| Column-I | Column-II | ||
| I. | Aspergillus niger | A. | Lactic acid |
| II. | Lactobacillus | B. | Butyric acid |
| III. | Clostridium butylicum | C. | Citric acid |
| IV. | Trichoderma polysporum | D. | Cyclosporin A |
| I | II | III | IV | |
| 1. | B | A | D | C |
| 2. | C | A | B | D |
| 3. | A | B | C | D |
| 4. | D | B | A | C |
In pea plant, the intermediate size of starch grains is due to:
1. Dominant epistasis
2. Codominance
3. Incomplete dominance
4. Recessive epistasis
Advantage of cleistogamy:
1. Higher genetic variability
2. More vigorous offspring
3. No dependence on pollinators
4. Vivipary
Given below are a few statements related to external fertilization. Choose the correct statements:
| 1. | The male and female gametes are formed and released simultaneously |
| 2. | Only a few gametes are released into the medium |
| 3. | Water is the medium in a majority of organisms exhibiting external fertilization |
| 4. | Offspring formed as a result of external fertilization have better chance of survival than those formed inside the organism |
| 1. | iii and iv |
| 2. | i and iii |
| 3. | ii and iv |
| 4. | i and iv |
In DNA replication, the two strands of a daughter DNA molecules are derived as:
| 1. | one strand from parent DNA and the other freshly synthesized |
| 2. | both strands from parent DNA |
| 3. | both strands synthesized as templates on parent strands |
| 4. | both strands synthesized on RNA templates of parent strands |
Which of the following is not correct with respect to salient features of the double helix structure of DNA?
| 1. | The plane of one base pair stacks over the other |
| 2. | The backbone is constituted by sugar-phosphate and the bases project outside |
| 3. | The two chains have anti-parallel polarity |
| 4. | The pitch of the helix is 3.4 nm |
Domestic wheat, which has 42 chromosomes, is probably hexaploid (6n), whereas the haploid number in the ancestral ones was 7. Find out the right reason as to how are such plants produced:
| 1. | Due to failure of segregation of chromatids during cell division cycle |
| 2. | Due to the gain of extra copy of chromosome |
| 3. | Due to failure of cytokinesis after telophase stage of cell division |
| 4. | Due to the loss of extra copy of chromosome |
Evolutionary convergence is the development of:
| 1. | common set of characters in the group of different ancestry |
| 2. | dissimilar characters in closely related groups |
| 3. | common set of characters in closely related groups |
| 4. | random mating |
The tobacco roots are infected by:
1. Nematode Meloidogyne incognita
2. Diptera beetles
3. Coleoptera fly
4. Lepidopterans
Which one of the following statements for the pyramid of energy is incorrect?
| 1. | It shows the energy content of different trophic-level organisms |
| 2. | It is inverted in shape |
| 3. | It is upright in shape |
| 4. | Its base is broad |
Select the incorrect statement with respect to SCP:
| 1. | Alternate source of proteins for human and animal nutrition |
| 2. | Can be grown on waste material |
| 3. | Decreases environmental pollution |
| 4. | Methylophilus methylotrophus can be expected to produce 250 tonnes of protein |
Which of the following statements are true for Heterotrophic bacteria?
| 1. | They fix nitrogen in legume roots. |
| 2. | The majority are important decomposers |
| 3. | Helpful in making curd from milk and production of antibiotics |
| 4. | All of these |
All the following plants are biennials except:-
1. Sugarbeet
2. Cabbages
3. Carrots
4. Wheat
Essential elements are grouped into _________ broad categories on the basis of function while these elements divided into _________ broad categories based on their quantitative requirements.
1. Four, One
2. Three, One
3. Four, Two
4. Three, Two
Select the incorrect option with respect to the modified structural name, modified organ and examples:
| Modified plant organ | Modified structures | Examples | |
| 1. | Tendril | Axillary bud | Gourds |
| 2. | Thorn | Axillary bud | Citrus |
| 3. | Phylloclade | Stem | Opuntia |
| 4. | Cladode | Stem | Australian Acacia |
Match the following and choose the correct option:
| Column I | Column II |
| A. Family | 1. Tuberosum |
| B. Kingdom | 2. Polymoniales |
| C. Order | 3. Solanum |
| D. Species | 4. Plantae |
| E. Genus | 5. Solanaceae |
| A | B | C | D | E | |
| 1. | 4 | 3 | 5 | 2 | 1 |
| 2. | 5 | 4 | 2 | 1 | 3 |
| 3. | 4 | 5 | 2 | 1 | 3 |
| 4. | 5 | 3 | 2 | 1 | 4 |
Find the incorrect statement:
| 1. | Middle lamella is mainly made up of calcium and magnesium pectate |
| 2. | Cell wall is formed on the inner side of the cell therefore secondary wall is formed first |
| 3. | Middle lamella glues the neighbouring cells together |
| 4. | Cell wall helps in cell to cell interaction and provides barrier to undesirable macromolecules |
Which of the following is not a part of Evil Quartet?
1. Habitat loss and Fragmentation
2. Dosage compensation
3. Alien Species Invasions
4. Co-extinctions
Genetically and functionally self-pollination is cent percent ensured with seed formation even in the absence of pollinators by:
| 1. | Bisexuality | 2. | Dicliny |
| 3. | Geitonogamy | 4. | Cleistogamy |
Read the following statements and choose the option which correctly marks T/F for given statements:
| a. | Viruses and some bacteria can transform eukaryotic cells and force them to do what the bacteria or viruses want. |
| b. | Recombinant bacterial colonies produce blue colour in presence of chromogenic substrate due to insertional inactivation. |
| c. | Disarmed pathogens are used to transfer the recombinant DNA into the host. |
| d. | Some divalent cations at specific concentration make the bacterial cell wall more porous. |
| a | b | c | d | |
| 1. | T | T | F | F |
| 2. | T | F | T | T |
| 3. | T | F | T | F |
| 4. | F | F | T | F |
The figure given below is a diagrammatic representation of lac operon. What does A, B, C and D represent respectively?
