom is surrounded by four oxygen atoms and each oxygen atom is bonded to two silicon atoms. |
The value of the 'spin only' magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
1. d5 (in strong ligand field)
2. d3 (in weak as well as in strong fields)
3. d4 (in weak ligand field)
4. d4 (in strong ligand field)
Which of the following reactions are not expected to give 'A' in yields of more than 50%?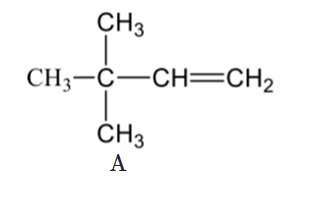
| 1. | 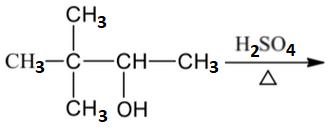 |
| 2. | 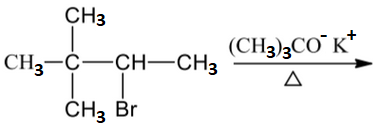 |
| 3. | 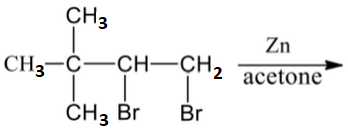 |
| 4. | None of the above |
For a given solution, pH = 6.9 at 60 ℃, where Kw = 10-12. The solution is:
| 1. | Acidic | 2. | Basic |
| 3. | Neutral | 4. | Unpredictable |
The final product 'C' of the following sequence of reactions is:
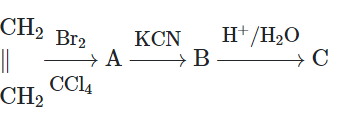
| 1. | \(\mathrm{CH_2-COOH\\ |\\ CH_2-COOH}\) | 2. | \(\mathrm{CH_2-Br\\ |\\ CH_2-Br}\) |
| 3. | \(\mathrm{CH_2-COOH\\ |\\ CH_2-CN}\) | 4. | \(\mathrm{CH_2-CN\\ |\\ CH_2-CN}\) |
For the following cell with hydrogen electrodes at two different pressures p1 and p2 , then the emf is given by:
Pt(H2) | H+(aq) |Pt (H2)
p1 1M p2
| 1. | \(\frac{R T}{F} \log _{e} \frac{P_{1}}{p_{2}}\) | 2. | \(\frac{R T}{2F} \log _{e} \frac{P_{1}}{p_{2}}\) |
| 3. | \(\frac{R T}{F} \log _{e} \frac{P_{2}}{p_{1}}\) | 4. | \(\frac{R T}{2F} \log _{e} \frac{P_{2}}{p_{1}}\) |
If a reaction is non-spontaneous at the freezing point of water but is spontaneous at the boiling point of water, then:
| \(\Delta H\) | \(\Delta S\) | |
| 1. | +ve | +ve |
| 2. | -ve | -ve |
| 3. | -ve | +ve |
| 4. | +ve | -ve |
Following types of compounds (as I, II) are studied in terms of isomerism in:
| (I) | CH3CH=CHCH3 |
| (II) |  |
1. Chain isomerism
2. Position isomerism
3. Conformers
4. Stereisomerism
The correct statement among the following is:
| 1. | When a covalent bond is formed, transfer of electrons takes place |
| 2. | Pure H2O does not contain any ion |
| 3. | A bond is formed when attractive forces overcome repulsive forces |
| 4. | HF is less polar than HBr |
| Assertion (A): | Λm for weak electrolytes shows a sharp increase when the electrolytic solution is diluted. |
| Reason (R): | For weak electrolytes degree of dissociation increases with a dilution of the solution. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
A brown ring is formed in the ring test for ion. It is due to the formation of:
1. [Fe(H2O)5(NO)]2+
2. FeSO4.NO2
3. [Fe(H2O)4 (NO)2]2+
4. FeSO4. HNO3
Which of the following complexes formed by Cu++ ions is most stable?
