The arrangement of nuclei in a normal embryo sac in the dicot plant is:
1. 2 + 4 + 2
2. 3 + 2 + 3
3. 2 + 3 + 3
4. 3 + 3 + 2
DNA polymerase:
| A. | is the main enzyme for RNA synthesis |
| B. | is DNA dependent DNA polymerizing enzyme |
| C. | is a highly efficient enzyme |
| D. | catalyzes a reaction with a high degree of accuracy |
2. A, B, C and D
3. B, C and D
4. A, B and C
In 1900, Mendel’s work was rediscovered by:
1. Korana, Nirenberg and Mathei
2. De Vries, Correns and von Tschermak
3. Avery, McCleod and McCarty
4. Watson, Crick and Wilkins
The technology of biogas production was developed in India mainly due to the efforts of:
1. IARI
2. KVIC
3. Both 1 and 2
4. IAIR
Which of the following criteria should be fulfilled by a molecule to act as a genetic material?
| (i) | It should not be able to generate its replica |
| (ii) | It should chemically and structurally be stable |
| (iii) | It should not allow slow mutation |
| (iv) | It should be able to express itself in the form of Mendelian Characters |
| 1. | (i) and (ii) |
| 2. | (ii) and (iii) |
| 3. | (iii) and (iv) |
| 4. | (ii) and (iv) |
Which of the following is not true about Big Bang theory?
| 1. | A singular huge explosion |
| 2. | Universe expanded and the temperature came down |
| 3. | Hydrogen and Helium formed along with this expansion |
| 4. | The gases condensed under gravitation and formed the galaxies of the present-day universe. |
An inverted pyramid of biomass can be found in which ecosystem?
1. Forest
2. Marine
3. Grassland
4. Tundra
Which of the following statement is incorrect?
| 1. | Cyanobacteria often form blooms in polluted water bodies |
| 2. | Body of slime moulds moves along decaying twigs and leaves engulfing inorganic material |
| 3. | RNA of the viroid is low molecular weight |
| 4. | Lichens do not grow in polluted areas |
Which of the following roots are respiratory roots?
1. Epiphytic Roots
2. Stilt Roots
3. Prop Roots
4. Pneumatophores
Which of the following is true about chromatophores?
1. It’s a plasma membrane infolding
2. site of photosynthesis
3. contain pigments
4. All of the above
Mark the incorrect option with respect to abscisic acid:
| 1. | Increases the tolerance of plants to various kinds of stresses |
| 2. | It induces flowering in some long day plants |
| 3. | It plays an important role in seed development and maturation |
| 4. | In most situations, it acts as an antagonist to gibberellins |
A place used for storing, preservation, and exhibition of both plants and animals is called:
1. Museum
2. Botanical garden
3. Herbarium
4. Flora
Match Column-I with Column- II.
| Column I | Column II | ||
| (a) | Nitrococcus | (i) | Denitrification |
| (b) | Rhizobium | (ii) | Conversion of ammonia to nitrite |
| (c) | Thiobacillus | (iii) | Conversion of nitrite to nitrate |
| (d) | Nitrobactèr | (iv) | Conversion of atmospheric nitrogen to ammonia |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iv) | (iii) | (ii) | (i) |
| 3. | (ii) | (iv) | (i) | (iii) |
| 4. | (i) | (ii) | (iii) | (iv) |
Which of the following timeline is correct for conventional plant breeding?
| 1. | Practiced for thousands of years, since the beginning of human civilization |
| 2. | The evidence of plant breeding dates back 9000-11000 years ago |
| 3. | The present-day crops are the result of domestication in ancient times |
| 4. | All of these |
A cell at telophase stage brought from the field is observed by a student. He tells his teacher that this cell is not like other cells at telophase stage. There is no formation of a cell plate and thus the cell is containing more chromosomes as compared to other dividing cells. This would result in:
1. polyploidy
2. somaclonal variation
3. polyteny
4. aneuploidy
Basmati rice is distinct for its unique aroma and flavour. How many documented varieties of Basmati are grown in India?
| 1. | 27 | 2. | 270 |
| 3. | 13 | 4. | 230 |
Match the following:
| (a) Eyes of | (i) Ginger |
| (b) Rhizome of | (ii) Potato |
| (c) Bulbil of | (iii) Agave |
| (d) Leaf buds of | (iv) Bryophyllum |
| (e) Offset of | (v) Water hyacinth |
| (a) | (b) | (c) | (d) | (e) | |
| 1. | (ii) | (iii) | (i) | (iv) | (v) |
| 2. | (iv) | (i) | (iii) | (ii) | (v) |
| 3. | (ii) | (i) | (iii) | (iv) | (v) |
| 4. | (v) | (i) | (iii) | (iv) | (ii) |
Which is the most common type of embryo sac in angiosperms?
1. Tetrasporic with one mitotic stage of divisions
2. Monosporic with three sequential mitotic divisions
3. Monosporic with two sequential mitotic divisions
4. Bisporic with two sequential mitotic divisions
Which of the following helps in termination of transcription?
1. Sigma factor
2. Translocase
3. Ubiquitin
4. Rho factor
The most important cause driving animals and plants to extinction in the Evil Quartet is:
1. Habitat loss and Fragmentation
2. Over-Exploitation
3. Alien Species Invasions
4. Co-extinctions
DNA precipitation out of a mixture of biomolecules can be achieved by treatment with:
1. Chilled chloroform
2. isopropanal
3. Chilled ethanol
4. Methanol at room temperature
A species that was introduced for ornamentation but has become a troublesome weed in India is:
1. Parthenium hysterophorus
2. Eichhornia crassipes
3. Prosopis juliflora
4. Trapa Spinosa
How do herbivores and other animals obtain Phosphorous?