1. A - Inducer ; B - lac y ; C - Permease ; D - lac z
2. A - Repressor ; B - lac z ; C - Lactase ; D - lac a
3. A - Inducer ; B - lac z ; C - Permease ; D - lac a
4. A - Lactose ; B - lac y ; C - Permease ; D - lac a
In Kranz anatomy, the bundle sheath cells have:
| 1. | thin walls, many intercellular spaces, and no chloroplast |
| 2. | thick walls, no intercellular spaces, and a large number of chloroplasts |
| 3. | thin walls, no intercellular spaces, and several chloroplasts |
| 4. | thick walls, many intercellular spaces, and few chloroplasts |
Which of the following Algae shows anisogamy?
| 1. | Volvox | 2. | Eudorina |
| 3. | Spirogyra | 4. | Ulothrix |
Which of the following is not indicated by BOD test?
| 1. | Rate of uptake of oxygen by microorganisms in a sample of water |
| 2. | A measure of organic matter present in the water |
| 3. | Polluting potential |
| 4. | Amount of Flocs |
When protoxylem lies towards the centre (pith) and the metaxylem lies towards the periphery of the organ, the primary xylem is called?
| 1. | Endarch; present in stem |
| 2. | Endarch; present in roots |
| 3. | Exarch; present in stem |
| 4. | Exarch; present in roots |
Where is the respiratory electron transport system (ETS) located in plants?
1. Mitochondrial matrix
2. Outer mitochondrial membrane
3. Inner mitochondrial membrane
4. Intermembrane space
Xylem translocates:
| 1. | Water, minerals salts, some organic nitrogen and hormones |
| 2. | Water only |
| 3. | Water and mineral salts only |
| 4. | Water, mineral and some organic nitrogen only |
Which of the following ecological pyramids is generally inverted?
1. Pyramid of biomass in a sea
2. Pyramid of numbers in grassland
3. Pyramid of energy
4. Pyramid of biomass in a forest
Secondary metabolites such as nicotine, strychnine and caffeine are produced by plants for their:
| 1. | Growth response |
2. | Defence action |
| 3. | Effect on reproduction |
4. | Nutritive value |
Which of the following is incorrect about Cyanobacteria?
1. They are photoautotrophs
2. They lack heterocysts
3. They often form blooms in polluted water bodies
4. They have chlorophyll A similar to green plants
Which of the following statements is not correct?
| 1. | Some reptiles have also been reported as pollinators in some plant species. |
| 2. | Pollen grains of many species can germinate on the stigma of a flower, but only one pollen tube of the same species grows into the style. |
| 3. | Insects that consume pollen or nectar without bringing about pollination are called pollen/ nectar robbers. |
| 4. | Pollen germination and pollen tube growth are regulated by chemical components of pollen interacting with those of the pistil. |
Which one of the following is a correct statement?
| 1. | In the cymose type of inflorescence, the main axis continues to grow |
| 2. | The ovary is half inferior in the flowers of cucumber |
| 3. | In castor, the endosperm is not present in mature seeds |
| 4. | Seeds of dicot and monocot plants vary in shape, size and period viability |
How many possible genotypes can be observed in a human population for ABO blood group system?
1. 3
2. 6
3. 10
4. 4
| (a) | i gene | (i) | -galactosidase |
| (b) | Z gene | (ii) | Permease |
| (c) | A gene | (iii) | Repressor |
| (d) | Y gene | (iv) | transacetylase |
Select the correct option
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (iv) | (i) | (ii) |
| 2. | (i) | (iii) | (ii) | (iv) |
| 3. | (iii) | (i) | (ii) | (iv) |
| 4. | (iii) | (i) | (iv) | (ii) |
Which of the following is not a part of dissolved materials in waste water?
1. Nitrate
2. Ammonia, sodium
3. Phosphate, calcium
4. Bacteria
The first living cell was described by:
1. Anton Von Leeuwenhoek
2. Robert Brown
3. Robert Hooke
4. Both A and B
If a plant needs exposure to light less than a defined period for flowering, such a plant is called:
1. Long day plant
2. Short day plant
3. Day neutral plant
4. Far red light plant
Vegetative cell in a pollen grain:
1. Floats in the cytoplasm of the generative cell
2. Is small and spindle-shaped
3. Is larger than generative cell but lacks reserve food
4. Has a large irregularly shaped nucleus
What is absent in Euglenoids?
1. Two flagella, a short and a long one
2. Pellicle, a protein-rich layer
3. Pigments identical to those present in higher plants
4. Cell wall with stiff cellulose plates
Phyllotaxy is:
1. Pattern of arrangement of leaves on the stem or branch
2. Pattern of arrangement of petals on the stem or branch
3. Pattern of arrangement of sepals on the flower
4. Pattern of arrangement of perianth on the flower
The classification given by Bentham and Hooker is:-
1. Artificial
2. Natural
3. Phylogenetic
4. Numerical
All of the given characteristics are related to parenchyma tissue, except:
| 1. | Composed of living cells |
| 2. | Generally isodiametric cells |
| 3. | Cell wall is mainly made up of suberin |
| 4. | Either closely packed cells with no intercellular spaces or have small intercellular spaces |
Find the incorrect match with respect to transport in plants:
1. Simple diffusion – Do not require special membrane protein
2. Active transport – Do not show uphill transport
3. Facilitated transport – Transport saturation can occur
4. Active transport – Highly selective nature
Organisms that can survive a wide range of temperatures are called?