| 1. | Cu2+ + 4NH3 → [Cu(NH3)4]2+ logK = 11.6 |
| 2. | Cu2+ + 4CN- → [Cu(CN)4]2- logK = 27.3 |
| 3. | Cu2+ + 2en- → [Cu(en)2]2+ logK = 15 .4 |
| 4. | Cu2+ + 4H2O → [Cu(H2O)4]2+ logK = 8.9 |
IUPAC name of m-cresol is:
| 1. | 3-Methylphenol | 2. | 3-Chlorophenol |
| 3. | 3-Methoxyphenol | 4. | Benzene-1,3-diol |
Compounds A and C in the following reaction are:
\(\mathrm{\small{{CH}_3 {CHO} \xrightarrow[{(ii) ~H_{2}O}]{(i) ~CH_{3}MgBr}({A}) \stackrel{{H}_2 {SO}_4}{\longrightarrow}({B}) \xrightarrow[]{Hydroboration \ oxidation}(C)}}\)
1. Identical
2. Positional isomers
3. Functional isomers
4. Optical isomers
16 g of oxygen has the same number of molecules as in:
(a) 16 g of CO
(b) 28 g of N2
(c) 14 g of N2
(d) 1.0 g of H2
1. (a), (b)
2. (b), (c)
3. (c), (d)
4. (b), (d)
For the reaction \(2 A+B \rightarrow C,\) the values of initial rate at different reactant concentrations are given in the table below. The rate law for the reaction is:
| [A] (mol L-1) | [B] (mol L-1) | Initial Rate (mol L-1 s-1) |
| 0.05 | 0.05 | 0.045 |
| 0.10 | 0.05 | 0.090 |
| 0.20 | 0.10 | 0.72 |
| 1. | \(\text { Rate }=\mathrm{k}[A][B]\) | 2. | \(\text { Rate }=\mathrm{k}[\mathrm{~A}][B]^2\) |
| 3. | \(\text { Rate }=\mathrm{k}[A]^2[B]^2\) | 4. | \(\text { Rate }=\mathrm{k}[A]^2[B]\) |
The correct statement about ICl5 and ICl4- is:
| 1. | Both are isostructural |
| 2. | ICl5 is square pyramidal and ICl4- is square planar |
| 3. | ICl5 is trigonal bipyramidal and ICl4- is tetrahedral |
| 4. | ICl5 is square pyramidal and ICl4- is tetrahedral |
A solution is prepared by dissolving 0.6 g of urea (molar mass = 60 g mol–1) and 1.8 g of glucose (molar mass = 180 g mol–1) in 100 mL of water at 27 ºC. The osmotic pressure of the solution is:
(R = 0.08206 L atm K–1mol–1)
1. 8.2 atm
2. 2.46 atm
3. 4.92 atm
4. 1.64 atm
If the equilibrium constant for and that of , the equilibrium constant for is:
1.
2.
3.
4.
Lattice energy and enthalpy of the solution of NaCl are 788 kJ mol–1 and 4 kJ mol–1 , respectively. The hydration enthalpy of NaCl is:
1. –780 kJ mol–1
2. –784 kJ mol–1
3. 780 kJ mol–1
4. 784 kJ mol–1
The isostructural pairs among the following are:
| A. | \( \mathrm{SO}_{4}^{2-} and \ \mathrm{CrO}_{4}^{2-}\) |
| B. | \(\mathrm{SiCl}_{4} \ and \ \mathrm{TiCl}_{4}\) |
| C. | \(\mathrm{NH}_{3} \ and \ \mathrm{NO}_{3}^{-} \) |
| D. | \(\mathrm{BCl}_{3} \ and \ \mathrm{BrCl}_{3}\) |
1. C and D only
2. A and B only
3. A and C only
4. B and C only
The major product among the following reaction is:
| 1. | 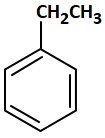 |
2. |  |
| 3. |  |
4. |  |
For the estimation of nitrogen, 1.4 g of an organic compound was digested by the Kjeldahl method and the evolved ammonia was absorbed in 60 mL of M/10 sulphuric acid. The unreacted acid required 20 mL of M/10 sodium hydroxide for complete neutralization. The percentage of nitrogen in the compound is:
1. 10%
2. 3%
3. 5%
4. 6%
What organic compound yields 17.6 g of 𝐶𝑂2 and 7.2 g of 𝐻2𝑂 when 5.6 g is combusted in excess oxygen?