1. From soil in the dissolved form
2. From plants
3. From drinking water
4. Synthesized in the body.
If one wants to write a whole reaction of Krebs’ cycle, then how many water molecules are utilized in net calculation?
| 1. | Two | 2. | One |
| 3. | Three | 4. | Four |
Study the pathway given below. In which of the following options correct words for all the three blanks a,b and c are indicated?
|
(a) |
(b) |
(c) |
|
|
1. |
Carboxylation |
Decarboxylation |
Reduction |
|
2. |
Decarboxylation |
Reduction |
Regeneration |
|
3. |
Fixation |
Transamination |
Regeneration |
|
4. |
fixation |
Decarboxylation |
Regeneration |
A biologist studied the population of rats in a barn. He found that the average natality was 250, average mortality 240, immigration 20, and emigration 30. The net increase in population is:
| 1. | 10 | 2. | 15 |
| 3. | 5 | 4. | zero |
Companion cells are closely associated with:
| 1. | Sieve elements | 2. | Vessel elements |
| 3. | Trichomes | 4. | Guard cells |
Isogamous condition with non-flagellated gametes is found in:
| 1. | Spirogyra | 2. | Volvox |
| 3. | Fucus | 4. | Chlamydomonas |
What does Xylem transport from roots to the aerial parts of the plants?
1. Water, mineral salt
2. Some organic Nitrogen
3. Inorganic solutes, Hormones
4. All of these
Wine and beer are produced:
1. By distillation only
2. By fermentation and distillation
3. By fermentation but without distillation
4. Without fermentation and distillation
Mad cow disease in cattle is caused by an organism which has:
1. Inert crystalline
2. Abnormally folded protein
3. Free RNA without protein coat
4. Free DNA without protein coat
Match Column I with Column II
| Column I | Column II | ||
| (a) | 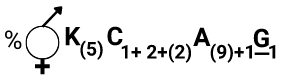 |
(i) | Brassicaceae |
| (b) |  |
(ii) | Liliaceae |
| (c) |  |
(iii) | Fabaceae |
| (d) |  |
(iv) | Solanaceae |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (iv) | (ii) | (i) | (iii) |
| 3. | (iii) | (iv) | (ii) | (i) |
| 4. | (i) | (ii) | (iii) | (iv) |
Which of the following statement is not associated with the phenomenon of flowering?
| 1. | Decision for flowering takes place much before actual flower appears. |
| 2. | During flowering several structural and hormonal changes takes place. |
| 3. | During flowering differentiation takes place. |
| 4. | Flowering is independent of environmental factors. |
Match the terms in Column I with their description in Column II and choose the correct option.
| Column I | Column II | ||
| (a) | Dominance | (i) | Many govern a single character |
| (b) | Codominance | (ii) | In a heterozygous organism only one allele expresses itself |
| (c) | Pleiotropy | (iii) | In a heterozygous organism both alleles express themselves fully |
| (d) | Polygenic inheritance | (iv) | A single gene influences many characters |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (iv) | (i) | (ii) | (iii) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (ii) | (i) | (iv) | (iii) |
In genetic code dictionary, codons used to code for all the 20 essential amino acids are:
| 1. | 20 | 2. | 60 |
| 3. | 61 | 4. | 64 |
The incorrect statement about "Montreal Protocol" is:
| 1. | An International Treaty |
| 2. | Was signed at Montreal (Canada) |
| 3. | To control the emission of Ozone depleting substances |
| 4. | Was effective in 1987 |
Which of the following is incorrect?
| 1. | Pili and Fimbriae play role in motility |
| 2. | Flagella, pili, and fimbriae are surface structures |
| 3. | Fimbriae help in attachment to surfaces |
| 4. | Both 1 and 3 |
Find the incorrect statement about Seed Dormancy.
| 1. | Can be caused by immature embryos |
| 2. | Can be overcome by natural means and various other man-made measures |
| 3. | ABA has no effects over seed Dormancy |
| 4. | GA can overcome seed Dormancy |
Select the correct statements:
| a. | Tapetum cells possess dense cytoplasm and generally have more than one nucleus |
| b. | Sporopollenin is absent in the areas called germ pore |
| c. | The generative cell is large, spindle-shaped with a irregularly shaped nuclei |
| d. | The spindle of first mitosis in pollen is asymmetric |
| 1. | a, b, c, & d |
| 2. | a, b, & c |
| 3. | a, c, & d |
| 4. | a, b, & d |
In the seeds of maize, the seed coat:
1. Is fused with the pericarp
2. Store aleurone grains
3. Is membranous and triploid
4. Has an outgrowth, called strophiole
Which fungi are edible?
1. Truffles
2. Morels
3. Agaricus
4 All of these
Gause’s principle of competitive exclusion states that:
| 1. | More abundant species will exclude the less abundant species through competition |
| 2. | Competition for the same resources excludes species having different food preferences |
| 3. | No two species can occupy the same niche indefinitely for the same limiting resources |
| 4. | Larger organisms exclude smaller ones through competition |
If is absent in the atmosphere of the earth then:
1. Temperature will decrease
2. Temperature will increase
3. Plants will flourish well
4. No effect
Diffusion is very important to plants since:
| 1. | The cells have a permeable cell wall |
| 2. | It is the only means for gaseous movement within the plant body. |
| 3. | Plants cannot transport material by active transport. |
| 4. | They are unable to move towards the source of the nutrients. |
Who gave the law of limiting factors and when?
1. Lieman-1906
2. Blackman-1905
3. Lieman-1905
4. Blackman-1906
When xylem and phloem within a vascular bundle are arranged in an alternate manner on different radii, such arrangement is called _______ and found mostly in __________.
| 1. | Radial, roots | 2. | Radial, shoots |
| 3. | Conjoint, roots | 4. | Conjoint, shoots |
After glycolysis, the fate of glucose in the mitochondrial matrix is:
1. oxidation
2. reduction
3. oxidative decarboxylation
4. hydrolysis.