1. Ectotherms
2. Eurytherms
3. Endotherms
4. Stenotherms
Nuisance growth of aquatic plants and bloom-forming algae in natural water is generally due to high concentrations of:
1. carbon
2. sulfur
3. calcium
4. phosphorus
PGA as the first CO2 fixation was discovered in photosynthesis of:
1. bryophyte
2. gymnosperm
3. angiosperm
4. algae
In Krebs' cycle, GTP is formed in:
1. substrate level phosphorylation
2. oxidative phosphorylation
3. non-cyclic photophosphorylation
4. cyclic photophosphorylation
In which of the following groups, all three are examples of polysaccharides?
| 1. | Starch, glycogen, cellulose |
| 2. | Sucrose, maltose, glucose |
| 3. | Glucose, fructose, lactose |
| 4. | Galactose, starch, sucrose |
At which stage of mitotic cell division, the following characteristics are first seen:
| (a) | Chromosome as two stranded structure |
| (b) | Spindle fibre formation at poles |
| (c) | Formation of interzonal fibres (IZF) |
| 1. | (a)-Prophase, (b)-Metaphase, (c)-Anaphase |
| 2. | (a) and (b)-Prophase, (c)-Anaphase |
| 3. | (a)-Prophase, (b) and (c)-Metaphase |
| 4. | (a)-Prophase, (b)-Metaphase, (c)-Telophase |
Absorption is the process-
| 1. | By which end products of digestion pass through lumen to intestinal Mucosa |
| 2. | By which end products of digestion pass through intestinal Mucosa into the circulatory fluids of the body |
| 3. | By which water and essential nutrients are taken from the alimentary canal to lymph |
| 4. | More than one of the above options is correct |
Find the incorrect statement with respect to Kwashiorkor:
1. It occurs in children whose diets are deficient in protein
2. It occurs in children more than one year of age
3. Subcutaneous fat is preserved
4. Oedema is absent
| 1. | several ribosomes attached to a single mRNA |
| 2. | many ribosomes attached to a strand of endoplasmic reticulum |
| 3. | a ribosome with several subunits |
| 4. | ribosomes attached to each other in a linear arrangement |
Select the wrong statement:
| 1. | The substrate binds to the active site of enzymes. |
| 2. | Enzymes isolated from thermophilic organisms get denatured at 50 ° C |
| 3. | The active site of enzyme breaks the chemical bonds of the product. |
| 4. | Prosthetic groups are tightly bound to apoenzyme. |
Match the following:
| List I | List II |
| (a) Physalia (b) Limulus (c) Ancylostoma (d) Pinctada |
I. Pearl oyster II. Portuguese Man of War III. Living fossil IV. Hookworm |
Choose the correct answer from the options given below.
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (i) | (iv) | (iii) | (ii) |
| 3. | (ii) | (iii) | (i) | (iv) |
| 4. | (iv) | (i) | (iii) | (ii) |
Which of the following combination of chromosome numbers represents the correct sex determination pattern in honey bees?
1. Male 32, Female 16
2. Male 16, Female 32
3. Male 31, Female 32
4. Female 32, Male 31
Arrange the following events in a proper sequence:
| (i) | Increased ventricular pressure |
| (ii) | Attempted backflow of blood into the atria |
| (iii) | Ventricular systole |
| (iv) | Closure of AV valves |
| 1. | (i)-(ii)-(iii)-(iv) |
| 2. | (ii)-(i)-(iii)-(iv) |
| 3. | (iii)-(i)-(ii)-(iv) |
| 4. | (i)-(iii)-(ii)-(iv) |
Following is a tabular representation of differences between Juxtamedullary and Cortical nephron. Find out the correct difference.
| Feature | Juxtamedullary nephron | Cortical nephron | |
| 1. | Size | Smaller | Larger |
| 2. | Loop of Henle | Longer | Short |
| 3. | Presence | 85% of total nephrons | 15% of total nephrons |
| 4. | Function | Control plasma volume when water supply is normal | Control plasma volume when water supply is short |
Which of the following neurons are usually found in the embryonic stage?
| 1. | Multipolar | 2. | Unipolar |
| 3. | Bipolar | 4. | Pseudounipolar |
| 1. | 500 - 800 ml | 2. | 1000 - 1100 ml |
| 3. | 6000 -8000 ml | 4. | 2500 - 3000 ml |
Recombinant DNA technology involves several steps in which initial step is of isolation of the DNA. Which enzymes are used in the process for the breakdown of fungal cell, plant cell and bacterial cell respectively?
1. Lysozyme, lipases, trypsin
2. Chitinase, cellulase, lysozyme
3. Chitinase, cellulase, trypsin
4. Trypsin, lipases, cellulase
Select the incorrect statement regarding inbreeding:
| 1. | Inbreeding helps in the elimination of deleterious alleles from the population |
| 2. | Inbreeding is necessary to evolve a pure-line in any animal |
| 3. | Continued inbreeding reduces fertility and leads to inbreeding depression |
| 4. | Inbreeding depression cannot be overcome by out-crossing |
The ADA deficiency is because of:
| 1. | Substitution | 2. | Inversion |
| 3. | Deletion | 4. | Insertion |
| (a) | Resistance to ampicillin |
| (b) | Sensitivity to tetracycline |
| (c) | Resistance to tetracycline |
| (d) | Sensitivity to ampicillin |
| 1. | a and b |
| 2. | a and c |
| 3. | b and d |
| 4. | c and d |
_____ acts on ____ of nephron and helps in reabsorption of water and electrolytes.
1. ADH, PCT
2. ADH, DCT
3. ADH, Loop of Henle
4. None of these
Which of the following bone(s) is/are not a part of the axial skeleton?
1. Skull bones
2. Bones of vertebral column
3. Bones of sternum and ribs
4. Bones of pectoral and pelvic girdles
Which one of the following is categorized as a parasite in a true sense?
| 1. | Human foetus developing inside the uterus draws nourishment from the mother |
| 2. | Head louse living on the human scalp as laying eggs on human hair |
| 3. | The cuckoo (koel) lays its eggs in a crow's nest |
| 4. | The female Anopheles bites and sucks blood from humans |
| 1. | Ovulation | LH surge |
| 2. | Follicular phase of menstrual cycle | Regeneration of endometrium |
| 3. | Luteal phase | Secretion of FSH |
| 4. | Menstruation | Withdrawal of progesterone |
Nowadays it is possible to detect the mutated gene causing cancer by allowing the radioactive probe to hybridise its complementary DNA in clones of cells, followed by its detection using autoradiography because:
| 1. | mutated gene does not appear on a photographic film as the probe has no complementarity with it |
| 2. | mutated gene does not appear on photographic film as the probe has complementarity with it |
| 3. | mutated gene partially appears on a photographic film |
| 4. | mutated gene completely and clearly appears on a photographic film |
Which of the following is not the product of exocrine gland?