1. C6H6
2. C4H8
3. C3H8
4. C2H2
The wavelength of the photon emitted by a hydrogen atom when an electron makes a transition from \(n=2\) to \(n=1\) state is:
1. \(194.8~\text{nm}\)
2. \(913.3~\text{nm}\)
3. \(490.7~\text{nm}\)
4. \(121.8~\text{nm}\)
A common transistor radio set requires \(12~\text{V}\) (D.C.) for its operation. The D.C. source is constructed by using a transformer and a rectifier circuit, which are operated at \(220~\text{V}\) (A.C.) on standard domestic A.C. supply. If the number of turns of secondary coil are \(24\), then the number of turns of primary are:
1. \(110\)
2. \(330\)
3. \(440\)
4. \(550\)
A person of \(80~\text{kg}\) mass is standing on the rim of a circular platform of mass \(200~\text{kg}\) rotating about its axis at \(5\) revolutions per minute (rpm). The person now starts moving towards the centre of the platform. What will be the rotational speed (in rpm) of the platform when the person reaches its centre?
| 1. | \(3\) | 2. | \(5\) |
| 3. | \(7\) | 4. | \(9\) |
The longitudinal strain of a string equals twice the magnitude of its lateral strain. What is Poisson's ratio of the material of the string?
1. \(0.4\)
2. \(0.5\)
3. \(0.1\)
4. \(0.2\)
A particle is oscillating under a force, \(\vec F=-k\vec x-b\vec v\) where \(k\) and \(b\) are constants. It shows:
1. linear oscillation
2. forced oscillation
3. damped oscillation
4. simple harmonic motion
| 1. | increased by a factor of \(2\) |
| 2. | decreased by a factor of \(2\) |
| 3. | unchanged |
| 4. | increased by a factor of \(4\) |
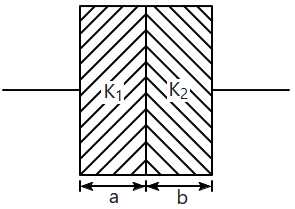
| 1. | \(\dfrac{{K}_{1}b}{{K}_{2}a}\) | 2. | \(\dfrac{{K}_{1}a}{{K}_{2}b}\) |
| 3. | \(\dfrac{{K}_{2}b}{{K}_{1}a}\) | 4. | \(\dfrac{{K}_{2}a}{{K}_{1}b}\) |
| 1. | Attracting magnetic substances |
| 2. | Hysteresis |
| 3. | Susceptibility independent of temperature |
| 4. | Directional property |
A magnet is made to oscillate with a particular frequency through a coil as shown in the figure. The time variation of the magnitude of emf generated across the coil during one cycle is:
| 1. | 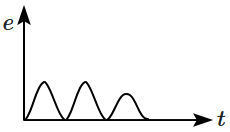 |
2. | 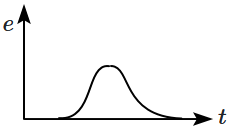 |
| 3. | 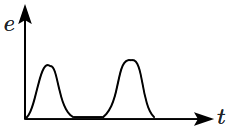 |
4. | 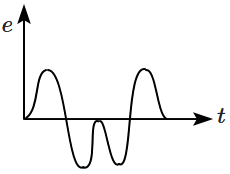 |
| 1. | 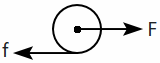 |
2. | 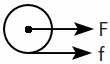 |
| 3. | 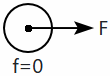 |
4. | cannot be interpreted |
| 1. | no beats |
| 2. | beats at a frequency of 64 Hz |
| 3. | beats at a frequency of 30 Hz |
| 4. | beats at a frequency of 32 Hz |
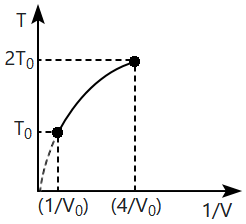
| 1. | X-rays | 2. | Visible rays |
| 3. | UV -rays | 4. | IR -rays |

| 1. | \(\dfrac{1}{\sqrt{3}}~\text{m/s}\) | 2. | \(\sqrt{3}~\text{m/s}\) |