Which of the following is not matched correctly?
| 1. | Artificial system of classification | Equal weightage to vegetative and sexual character |
| 2. | Natural system of classification | Based on natural affinities |
| 3. | Phylogenetic system | Common ancestor for unrelated taxa |
| 4. | Numerical taxonomy | Equal importance to all character |
Concanavalin A is:
| 1. | a pigment | 2. | an alkaloid |
| 3. | an essential oil | 4. | a lectin |
A stage in cell division is shown in the figure. Select the answer which gives correct identification of the stage with its characteristics:
| 1. | Late anaphase | Chromosomes move away from equatorial plate, Golgi complex not present |
| 2. | Cytokinesis | Cell plate formed, mitochondria distributed between two daughter cells |
| 3. | Telophase | Endoplasmic reticulum and nucleolus not reformed yet |
| 4. | Telophase | Nuclear envelop reforms, Golgi complex reforms |
Which one of the following statements is incorrect with respect to the transport of carbon dioxide in blood?
| 1. | About 20-25 percent of carbon dioxide is carried by haemoglobin in the form of carbaminohaemoglobin |
| 2. | 70 percent of is carried in a dissolved state through plasma |
| 3. | As the blood picks up carbon dioxide from tissue, accumulates inside RBCs of which some move out into the blood plasma, and in exchange chloride ions move from plasma into RBCs |
| 4. | The greatest percentage of carbon dioxide is transported in blood plasma as bicarbonate ions |
The embryo at the morula stage contains:
| 1. | 4 blastomeres | 2. | 30 blastomeres |
| 3. | 32 to 64 blastomeres | 4. | 8 to 16 blastomeres |
What will be the ratio of number of microtubules in peripheral and central parts of the axoneme in a eukaryotic flagellum?
| 1. | 9 : 2 | 2. | 9 : 0 |
| 3. | 9 : 1 | 4. | 27 : 0 |
Upon complete burning of the tissue, all the carbon compounds are oxidized to:
1. Gaseous form (NH3, CO2)
2. Gaseous form (CO2, water vapour)
3. Solid form (CO2, water vapour)
4. Liquid form (CH4, CO2)
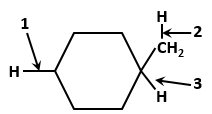
In the given diagram showing anatomical regions of the human stomach, A, B and C respectively represent:
| (i) | Dorsally by vertebral column |
| (ii) | Ventrally by sternum |
| (iii) | Laterally by ribs |
| (iv) | Lower side by diaphragm |
| 1. | (i) and (iii) only |
| 2. | (i), (ii) and (iii) only |
| 3. | (ii), (iii) and (iv) only |
| 4. | (i), (ii), (iii) and (iv) |
| Column-I | Column-II | ||
| (a) | Heart attack | (I) | Heart stops beating |
| (b) | Cardiac arrest | (II) | Not enough oxygen is reaching heart muscles leading to chest pain |
| (c) | Heart failure | (III) | Heart is not pumping blood effectively to meet the body's demand |
| (d) | Angina | (IV) | Heart muscles suddenly damaged by the inadequate blood supply |
| (a) | (b) | (c) | (d) | |
| 1. | (IV) | (I) | (II) | (III) |
| 2. | (IV) | (I) | (III) | (II) |
| 3. | (I) | (IV) | (III) | (II) |
| 4. | (I) | (III) | (II) | (IV) |
Match the columns:-
| Column-l | Column-ll | ||
| (a) | Rosie | (i) | α - 1 antitrypsin |
| (b) | ELISA | (ii) | Protein enriched milk |
| (c) | ROP | (iii) | Test to detect antigen or antibody |
| (d) | Emphysema | (iv) | Codes for protein involved in plasmid replication |
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (i) | (iii) | (iv) | (ii) |
| 3. | (ii) | (iii) | (i) | (iv) |
| 4. | (iv) | (iii) | (ii) | (i) |
Inbreeding depression is -
| 1. | Reduced motility and immunity due to close inbreeding |
| 2. | Decreased productivity due to mating of superior male and inferior female |
| 3. | Decrease in body mass of progeny due to continued close inbreeding |
| 4. | Reduced fertility and productivity due to continued close inbreeding |
Visceral nervous system:
| 1. | is a part of PNS |
| 2. | is made up of Nerves, Fibres, Ganglia and Plexuses |
| 3. | transmits information from CNS to the viscera and viscera to CNS |
| 4. | All of the above |
A man has tumor in thyroid gland which adversely affects the body physiology. This is because of:
| 1. | hyperthyroidism | 2. | hypothyroidism |
| 3. | Both 1 and 2 | 4. | None of the above |
Stinging capsules (nematocysts) are found in:
1. Wasp and Physalia
2. Adamsia and Scorpion
3. Sea pen and Sea fan
4. Meandrina and Tapeworm
Insertional inactivation of the lac Z gene forms-
| 1. | Blue recombinant colonies |
| 2. | Colourless recombinant colonies |
| 3. | Fluorescent green colonies |
| 4. | There is no relation between the lac Z gene and colour of the colony. |
Which of the following pairs of organs do not represent analogous organs?
1. Eye of an octopus and eye of a mammal
2. Flippers of penguin and flippers of dolphin
3. Wings of bat and forelimbs of humans
4. Wings of birds and wings of butterfly
Which of the following is correctly matched?