| 1. | Earwax | 2. | Oil |
| 3. | Mucus | 4. | Hormone |
Which of the following is true about nucleolus?
| 1. | membrane-less structure |
| 2. | active site for rRNA synthesis |
| 3. | numerous in cells which are involved in protein synthesis |
| 4. | All of the above |
When compared to the cones of the retina, the rods:
1. Are more sensitive to low intensity of light
2. Are most concentrated in the fovea
3. Are primarily involved in colour vision
4. Have higher visual acuity
Kwashiorkor disease is due to:
| 1. | simultaneous deficiency of proteins and fats |
| 2. | simultaneous deficiency of proteins and calories |
| 3. | deficiency of carbohydrates |
| 4. | protein deficiency not accompanied by calorie deficiency |
Which of the following equipments is essentially required for growing microbes on a large scale for industrial production of enzymes?
1. Bioreactor
2. BOD incubator
3. Sludge digester
4. Industrial oven
Match the source gland with its respective hormone and function and select the correct option:
| Source gland | Hormone | Function | |
| 1. | Anterior pituitary | Oxytocin | Contraction of uterine muscles |
| 2. | Anterior pituitary | Vasopressin | Induces reabsorption of water in nephron |
| 3. | Thymus | Thymosin | Proliferation of T-lymphocytes |
| 4. | α -cells of islets of Langerhans | Glucagon | Uptake of glucose into the cell |
What is true for an ideal contraceptive?
| I. | It should be user-friendly |
| II. | It should be easily available |
| III. | It should be ineffective and reversible with least side effects |
| IV. | It should be effective and reversible with least side effects. |
| V. | It should interfere with the sexual act of the user. |
| 1. | All of the above |
| 2. | I, II, III |
| 3. | I, II, IV |
| 4. | I, II, IV, V |
Bicarbonate is not reabsorbed by:
| 1. | PCT | 2. | DCT |
| 3. | Henle's Loop | 4. | All of these |
Serum differs from blood in:
1. lacking globulins
2. lacking albumins
3. lacking clotting factors
4. lacking antibodies
Enzymes enhance the rate of reaction by:
1. forming a reactant-product complex
2. changing the equilibrium point of the reaction
3. combining with the product as soon as it is formed
4. lowering the activation energy of the reaction
Which one of the following statements about certain given animals is correct?
1. Molluscs are acoelomates
2. Insects are pseudocoelomoates
3. Flatworms (Platyhelminthes) are coelomates
4. Roundworms (Aschelminthes) are pseudocoelomates.
Consider the following four statements (a-d) regarding kidney transplant and select the two correct ones out of these:
| (a) | Even if a kidney transplant is proper the recipient may need to take immuno suppressants for a long time. |
| (b) | The cell-mediated immune response is responsible for graft rejection. |
| (c) | The B- lymphocytes are responsible for the rejection of the graft. |
| (d) | The acceptance or rejection of a kidney transplant depends on specific interferons. |
The two correct statements are:
| 1. | (c) and (d) |
| 2. | (a) and (c) |
| 3. | (a) and (b) |
| 4. | (b) and (c) |
At a particular locus, the frequency A allele is 0.6 and that of allele a is 0.4. What would be the frequency of heterozygotes in a random mating population at equilibrium?
| 1. | 0.24 | 2. | 0.16 |
| 3. | 0.48 | 4. | 0.34 |
In alveoli,
1. pCO2 is high and pO2is low
2. pCO2 is low and pO2 is high
3. pCO2 is low and pO2 low
4. None of the above
Acrosome is:
1. Part of sperm head
2. Caps the anterior portion of haploid nucleus
3. Has enzymes for fertilization
4. All of these
Which of the following constitutes the conduction part of the respiratory system?
1. external nostrils to trachea
2. external nostrils to primary bronchioles
3. Respiratory bronchioles to alveoli
4. external nostrils to terminal bronchioles
Congestion of the lungs is one of the main symptoms in:
1. Hypotension
2. Coronary artery disease
3. Angina
4. Heart failure
Excretion of dilute urine requires:
| 1. | relative permeability of the distal tubule to water |
| 2. | transport of sodium and chloride ions out of the descending loop of Henle |
| 3. | the presence of ADH |
| 4. | impermeability of the collecting tubule to water |
Which of the following is an ovarian hormone?
1. estrogen
2. Progesterone
3. Inhibin
4. All of these
Limbic system comprises:
1. Hypothalamus and optic lobes
2. Hippocampus and amygdala
3. Hippocampus and cerebellum
4. Brain stem
Which of the following is mismatched regarding disorders of the muscular and skeletal system?
| 1. | Myasthenia gravis: Autoimmune disorder affecting neuromuscular junction can cause paralysis of skeletal muscles |
| 2. | Muscular dystrophy: Progressive degeneration of skeletal muscles mostly due to genetic disorder |
| 3. | Tetany: Rapid spasms in muscle due to low Ca2+ in body fluid |
| 4. | Osteoporosis: Inflammation of joints due to accumulation of uric acid crystals |
Sperm motility is facilitated by:
1. Middle piece
2. Neck
3. Tail
4. Acrosomal enzymes
The below diagram represents the ovum surrounded by few sperms.
Identify A, B, C and D :-
| 1. | A-Zona pellucida, B-Sperm, C-Cells of the corona radiata, D-Perivitelline space |
| 2. | A-Cells of corona radiata, B-Sperm, C-Zona pellucida, D-Perivitelline space |
| 3. | A-Perivitelline space, B-Sperm, C-Zona pellucida, D-Cells of the corona radiata |
| 4. | A-Perivitelline space, B-Sperm, C-Cells of corona radiata, D-Zona pellucida |
______ protects the user from contracting STIs and AIDS.
Fill in the blank with a suitable option:
1. Tubectomy
2. Condom
3. Progestasert
4. Withdrawal method
In which of the following phases do centromeres split and chromatids separate, and then chromatids move to opposite poles?
| 1. | Metaphase-l, Anaphase |
| 2. | Anaphase, Anaphase-l |
| 3. | Anaphase-l, Metaphase-ll |
| 4. | Anaphase, Anaphase-ll |
Which of the following groups of animals depicted in the diagrams given below are dioecious, coelomate, metamerically segmented and have closed circulatory systems?