| 3. | \(3\sqrt{3}~\text{m/s}\) | 4. | \(\dfrac{\sqrt{3}}{2}~\text{m/s}\) |
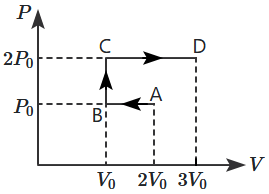
A particle is moving along a straight line such that its position depends on time as \(x=1-at+bt^{2} \), where \(a=2~\text{m/s}\), \(b=1~\text{m/s}^2\). The distance covered by the particle during the first \(3\) seconds from start of the motion will be:
| 1. | \(2~\text{m}\) | 2. | \(5~\text{m}\) |
| 3. | \(7~\text{m}\) | 4. | \(4~\text{m}\) |
The focal length of a convex lens is \(f\). An object is placed at a distance \(x\) from its second focal point. The ratio of the size of the real image to that of the object is:
| 1. | \(\dfrac{f}{x^2}\) | 2. | \(\dfrac{x^2}{f}\) |
| 3. | \(\dfrac{f}{x}\) | 4. | \(\dfrac{x}{f}\) |
| 1. | \(\dfrac{a}{c}{=}\dfrac{4}{3}\) | 2. | \(\dfrac{a}{c}{=}\dfrac{3}{4}\) |
| 3. | \(\dfrac{b}{d}{=}\dfrac{4}{3}\) | 4. | \(\dfrac{b}{d}{=}\dfrac{3}{4}\) |
| 1. | that the central maximum is wider. |
| 2. | more number of fringes. |
| 3. | less number of fringes. |
| 4. | no diffraction pattern. |
| 1. | 2p | 2. | \({2}{p}\cos\mathit{\theta}\) |
| 3. | \({2}{p}\sin\mathit{\theta}\) | 4. | \({2}{p}\tan\mathit{\theta}\) |
| 1. | \(2E\) | 2. | \(E/\sqrt{2}\) |
| 3. | \(E/2\) | 4. | \(E\) |
| 1. | \(\dfrac{1}{r}\) only | 2. | \(\dfrac{m}{r}\) |
| 3. | \( \left ( \dfrac{m}{r} \right )^{1/2} \) | 4. | \(m\) only |
| 1. | both remain unaffected |
| 2. | path of \(e^-\) will be more curved than the proton |
| 3. | path of proton will be more curved than electron |
| 4. | path of both particles will be equally curved |
| 1. | \(\mathit{\beta}\) times the base current |
| 2. | \(\mathit{\beta}\) times the emitter's current |
| 3. | \(\mathit{\alpha}\) times the base current |
| 4. | \({\mathit{\alpha}}^{2}\) times the base current |
The value of acceleration due to gravity at Earth's surface is \(9.8~\text{ms}^{-2}\). The altitude above its surface at which the acceleration due to gravity decreases to \(4.9~\text{ms}^{-2}\) is close to: (Radius of earth = \(6.4\times 10^6~\text{m}\))
| 1. | \( 6.4 \times 10^6 \mathrm{~m} \) | 2. | \( 2.6 \times 10^6 \mathrm{~m} \) |
| 3. | \( 1.6 \times 10^6 \mathrm{~m} \) | 4. | \( 9.0 \times 10^6 \mathrm{~m}\) |
A cricket ball of mass \(0.15~\text{kg}\) is thrown vertically up by a bowling machine so that it rises to a maximum height of \(20~\text{m}\) after leaving the machine. If the part pushing the ball applies a constant force \(F\) on the ball and moves horizontally a distance of \(0.2~\text{m}\) while launching the ball, the value of \(F\) (in N) is: \((g=10~\text{m/s}^2) \)
1. \(50 \mathrm{~N} \)
2. \(100 \mathrm{~N} \)
3. \(150 \mathrm{~N} \)
4. \(200 \mathrm{~N}\)