1. Mendelism - Genetics of haploid
2. Polydactyly - X-linked dominant
3. Blood group - More than two forms of a gene
4. Lethal gene - 1 : 2 : 1
Read the given statement:
For normal fertility in a healthy human male, at least __a__ % of ejaculated sperms must have normal shape and size and at least __b__ % of them must show vigorous motility. Choose the option that fills the respective blanks correctly 'a' & 'b':
1. 60 & 40
2. 40 & 60
3. 80 & 20
4. 70 & 30
| Energy Production, Waste treatment, Bioremediation, Diagnostics |
Carnivorous animals - lions and leopards, occupy the same niche but lions predate mostly larger animals and leopards take smaller ones. This mechanism of competition is referred to as -
1. Character displacement
2. Altruism
3. Resource partitioning
4. Competitive exclusion
The principal nitrogenous excretory compound in humans is synthesized:
1. in kidneys but eliminated mostly through liver
2. in kidneys as well as eliminated by kidneys
3. in liver and also eliminated by the same through bile
4. in the liver, but eliminated mostly through kidneys
If for some reason our goblet cells are non-functional, this will adversely affect:
1. production of somatostatin
2. secretion of sebum from the sebaceous glands
3. maturation of sperms
4. smooth movement of food down the intestine
Given diagram shows bone of the left human hind limb as seen from front. It has certain mistakes in labeling. Two of the wrongly labelled bones are:
1. tibia and tarsals
2. femur and fibula
3. fibula and phalanges
4. tarsals and femur
The structures that help some bacteria to attach to rocks and / or host tissues are:
1. Fimbriae
2. Mesosomes
3. Holdfast
4. Rhizoids
Plasmid pBR322 has a PstI restriction enzyme site within gene ampR that confers ampicillin resistance. If this enzyme is used for inserting a gene for -galactoside production and the recombinant plasmid is inserted in an E.coli strain, then:
| 1. | it will lead to the lysis of host cells. |
| 2. | it will be able to produce a novel protein with dual abilities. |
| 3. | it will not be able to confer ampicillin resistance to the host cell. |
| 4. | the transformed cells will have the ability to resist ampicillin as well as produce -galactoside. |
Vitreous humor is found in vitreous chamber:
1. Between lens and retina
2. Between cornea and lens
3. Between sclera and lens
4. Between choroid and lens
Which of the following hormone regulates sleep-wake cycle ?
1. Melatonin
2. Thyroxine
3. Vasopressin
4. MSH
Choose the correct pair:
| 1. | Physical barriers | HCl in stomach, saliva in mouth |
| 2. | Cellular barriers | PMNL, NK cells |
| 3. | Physiological barriers | Mucosa of gut, urogenital tract |
| 4. | Cytokine barriers | Exotoxins |
In a specie, the weight of newborn ranges from 2 to 5 kg. 97% of the newborn with an average weight between 3 to 3.3 kg survive whereas 99% of the infants born with weights from 2 to 2.5 kg or 4.5 kg to 5 kg die. Which type of selection process is taking place?
| 1. | Cyclical selection | 2. | Directional selection |
| 3. | Stabilizing selection | 4. | Disruptive selection |
Match the Column-I and Column-II
| Column-I | Column-II | ||
| (a) | P-waves | (i) | Depolarisation of ventricles |
| (b) | QRS complex | (ii) | Repolarization of ventricles |
| (c) | T-wave | (iii) | Coronary ischemia |
| (d) | Reduction in the size of T-wave | (iv) | Depolarisation of atria |
| (v) | Repolarization of atria |
||
Select the correct option
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (iii) | (v) | (iv) |
| 2. | (iv) | (i) | (ii) | (iii) |
| 3. | (iv) | (i) | (ii) | (v) |
| 4. | (ii) | (i) | (v) | (iii) |
Select the incorrectly matched pair from the following:
| 1. | Chondrocytes | Smooth muscle cells |
| 2. | Neurons | Nerve cells |
| 3. | Fibroblast | Areolar tissue |
| 4. | Osteocytes | Bone cells |
Match the following columns and select the correct option:
| Column-I | Column-II | ||
| (a) | Pneumotaxic Centre | (i) | Alveoli |
| (b) | O2 Dissociation curve | (ii) | Pons region of brain |
| (c) | Carbonic Anhydrase | (iii) | Haemoglobin |
| (d) | Primary site of exchange of gases | (iv) | R.B.C. |
Options:
| (a) | (b) | (c) | (d) | |
| 1. | (i) | (iii) | (ii) | (iv) |
| 2. | (ii) | (iii) | (iv) | (i) |
| 3. | (iii) | (ii) | (iv) | (i) |
| 4. | (iv) | (i) | (iii) | (ii) |
Match Column I with Column II.
| Column I | Column II | ||
| (a) | Vaults | (i) | Entry of sperm through the cervix is blocked |
| (b) | IUDs | (ii) | Removal of Vas deferens |
| (c) | Vasectomy | (iii) | Phagocytosis of sperms within the uterus |
| (d) | Tubectomy | (iv) | Removal of the fallopian tube |
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (iv) | (iii) | (i) |
| 2. | (iii) | (i) | (iv) | (ii) |
| 3. | (iv) | (ii) | (i) | (iii) |
| 4. | (i) | (iii) | (ii) | (iv) |
Which one of the following organisms bears hollow and pneumatic long bones?
| 1. | Macropus | 2. | Ornithorhynchus |
| 3. | Neophron | 4. | Hemidactylus |

| A | B | C | D | |
| 1. | Potential energy | Transition state | Activation energy with enzyme |
Activation energy without enzyme |
| 2. | Transition state | Potential energy | Activation energy without enzyme |
Activation energy with enzyme |
| 3. | Potential energy | Transition state | Activation energy with enzyme |
Activation energy without enzyme |
| 4. | Activation energy with enzyme |
Transition state | Activation energy without enzyme |
Potential energy |
The figure given below depicts a diagrammatic sectional view of the female reproductive system of humans. Which one set of three parts out of A-F have been correctly identified?
| 1. | C | lnfundibulum | D | Fimbriae | E | Cervix |
| 2. | D | Oviducal funnel | E | Uterus | F | Cervix |
| 3. | A | Perimetrium | B | Myometrium | C | Fallopian tube |
| 4. | B | Endometrium | C | Infundibulum | D | Fimbriae |
In the process of respiration, how many times exchange of gases by the process of diffusion takes place in respiration?
1. 4
2. 3
3. 2
4. 1
The number of RBC in a healthy individual are:
1. 5 million to 5.5 million RBCs
2. 5 billion to 5.5 billion RBCs
3. 1 million to 1.5 million RBCs
4. 1 billion to 1.5 billion RBCs
Consider the following statements:
| I. | Many bony fishes, aquatic amphibians and aquatic insects are ammonotelic |
| II. | Mammals, many terrestrial amphibians and cartilaginous fishes are ureotelic |
| III. | Reptiles, birds, land snails and insects are uricotelic |
Which of the above statements are true ?
| 1. | I and II only |
| 2. | I and III only |
| 3. | II and III only |
| 4. | I,II, and III |
Which event surely leads to pregnancy?
| 1. | Unprotected coitus | 2. | Fertilization |
| 3. | Implantation | 4. | Gestation |
Read the following paragraph having three blanks A, B and C.