Options
1. A only
2. A & B
3. C only
4. A,B & C
The percentage of base sequences that are dissimilar among humans is:
1. 99.9%
2. 0.01%
3. 0.10%
4. 1.01%
In the case of peppered moth (Biston betularia) the black-coloured form became dominant over the light-coloured form in England during the industrial revolution. This is an example of:
| 1. | natural selection whereby the darker forms were selected |
| 2. | appearance of the darker colour individuals due to very poor sunlight |
| 3. | protective mimicry |
| 4. | inheritance of darker colour character acquired due to the darker environment |
Which of the following statement is true for the structure given below?
| 1. | It interferes with the transport of the neurotransmitter dopamine |
| 2. | Useful in patients who have undergone surgery |
| 3. | Obtained from Cannabis sativa |
| 4. | It has a potent stimulating action on the nervous system |
Which one of the following techniques made it possible to genetically engineer living organisms?
1. Recombinant DNA techniques
2. X-ray diffraction
3. Heavier isotope labeling
4. Hybridization
| Assertion (A): | Acylation of amines gives a monosubstituted product whereas alkylation of amines gives a polysubstituted product. |
| Reason (R): | Acyl group sterically hinders the approach of further acyl groups. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Given below is a reaction sequence:
\(\begin{aligned} & CH_3CH_2Cl\xrightarrow{NaCN}X\xrightarrow{H_2/Ni}Y\\ &Y\xrightarrow{\text{Acetic Anhydride}}Z \end{aligned}\)
Z in the above reaction is:
1. CH3CH2CH2NHCOCH3
2. CH3CH2CH2NH2
3. CH3CH2CH2CONHCH3
4. CH3CH2CH2CONHCOCH3
Which one of the following polymers are prepared by addition polymerization?
1. Novolac
2. Dacron
3. Teflon
4. Nylon-66
The right option for the statement "Tyndall effect is exhibited by", is:
1. Starch solution
2. Urea solution
3. NaCl solution
4. Glucose solution
| I: | Aspirin and Paracetamol belong to the class of narcotic analgesics. |
| II: | Morphine and Heroin are non-narcotic analgesics. |
| 1. | I is correct but II is false. |
| 2. | I is incorrect but II is true. |
| 3. | Both I and II are true. |
| 4. | Both I and II are false. |
| Assertion (A): | The [Fe(NH3)2(CN)4]– shows geometrical isomerism. |
| Reason (R): | [Fe(NH3)2(CN)4]– also show optical isomerism. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Cast iron is used for the manufacture of :
1. Wrought iron and pig iron
2. Wrought iron and steel
3. Wrought iron, pig iron and steel
4. Pig iron, scrap iron and steel
If the boiling point of H2O is 373 K, the boiling point of H2S will be:
1. Greater than 300 K but less than 373 K
2. Less than 300 K
3. More than 373 K
4. Equal to 373 K
How many chiral center(s) is/are present in threonine?
| 1. | Zero | 2. | One |
| 3. | Two | 4. | Three |
The correct order of acid character of the following compounds is:
 |
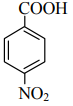 |
 |
 |
| (I) | (II) | (III) | (IV) |
| 1. | III > II > I > IV | 2. | IV > III > II > I |
| 3. | I > II > III > IV | 4. | II > III > IV > I |
The unit cell of copper corresponds to a face-centred cube of edge length 3.596 Å with one copper atom at each lattice point. The calculated density of copper in kg/m3 is-
[Molar mass of Cu : 63.54 g ; Avogadro Number = 6.022× 1023]
1. 9076
2. 9100
3. 8994
4. 9087
The correct order of the following compounds showing increasing tendency towards nucleophilic substitution reaction is:
 |
 |
||
| (i) | (ii) | (iii) | (iv) |
1. (iv) < (iii) < (ii) < (i)
2. (iv) < (i) < (ii) < (iii)
3. (iv) < (i) < (iii) < (ii)
4. (i) < (ii) < (iii) < (iv)
A ball weighing 10 g is moving with a velocity of 90 ms–1. If the uncertainty in its velocity is 5 %, then the uncertainty in its position is X × 10–33 m. The value of X will be :
[Given : h = 6.63 × 10–34 Js]
1. 1.17
2. 1.37
3. 1.67
4. 1.97
A car tyre is filled with nitrogen gas at 35 psi at 27 °C. It will burst if pressure exceeds 40 psi. The temperature in °C at which the car tyre will burst is-
(Rounded-off to the nearest integer)
(Hint: psi is unit of pressure)
1. 65
2. 70
3. 75
4. 77
The molarity of a solution obtained by mixing 750 mL of 0.5M HCl with 250 ml of 2M HCl will be:
1. 1.75 M
2. 0.975 M
3. 0.875 M
4. 1.00 M
The correct sequence of increasing the basic nature of the following oxides is:
1.
2.
3.
4.