A particle starts from the origin \((0,0)\) at time \(t=0\) with an initial velocity of \(5\hat{j}\) m/s. It moves in the \(xy \text-\)plane under a constant acceleration of \((10 \hat{i}+4 \hat{j})\) m/s2. At some instant, its coordinates are \((20~\text{m},~y_0~\text{m}).\) The value of \(y_0\) is:
| 1. | \(52~\text{m}\) | 2. | \(25~\text{m}\) |
| 3. | \(18~\text{m}\) | 4. | \(24~\text{m}\) |
A closed vessel contains \(0.1\) mole of a monatomic ideal gas at \(200~\text{K}\). If \(0.05\) mole of the same gas at \(500~\text{K}\) is added to it, the final equilibrium temperature (in \(\text{K}\)) of the gas in the vessel is:
1. \(100~\text{K}\)
2. \(200~\text{K}\)
3. \(300~\text{K}\)
4. \(400~\text{K}\)
If two soap bubbles of different radii are connected by a tube:
| 1. | air flows from the bigger bubble to the smaller bubble till the sizes become equal. |
| 2. | air flows from the bigger bubble to the smaller bubble till the sizes are interchanged. |
| 3. | air flows from the smaller bubble to the bigger. |
| 4. | there is no flow of air. |
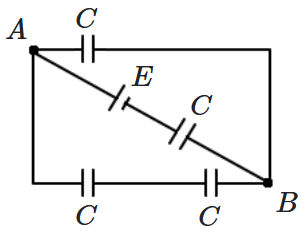
| 1. | \(E\) | 2. | \( \dfrac{2E}{3}\) |
| 3. | \( \dfrac{2E}{5}\) | 4. | zero |
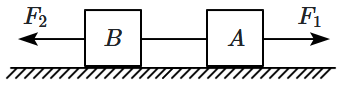
| 1. | the direction of friction on block A is towards right. |
| 2. | the direction of friction on block B may be towards left or right. |
| 3. | tension in the string must be zero. |
| 4. | friction force on block B must be zero. |
The figure shows two blocks of masses \(m\) and \(M\) connected by a string passing over a pulley. The horizontal table over which the mass \(m\) slides is smooth. The pulley has a radius \(r\) and moment of inertia \(I\) about its axis and it can freely rotate about this axis. The acceleration of the mass \(M\) assuming that the string does not slip on the pulley is:
| 1. | \(\dfrac{mg}{M+m+I/r^2}\) | 2. | \(\dfrac{Mg}{M+m+I/r^2}\) |
| 3. | \(\dfrac{Mg}{M+m+2I/r^2}\) | 4. | \(\dfrac{mg}{M+m+2I/r^2}\) |
| 1. | a wave traveling in the positive x-direction with a velocity \(1.5~\text{m/s} \) |
| 2. | a wave traveling in the negative x-direction with a velocity \(2.5~\text{m/s} \) |
| 3. | a wave traveling in the negative x-direction having a wavelength \(0.2~\text{m} \) |
| 4. | a wave traveling in the positive x-direction having a wavelength \(0.2~\text{m} \) |
| 1. | simple harmonic motion of frequency \(\mathit{\omega}{/}\mathit{\pi}\). |
| 2. | simple harmonic motion of frequency \({3}\mathit{\omega}{/}{2}\mathit{\pi}\). |
| 3. | non-simple harmonic motion. |
| 4. | simple harmonic motion of frequency \(\mathit{\omega}{/}{2}\mathit{\pi}\). |
| 1. | \(\dfrac{{E}_{e}}{{E}_{ph}}=\dfrac{2c}{v}\) | 2. | \(\dfrac{{E}_{e}}{{E}_{ph}}=\dfrac{v}{2c}\) |
| 3. | \(\dfrac{{P}_{e}}{{P}_{ph}}=\dfrac{2c}{v}\) | 4. | \(\dfrac{{P}_{e}}{{P}_{ph}}=\dfrac{v}{2c}\) |