Subphyla Urochordata and Cephalochordata are often referred to as protochordates and are A , marine. In B , notochord is present only in larval tail, while in C it extends from head to tail region and is persistent throughout their life.
The correct option for the three blanks is:
| A | B | C | |
| 1. | Generally | Urochordata | Cephalochordata |
| 2. | Exclusively | Cephalochordata | Urochordata |
| 3. | Exclusively | Herdmania | Branchiostoma |
| 4. | Generally | Ascidia | Branchiostoma |
Which of the following is wrongly matched in the given table?
| Microbe | Product | Application | |
| 1. | Trichoderma | Cyclosporin A | Immunosuppressive drug |
| 2. | Monascus purpureus | Statins | Lowering of blood cholesterol |
| 3. | Streptococcus | Streptokinase | Removal of clot from blood vessel |
| 4. | Clostridium butylicum | Lipase | Removal of oil stains |
Which of the following step is incorrect with respect to DNA fingerprinting?
| 1. | Separation of DNA fragments by electrophoresis |
| 2. | Isolation of DNA |
| 3. | Hybridisation using labeled VNTR probe |
| 4. | Detection of hybridized DNA fragments by ultracentrifugation |
Mammals evolved from:
1. Therapsids
2. Sauropsids
3. Thecodonts
4. Tuataras
A diagram showing axon terminal and synapse is given. Identify correctly A-D:
| 1. | A | Receptor | C | Synaptic vesicles |
| 2. | B | Synaptic connection | D | K+ |
| 3. | A | Neurotransmitter | B | Synaptic cleft |
| 4. | C | Neurotransmitter | D | Ca2+ |
Match the source gland with its respective hormone as well as the function:
| Source gland | Hormone | Function | |
| 1. | Posterior pituitary | Vasopressin | Stimulates reabsorption of water in the distal tubules in the nephron |
| 2. | Corpus luteum | Prolactin | Supports pregnancy |
| 3. | Thyroid | Thyroxine | Regulates blood calcium level |
| 4. | Anterior pituitary | Oxytocin | Contraction of uterus muscles during childbirth |
A neural signal reaching the neuromuscular junction releases …………. which generates an action potential in the ………..
1. Acetylcholine, Sarcolemma
2. Inositol, Sarcolemma
3. GABA, Sarcoplasmic reticulum
4. GABA, Sarcoplasm
Every……….Keoladeo National Park…… in Rajasthan hosts thousands of migratory birds coming from …….
1. Summer, Udaypur, Himachal
2. Winter, Bharatpur, Siberia
3. Summer, Bharatpur, Madhya Pradesh
4. Winter, Jodhpur, Himachal
The figure below is the diagrammatic representation of the E.Coli vector pBR 322. Which one of the given options correctly identifies its certain component(s)?
| 1. | ori | original restriction enzyme |
| 2. | rop | reduced osmotic pressure |
| 3. | Hind III, EcoRI | selectable markers |
| 4. | ampR, tetR | antibiotic resistance genes |
Which one of the following statements is correct with respect to AIDS?
| 1. | The HIV can be transmitted through eating food together with an infected person |
| 2. | Drug addicts are least susceptible to HIV infection |
| 3. | AIDS patients are being fully cured cent percent with proper care and nutrition |
| 4. | The causative HIV retrovirus enters helper T-lymphocytes thus reducing their numbers |
Which of the following statements are correct about IUDs?
| I. | Increase phagocytosis of sperms |
| II. | Release Cu ions that suppress sperm motility and fertilising capacity of sperms |
| III. | Make the uterus unsuitable for implantation |
| IV. | Make the cervix hostile to sperms |
| V. | Prevent semen from entering the female reproductive tract. |
| 1. | I, II, III, IV and V |
| 2. | I, II, III and IV |
| 3. | I and II |
| 4. | II, III and IV |
Among the following, the incorrect statement about colloids is:
| 1. | The range of diameters of colloidal particles is between 1 and 1000 nm |
| 2. | They are larger than small molecules and have a high molar mass |
| 3. | They can scatter the light |
| 4. | The osmotic pressure of a colloidal solution is of a higher order than the true solution at the same concentration. |
Which of the following compounds will show retention in the configuration on nucleophilic substitution by OH– ion?
1. \(\mathrm{CH}_3-\mathrm{CH}\left(\mathrm{C}_2 \mathrm{H}_5\right)-\mathrm{CH}_2 \mathrm{Br}\)
2. \(\mathrm{CH}_3-\mathrm{CH}\left(\mathrm{C}_6 \mathrm{H}_5\right)-\mathrm{Br}\)
3. \(\begin{array}{lll}
\mathrm{CH}_3 -\mathrm{CH}\left(\mathrm{CH}_3\right) -\mathrm{Br}
\end{array}\)
4. \(\mathrm{CH}_3-\mathrm{CH}(\mathrm{Br})-\mathrm{C}_6 \mathrm{H}_{13}\)
Simplified absorption spectra of three complexes (i), (ii) and (iii) of Mn+ ion are provided below; their max values are marked as A, B and C respectively. The correct match between the complexes and their max values is:
 |
(i) | [M(NCS)6](-6+n) |
| (ii) | [MF6](-6+n) | |
| (iii) | [M(NH3)6]n+ |
Options:
| A | B | C | |
| 1. | (ii) | (i) | (iii) |
| 2. | (iii) | (i) | (ii) |
| 3. | (ii) | (iii) | (i) |
| 4. | (i) | (ii) | (iii) |
Match the following drugs with their therapeutic actions:
| Drugs | Therapeutic actions | ||
| (i) | Ranitidine | (a) | Antidepressant |
| (ii) | Nardil (Phenelzine) | (b) | Antibiotic |
| (iii) | Chloramphenicol | (c) | Antihistamine |
| (iv) | Dimetapp (Brompheniramine) | (d) | Antacid |
| (e) | Analgesic | ||
Options:
| (i) | (ii) | (iii) | (iv) | |
| 1. | (e) | (a) | (c) | (d) |
| 2. | (d) | (c) | (a) | (e) |
| 3. | (d) | (a) | (b) | (c) |
| 4. | (a) | (c) | (b) | (e) |
An Ellingham diagram provides information about:
| 1. | The pressure dependence of the standard electrode potentials of reduction reactions involved in the extraction of metals. |
| 2. | The kinetics of the reduction process. |
| 3. | The temperature dependence of the standard Gibbs energies of formation of some metal oxides. |
| 4. | The conditions of pH and potential under which a species is thermodynamically stable. |
'A' and 'B' in the following reactions are:

| A | B | A | B | ||
| 1. |  |
 |
2. |  |
 |
| 3. |  |
 |
4. |  |
 |
Match Column I with Column II.