The standard Gibbs energy change at 300 K for the reaction is 2494.2 J. At a given time, the composition of the reaction mixture is . The reaction proceeds in the:[assume R=8.314 J/K/mol; e-1 = 0.37]
| 1. | Forward direction because Q > KC |
| 2. | Reverse direction because Q > KC |
| 3. | Forward direction because Q < KC |
| 4. | Reverse direction because Q < KC |
Identify the incorrect statement from the following:
| 1. | Ozone absorbs the intense ultraviolet radiation of the sun. |
| 2. | Depletion of ozone layer is because of its chemical reactions with chlorofluorocarbon. |
| 3. | Ozone absorbs infrared radiation. |
| 4. | Oxides of nitrogen in the atmosphere can cause the depletion of ozone layer. |
Among the following statements about water, the FALSE statement is-
| 1. | Water is oxidized to oxygen during photosynthesis |
| 2. | Water can act both as an acid and as a base |
| 3. | There is extensive intramolecular hydrogen bonding in the condensed phase |
| 4. | Ice formed by heavy water sinks in normal water |
The pair in which phosphorous atoms have a oxidation state of +3 is :
1. Orthophosphorous and pyrophosphorous acids
2. Pyrophosphorous and hypophosphoric acids
3. Orthophosphorous and hypophosphoric acids
4. Pyrophosphorous and pyrophosphoric acids
The best description of the change that occurs when Na2O(s) are dissolved in water is-
1. Oxide ion accepts a shared pair of electrons
2. Oxide ion donates a pair of electrons
3. Oxidation number of oxygen increases
4. Oxidation number of sodium decreases
The major product obtained on interaction of phenol with sodium hydroxide and carbon dioxide is:
1. Benzoic acid
2. Salicylaldehyde
3. Salicylic acid
4. Phthalic acid
The point of dissimilarity between lanthanides and actinides is:
| 1. | Three outermost shells are partially filled. |
| 2. | They show oxidation state of +3 (common). |
| 3. | They are called inner transition elements. |
| 4. | They are radioactive in nature. |
The oxidation states of two S-atoms in are:
1. +2 and +4
2. +3 and -2
3. +4 and -2
4. +6 and -2
A reaction was found to be second order with respect to the concentration of carbon monoxide. If the concentration of carbon monoxide is doubled, with everything else kept the same, the rate of reaction will:
1. Remain unchanged
2. Triple
3. Increases by a factor of 4
4. Double
The structure of Al2Cl6 contains:
1. Four 2c-2e bonds and two 3c-2e bonds
2. Four 2c-2e bonds and four 3c-3e bonds
3. Four 2c-2e bonds and two 3c-4e bonds
4. Two 2c-2e bonds and four 3c-4e bonds
The aromatic compound among the following is:
| 1. | 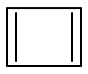 |
2. | 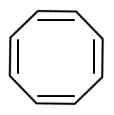 |
| 3. | 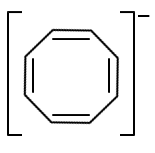 |
4. | 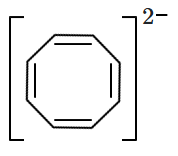 |
The complex that does not involve inner orbital hybridisation among the following is:
1. [CoF6]3-
2. [Co(NH3)6]3+
3. [Fe(CN)6]3-
4. [Cr(NH3)6]3+
For preparing a buffer solution of pH 6 by mixing sodium acetate and acetic acid, the ratio of concentration of salt and acid (Ka = 10−5) should be:
| 1. | 1 : 10 | 2. | 10 : 1 |
| 3. | 100 : 1 | 4. | 1 : 100 |
Complete the following reaction:
| 1. | 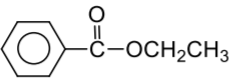 |
| 2. | 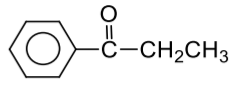 |
| 3. | 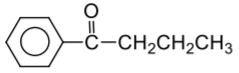 |
| 4. | 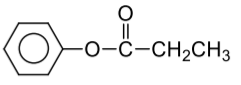 |
The potential of the cell containing two hydrogen electrodes as given below is:
| 1. | 2. | ||
| 3. | 4. |
The pair of species that has identical shapes among the following is:
| 1. | CF4, SF4 | 2. | XeF2, CO2 |
| 3. | BF3, PCl3 | 4. | PF5, IF5 |
If at 298 K the bond energies of C-H, C-C, C = C and H-H bonds are respectively 414, 347, 615, and 435 kJ mol–1, the value of enthalpy change for the reaction at 298 K will be:
\(\mathrm{H}_{2} \mathrm{C}=\mathrm{CH}_{2}(g)+\mathrm{H}_{2}(g) \longrightarrow \mathrm{H}_{3} \mathrm{C}-\mathrm{CH}_{3}(g)\)
1. +250 kJ
2. –250 kJ
3. +125 kJ
4. –125 kJ
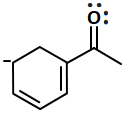
Which of the following are peroxoacids of sulphur?
| 1. | \(\mathrm{H_{2} SO_{5} } \text{ and } \mathrm{H_{2} S_{2} O_{8}}\) | 2. | \(\mathrm{H_{2} SO_{5 }} \text{ and } \mathrm{H_{2} S_{2} O_{7}}\) |
| 3. | \(\mathrm{H_{2} S_{2} O_{7}} \text{ and } \mathrm{H_{2} S_{2} O_{8}}\) | 4. | \(\mathrm{H_{2} S_{2} O_{6}} \text{ and } \mathrm{H_{2} S_{2} O_{7}}\) |
The colour of the coordination compounds depends on the crystal field splitting. What will be the correct order of absorption of the wavelength of light in the visible region for the complexes?
1. [Co(CN)6]3– > [Co(NH3)6]3+ > [Co(H2O)6]3+
2. [Co(CN)6]3– > [Co(H2O)6]3+ > [Co(NH3)6]3+
3. [Co(H2O)6]3+ > [Co(NH3)6]3+ > [Co(CN)6]3–
4. [Co(NH3)6]3+ > [Co(CN)6]3– > [Co(H2O)6]3+
Through which of the following reactions number of carbon atoms can be increased in the chain?
a. Grignard reaction
b. Cannizzaro’s reaction
c. Aldol condensation
d. HVZ reaction
Choose the correct option:
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)
The compound that gives p-cresol with p-methyl diazonium chloride is:
| 1. | H2O | 2. | H3PO2 |
| 3. | HCOOH | 4. | C6H5OH |
Which molecule among the following is non-polar?
| 1. | SbCl5 | 2. | NO2 |
| 3. | POCl3 | 4. | CH2O |
A solution of Ni(NO3)2 is electrolysed between platinum electrodes using 0.1 Faraday electricity. The number of moles of Ni that will be deposited at the cathode are:
| 1. | 0.10 | 2. | 0.05 |
| 3. | 0.20 | 4. | 0.15 |
What is the molality of a solution containing 20% (mass/mass) KI in water?