| Column I (Monomer Unit) |
Column II (Polymer) |
||
| (a) | Caprolactam | (i) | Natural rubber |
| (b) | 2-Chloro-1,3-butadiene | (ii) | Buna-N |
| (c) | Isoprene | (iii) | Nylon 6 |
| (d) | Acrylonitrile | (iv) | Neoprene |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (iv) | (iii) | (ii) | (i) |
| 2. | (ii) | (i) | (iv) | (iii) |
| 3. | (iii) | (iv) | (i) | (ii) |
| 4. | (i) | (ii) | (iii) | (iv) |
Among the following allotropic forms of sulphur, the allotropic form paramagnetic in nature is:
| 1. | -sulphur | 2. | -sulphur |
| 3. | S2-form | 4. | None of the above |
Which of the glycosidic linkage between galactose and glucose is present in lactose?
1. C-1 of galactose and C-4 of glucose
2. C-1 of glucose and C-6 of galactose
3. C-1 of glucose and C-4 of galactose
4. C-1 of galactose and C-6 of glucose
The correct match between Item-I (starting material) and Item-II (reagent) for the preparation of benzaldehyde is :
| Item-I | Item-II | ||
| (I) | Benzene | (P) | |
| (II) | Benzonitrile | (Q) | |
| (III) | Benzoyl Chloride | (R) | |
Options:
| (I) | (II) | (III) | |
| 1. | (Q) | (R) | (P) |
| 2. | (R) | (Q) | (P) |
| 3. | (R) | (P) | (Q) |
| 4. | (P) | (Q) | (R) |
Identify A and B in the chemical reaction:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The electrons identified by quantum numbers n and l can be placed in order of increasing energy as:
a. n = 4, = 1
b. n = 4, = 0
c. n = 3, = 2
d. n = 3, = 1
| 1. | (d) < (b) < (c) < (a) | 2. | (b) < (d) < (a) < (c) |
| 3. | (a) < (c) < (b) < (d) | 4. | (c) < (d) < (b) < (a) |
If 10−4 dm3 of water is introduced into a 1.0 dm3 flask at 300 K, then the total number of moles of water in the vapour phase (equilibrium is established) are-
(Given: Vapour pressure of H2O at 300 K is 3170 Pa; R = 0.0821 atm L K−1 mol−1; 1 Pa = 9.9 × 10-6 atm )
1. 1.26 x 10−3 mol
2. 5.56 x 10−3 mol
3. 1.53 x10−2 mol
4. 4346 x 10-2 mol
Percentages of free space in cubic close-packed structure and in body-centered packed structure are respectively-
1. 48% and 26%
2. 30% and 26%
3. 26% and 32%
4. 32% and 48%
The equilibrium constant at 298 K for a reaction A + B ⇋ C + D is 100.
If the initial concentration of all the four species were 1 M each, then equilibrium concentration of D (in mol L–1) will be :
| 1. | 0.182 | 2. | 0.818 |
| 3. | 1.818 | 4. | 1.182 |
The following do/does not cause water pollution:
1. Heavy metals such as Cd, Pb, Hg
2. Detergents
3. Polychlorobiphenyls
4. Freons
Determine the freezing point of a solution containing 8.1 g HBr in 100 g of water, assuming the acid to be 90% ionised:
(kf for water = 1.86 K mol-1)
1. 0.85 C
2. -3.53 C
3. 0 C
4. -0.35 C
The oxidation number of carbon in carbon suboxide is:
1.
2.
3. +4
4.
In which of the following arrangements the order is not according to the property indicated against it?
| 1. | Li < Na < K < Rb : Increasing metallic radius |
| 2. | I < Br < F < Cl: Increasing electron gain enthalpy (with negative sign) |
| 3. | B < C < N < O: Increasing first ionisation enthalpy |
| 4. | Al3+ < Mg2+ < Na+ < F- : Increasing ionic size |
How many moles of magnesium phosphate, Mg₃(PO₄)₂, contain 0.25 moles of oxygen atoms?
| 1. | 0.02 | 2. | 3.125 × 10–2 |
| 3. | 1.25 × 10–2 | 4. | 2.5 × 10–2 |
Beryllium and aluminum exhibit many properties which are similar. But, the two elements differ in:
1. Exhibiting maximum covalency in compounds.
2. Forming polymeric hydrides.
3. Forming covalent halides.
4. Exhibiting amphoteric nature in their oxides.
The half-life of a radioisotope is four hours. If the initial mass of the isotope was 200 g, then the mass remaining undecayed after 24 hours is:
1. 1.042 g
2. 2.084 g
3. 3.125 g
4. 4.167 g
The structure of the compound that gives a tribromo derivative on treatment with bromine water is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The number of EDTA (ethylenediaminetetraacetic acid) molecules that are required to make an octahedral complex with a Ca2+ ion is/are:
1. Six (6)
2. Three (3)
3. One (1)
4. Two (2)
Geometrical isomerism is not shown by:
1. 1,1-Dichloro-1-pentene
2. 1,2-Dichloro-1-pentene
3. 1,3-Dichloro-2-pentene
4. 1,4-Dichloro-2-pentene
Ce3+ , La3+ , Pm3+ and Yb3+ have ionic radii in the increasing order as:
1. La3+ < Ce3+ < Pm3+ < Yb3+
2. Yb3+ < Pm3+ < Ce3+ < La3+
3. La3+ = Ce3+ < Pm3+ < Yb3+
4. Yb3+ < Pm3+ < La3+ < Ce3+
pH of 0.005M calcium acetate (pka of CH3COOH = 4.74) is:
| 1. | 7.04 | 2. | 9.37 |
| 3. | 9.26 | 4. | 8.37 |
On vigorous oxidation by potassium permanganate solution, (CH3)2 C = CHCH2CHO gives:
| 1. | (CH3)2CO and OHCCH2CHO |
| 2. | 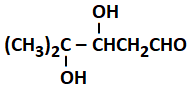 |
| 3. | (CH3)2CO and OHCH2CHO |
| 4. | (CH3)2CO and CH2(COOH)2 |
\(\mathrm{Zn}(s)+\mathrm{Cu}^{2+}(0.1 \mathrm{M}) \rightarrow \mathrm{Zn}^{2+}(1 \mathrm{M})+\mathrm{Cu}(s)\)
For the redox reaction taking place in a cell , \(E^o_{cell}\) is 1.10 volt . Ecell for the cell will be:
\(\left(2.303 \frac{R T}{F}=0.0591\right) \)
| 1. | 2.14 V | 2. | 1.80 V |
| 3. | 1.07 V | 4. | 0.82 V |
The substance used in Holmes signals of the ship is a mixture of :
1. CaC2 + Ca3P2
2. Ca3(PO4)2 + Pb3O4
3. H3PO4 + CaCl2
4. NH3 + HOCl
| 1. | Hydrogen never acts as cation in ionic salts |
| 2. | Hydronium ion, H3O+ exists freely in solution |
| 3. | Dihydrogen does not act as a reducing agent |
| 4. | Hydrogen has three isotopes of which protium is the most common |
Which of the following structure is similar to graphite ?
1. B
2. B4C
3. B2H6
4. BN
| Statement I: | The standard enthalpy change for the formation of one mole of a compound from its elements in their most stable states (reference states) is called standard molar enthalpy of formation. |
| Statement II: | For, CaO(s)+ CO2 (g) → CaCO3(s) ; ∆fH⊝ = -178.3 kJmol-1 , the ∆rH⊝ is equal to the ∆fH⊝ . |
1. Both statements I and II are true.
2. Statement I is true; and statement II is false.
3. Both statements I and II are false.
4. Statement I is false; statement II is true.
| 1. | 1 < 2 < 3 | 2. | 3 < 2 < 1 |
| 3. | 2 < 3 < 1 | 4. | 3 < 1 < 2 |
When 1 mole of is treated with excess of 3 moles of are obtained. The formula of the complex is:
1. [CrCl3(H2O)3].3H2O
2. [CrCl2(H2O)4]Cl.2H2O
3. [CrCl3(H2O)5]Cl2.H2O
4. [Cr(H2O)6]Cl3
In qualitative analysis when is passed through an aqueous solution of salt acidified with dil. HCl, a black precipitate is obtained. On boiling the precipitate with dil. , it forms a solution of blue colour. Addition of excess of aqueous solution of ammonia to this solution gives:
1. Deep blue precipitate of Cu(OH)2
2. Deep blue precipitate of [Cu(NH3)4]2+
3. Deep blue precipitate of Cu(NO3)2
4. Deep blue solution of Cu(OH)2 . Cu(NO3)2
Acetaldehyde reacts with semicarbazide to yield the following as a product:
| 1. | \( {CH_3CH=NHNH_2} ~\) |
| 2. | \({CH_3CH=NCONHNH_2}~~~\) |
| 3. | \({CH_3CH=NNH-CO-NH_2}~~~~~\) |
| 4. |  |
The major product of the following reaction is :

| 1. |  |
2. |  |
| 3. |  |
4. |  |
Among the following, the molecule expected to be stabilized by anion formation is : C2, O2, NO, F2
1. NO
2. O2
3. C2
4. F2
The decreasing order of electrical conductivity of the following aqueous solutions is :
0.1 M Formic acid (A),
0.1 M Acetic acid (B),
0.1 M Benzoic acid (C)
1. A > B > C
2. A > C > B
3. C > B > A
4. C > A > B
The mass of ammonia in grams produced when 2.8 kg of dinitrogen quantitatively reacts
with 1 kg of dihydrogen is:
| 1. | 3600 g | 2. | 3000 g |
| 3. | 3400 g | 4. | 4000 g |
For the reaction , the correct statement is:
| 1. | |
| 2. | |
| 3. | |
| 4. |
If 250 cm3 of an aqueous solution containing 0.73 g of a protein A is isotonic with one litre of another aqueous solution containing 1.66 g of a protein B at 298 K, the ratio of the molecular masses of A and B is:
1. 1.54
2. 1.77
3. 1.65
4. 1.60
At 1990 K and 1 atm pressure, there are an equal number of Cl2 molecules and Cl atoms in the reaction mixture.
The value of KP for the reaction under the above conditions is x × 10–1. The value of x is:
1. 4
2. 8
3. 5
4. 10
Which option correctly describes the shape and \(\text{I–I–I}\) bond angle of the \({I}_3^-\) ion, respectively?