(molar mass of KI = 166 g mol–1)
1. 1.51
2. 7.35
3. 4.08
4. 2.48
NO2 required for a reaction is produced by the decomposition of N2O5 in CCl4 as per the equation,
2N2O5(g) 4NO2(g) + O2(g)
The initial concentration of N2O5 is 3.00 mol L–1, and it is 2.75 mol L–1 after 30 minutes. The rate of formation of NO2 is:
1. 2.083 × 10–3 mol L–1 min-1
2. 8.333 × 10–3 mol L–1 min-1
3. 4.167 ×10–3 mol L–1 min-1
4. 1.667 × 10–2 mol L–1 min-1
The mole fraction of glucose (C6H12O6) in an aqueous binary solution is 0.1. The mass percentage of water in it to the nearest integer is:
| 1. | 44% | 2. | 49 % |
| 3. | 47 % | 4. | 41 % |
If, for a dimerization reaction, 2A(g) → A2(g) at 298 K , ∆UΘ = -20 kJ mol-1 ∆SΘ = - 30 J K-1mol-1 , then ∆GΘ will be:
1. -10. 4 kJ
2. 18.9 kJ
3. -13.5 kJ
4. 17. 4 kJ
For a reaction; , 1.0 mol of X, 1.5 mol of Y and 0.5 mol of Z were taken in a 1 L vessel and allowed to react. At equilibrium, the concentration of Z was 1.0 mol L–1 . The equilibrium constant of the reaction is . The value of x is:
1. 24
2. 13
3. 16
4. 19
According to molecular orbital theory, which of the following molecules is not expected to be stable?
| 1. | \(\mathrm{He}_2^{2+}\) | 2. | \(\mathrm{He}_2^{+}\) |
| 3. | \(\mathrm{H}_2^{-}\) | 4. | \(\mathrm{H}_2^{2-}\) |
The least resonance stabilized molecule among the following is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The order of stability of the following carbocations is:
 |
 |
|
| (I) | (II) | (III) |
1. I > II > III
2. III > I > II
3. III > II > I
4. II > III > I
The order of stability of the following carbanions is:
| 1. I>II>III>IV | 2. I>III>II>IV |
| 3. IV>III>II>I | 4. III>IV>I>II |
In Bohr model of hydrogen atom, the ratio of periods of revolution of an electron in \(n=2\) and \(n=1\) orbits is:
1. \(2:1\)
2. \(4:1\)
3. \(8:1\)
4. \(16:1\)
A cord is wound round the circumference of wheel of radius r. The axis of the wheel is horizontal and the moment of inertia about it is I. A weight mg is attached to the cord at the end. The weight falls from rest. After falling through a distance 'h', the square of angular velocity of wheel will be :
1.
2.
3. 2gh
4.
Kinetic energy of an ideal gas is proportional to:
1. \(T\)
2. \(1 \over T\)
3. \(T^0\)
4. \(T^2\)
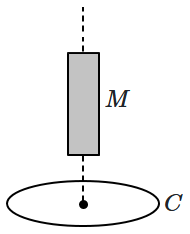
| 1. | \( v>2 g \) | 2. | \( v<g \) |
| 3. | \( s>2 g \) | 4. | \( s<2 g\) |
| 1. | \(\vec{\mathbf{E}}\) and \(\vec{\mathbf{B}}\) vectors vary sinusoidally in the same phase. |
| 2. | \(\vec{\mathbf{E}}\) and \(\vec{\mathbf{B}}\) vectors vary sinusoidally in the opposite phase. |
| 3. | \(\vec{\mathbf{E}}\) and \(\vec{\mathbf{B}}\) vectors contribute to energy equally. |
| 4. | \(\vec{\mathbf{E}}\) and \(\vec{\mathbf{B}}\) vectors vibrate orthogonally with the same frequencies. |
| 1. | \(1.5~\text{m}\) in the direction of displacement of the man. |
| 2. | \(0.75~\text{m}\) in the direction of displacement of the man. |
| 3. | \(1.5~\text{m}\) in the direction opposite to the displacement of the man. |
| 4. | \(0.75~\text{m}\) in the direction opposite to the displacement of the man. |
On which of the following temperature scales is the temperature never negative?
1. Celsius
2. Fahrenheit
3. Reaumur
4. Kelvin
| 1. | \(\frac{1}{2},0,\frac{1}{2}\) | 2. | \(\frac{1}{2},\frac{1}{2},0\) |
| 3. | \(\frac{1}{2}{,}{-}\frac{1}{2}{,}{0}\) | 4. | \(\frac{1}{2},0,1\) |
| 1. | diffraction of electron beam from a single slit. |
| 2. | diffraction of electron beam from crystals. |
| 3. | interference of electron beam in a double-slit experiment. |
| 4. | formation of standing waves between the lattice of crystals. |
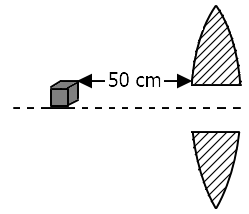
| 1. | Image/images formed are real |
| 2. | Image/images formed are magnified |
| 3. | Two images are formed |
| 4. | Image/images formed are virtual |
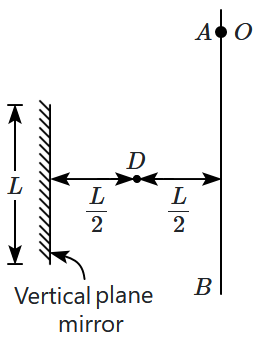
| 1. | \(\sqrt{\dfrac{6L}{g}}\) | 2. | \(\sqrt{\dfrac{2L}{g}}\) |
| 3. | \(\sqrt{\dfrac{3L}{g}}\) | 4. | \({t}\rightarrow\infty\) |
| 1. | \(25~\text{m}\) | 2. | \(55~\text{m}\) |
| 3. | \(65~\text{m}\) | 4. | \(75~\text{m}\) |
| 1. | \(800\) | 2. | \(200\) |
| 3. | \(600\) | 4. | \(400\) |
| 1. | \(\sqrt{\dfrac{3g}{L}}\) | 2. | \(\sqrt{3gL}\) |
| 3. | \(\sqrt{\dfrac{L}{3g}}\) | 4. | \(\sqrt{\dfrac{g}{3L}}\) |
| 1. | Newton's first law |
| 2. | Newton's second law |
| 3. | Special theory of relativity |
| 4. | Conservation of angular momentum |
| 1. | \(6f\) | 2. | \(f/\sqrt{6}\) |
| 3. | \(\sqrt{6}f\) | 4. | \(f\) |
| 1. | \(\left({{\mathit{\lambda}}_{1}/{\mathit{\lambda}}_{2}}\right)\) | 2. | \(\left({{\mathit{\lambda}}_{2}/{\mathit{\lambda}}_{1}}\right)\) |
| 3. | \(\left({{\mathit{\lambda}}_{1}^{2}/{\mathit{\lambda}}_{2}^{2}}\right)\) | 4. | \(\left({{\mathit{\lambda}}_{2}^{2}/{\mathit{\lambda}}_{1}^{2}}\right)\) |
| (I) | In forward bias |
| (II) | In reverse bias |
| (III) | In parallel with the load |
| (IV) | In series with the load |
| 1. | (I) and (II) only |
| 2. | (II) and (III) only |
| 3. | only (I) |
| 4. | only (IV) |
| 1. | IB against VBE at constant VCE |
| 2. | IC against VCE at constant VBE |
| 3. | IB against IC |
| 4. | IE against IC |
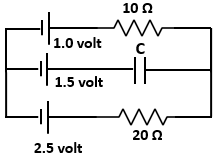
| 1. | \(nG\) | 2. | \((n-1)G\) |
| 3. | \(G/n\) | 4. | \(G/(n-1)\) |
The ratio of the weights of a body on the Earth’s surface to that on the surface of a planet is \(9:4\). The mass of the planet is \(\left(\dfrac{1}{9}\right)^\text{th} \) that of the Earth. If \(R\) is the radius of the Earth, what is the radius of the planet? (Take the planets to have the same mass density)
| 1. | \(\dfrac R9\) | 2. | \(\dfrac R2\) |
| 3. | \(\dfrac R 3\) | 4. | \(\dfrac R 4\) |
A block of mass \(1.9~\text{kg}\) is at rest at the edge of a table of height \(1~\text{m}\). A bullet of mass \(0.1~\text{kg}\) collides with the block and sticks to it. If the velocity of the bullet is \(20~\text{m/s}\) in the horizontal direction just before the collision, then the kinetic energy just before the combined system strikes the floor is:
(Take \(g=10~\text{m/s}^2\). Assume there is no rotational motion and loss of energy after the collision is negligible.)
1. \(21~\text{J}\)
2. \(23~\text{J}\)
3. \(20~\text{J}\)
4. \(19~\text{J}\)
The following are four different relations about displacement, velocity, and acceleration for the motion of a particle in general.
| (a) | \(v_{avg}=\frac{1}{2} [ v(t_{1})+v(t_{2}) ]\) |
| (b) | \(v_{avg}=\frac{r(t_{2})-r(t_{1})}{t_{2}-t_{1}}\) |
| (c) | \(r=\frac{1}{2}[ v(t_{2})-v(t_{1}) ](t_2-t_1)\) |
| (d) | \(a_{avg}=\frac{v(t_{2})-v(t_{1})}{t_{2}-t_{1}}\) |
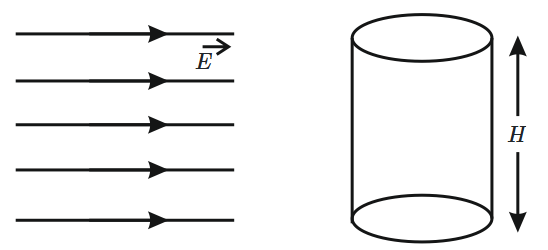
A cylinder of height \(20~\text{m}\) is completely filled with water. The velocity of efflux of water (in \(\text{ms}^{-1}\)) through a small hole on the side wall of the cylinder near its bottom, is:
1. \(10~\text{m/s}\)
2. \(20~\text{m/s}\)
3. \(25.5~\text{m/s}\)
4. \(5~\text{m/s}\)
| 1. | \(\dfrac{100}{473}P\) | 2. | \(\dfrac{273}{473}P\) |
| 3. | \(\dfrac{273}{100}P\) | 4. | \(\dfrac{473}{273}P\) |
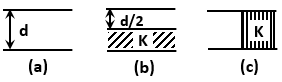
| 1. | \(mg=\dfrac{{mv}^{2}}{r}\) |
| 2. | \(mg\) is greater than \(\dfrac{{mv}^{2}}{r}\) |
| 3. | \(mg\) is not greater than \(\dfrac{{mv}^{2}}{r}\) |
| 4. | \(mg\) is not less than \(\dfrac{{mv}^{2}}{r}\) |
| 1. | \(1.5\) | 2. | \(1.33\) |
| 3. | \(3\) | 4. | \(2\) |
| 1. | \(\mathit{\pi}\) | 2. | \({2}\mathit{\pi}\) |
| 3. | \({4}\mathit{\pi}\) | 4. | \({6}\mathit{\pi}\) |
| 1. | zero | 2. | \(0.02\) |
| 3. | \(0.03\) | 4. | \(0.036\) |
| 1. | there is a minimum frequency of light, below which no photoelectric effect takes place. |
| 2. | by reverse potential, the photoelectric effect can be stopped. |
| 3. | intensity can alter the energy of photoelectron |
| 4. | even at low intensity, with frequency more than threshold, photoelectric emission takes place |