1. Distorted trigonal planar; 135° and 90°
2. T-shaped; 180° and 90
3. Trigonal planar; 120°
4. Linear; 180°
For a chemical reaction A + B C + D (rH0 = 80 kJ mol–1 ) the entropy change rS0 depends on the temperature T (in K) as rS0 = 2T (JK–1 mol–1 ). The minimum temperature at which it will become spontaneous is:
| 1. | 200 K | 2. | 250 K |
| 3. | 300 K | 4. | 350 K |
29.5 mg of an organic compound containing nitrogen was digested according to Kjeldahl's method and the evolved ammonia were absorbed in 20 mL of 0.1 M HCl solution. The excess of the acid required 15 mL of 0.1 M NaOH solution for complete neutralization. The percentage of nitrogen in the compound is:
| 1. | 29.5 | 2. | 23.7 |
| 3. | 47.4 | 4. | 59.0 |
Which of the following compounds is non-aromatic?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The strongest ortho/para directing group is:
1. −NH2
2. −CH3
3. −Cl
4. −C2H5
A bob of mass \(m\) attached to an inextensible string of length \(l\) is suspended from a vertical support. The bob rotates in a horizontal circle with an angular speed \(\omega\) rad/s about the vertical. About the point of suspension:
| 1. | angular momentum changes in magnitude but not in direction. |
| 2. | angular momentum changes in direction but not in magnitude. |
| 3. | angular momentum changes in both direction and magnitude. |
| 4. | angular momentum is conserved. |
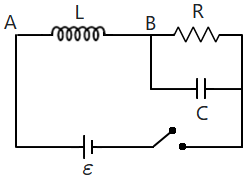

| 1. | increases with time | 2. | decreases with time |
| 3. | remains constant | 4. | no EMF is induced |
| 1. | lost its elastic property completely and it will not contract. |
| 2. | will contract to its original length. |
| 3. | will contract to its length at elastic limit. |
| 4. | will contract but the final length will be greater than the original length. |
| 1. | Coefficient of cubical expansion of liquid must be greater than that of bulb material |
| 2. | Coefficient of cubical expansion of liquid may be equal to that of bulb material |
| 3. | Coefficient of cubical expansion of liquid must be less than that of bulb material |
| 4. | None of the above |
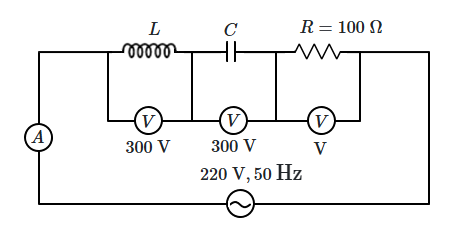
| 1. | \(4.0~\text{N}\) | 2. | \(12.5~\text{N}\) |
| 3. | \(0.5~\text{N}\) | 4. | \(6.25~\text{N}\) |
| 1. | \(x^{2}\) | 2. | \(x \) |
| 3. | \(\dfrac{1}{x}\) | 4. | \(\dfrac{1}{{x}^{2}}\) |
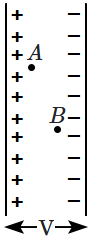
| 1. | \(F_A>F_B\) | 2. | \(F_A= F_B\) |
| 3. | \(F_A<F_B\) | 4. | nothing can be predicted |
| 1. | \(m\) | 2. | \(\frac{1}{m}\) |
| 3. | \(m+1\) | 4. | \(\frac{1}{{m}{+}{1}}\) |
| 1. | any angle between \(0\) and \(\pm{\pi}{/}{2}\) |
| 2. | \(\mathit{\pi}{/}{2}\) |
| 3. | \(\mathit{\pi}\) |
| 4. | any angle between \(0\) and \(\mathit{\pi}\) |
| 1. | \(I\) | 2. | \(3I\) |
| 3. | \(16I\) | 4. | \(25I\) |
| 1. | increase the maximum kinetic energy of the photoelectrons, as well as photoelectric current by a factor of two. |
| 2. | increase the maximum kinetic energy of the photoelectrons and would increase the photoelectric current by a factor of two. |
| 3. | increase the maximum kinetic energy of the photoelectrons by a factor of two and will have no effect on the magnitude of the photoelectric current produced. |
| 4. | not produce any effect on the kinetic energy of the emitted electrons but will increase the photoelectric current by a factor of two. |

| 1. | \(2B\) | 2. | \(B\) |
| 3. | \(B /2\) | 4. | \(3B\) |
| 1. | three \({\beta}^{+}\text-\)particles |
| 2. | one \(\alpha\text-\)particle and two \(\beta^{+}\text-\)particles |
| 3. | one \(\alpha\text-\)particle and two \(\beta^{-}\text-\)particles |
| 4. | two \(\alpha\text{-}\)particles and one \(\beta^{-}\text-\)particle |
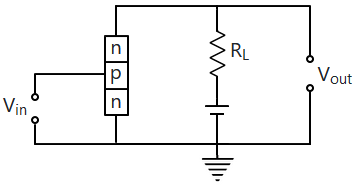
| 1. | a common-base amplifier circuit. |
| 2. | a common-emitter amplifier circuit. |
| 3. | a common-collector amplifier circuit. |
| 4. | none of the above. |
| 1. | \(25~\text{m/s} \) | 2. | \(40~\text{m/s}\) |
| 3. | \(31.25~\text{m/s}\) | 4. | \(12.5~\text{m/s}\) |
| 1. | constant amplitude | 2. | decreasing amplitude |
| 3. | increasing amplitude | 4. | none of these |
| 1. | \(5~\text{cm}\) | 2. | \(10~\text{cm}\) |
| 3. | \(20~\text{cm}\) | 4. | none of these |
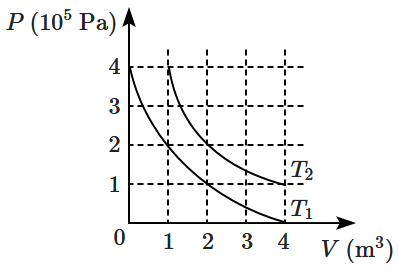
| 1. | \(2\sqrt{2}\) | 2. | \(\sqrt{2}\) |
| 3. | \(2\) | 4. | \(4\) |
| 1. | the charges are deflected in opposite directions |
| 2. | the charges continue to move in a straight line |
| 3. | the charges move in circular paths |
| 4. | the charges move in circular paths but in opposite directions |
| 1. | \(T= T_0\) | 2. | \(T>T_0\) |
| 3. | \(T<T_0\) | 4. | \(T=2T_0\) |
The variation of acceleration due to gravity \(g\) with distance \(d\) from centre of the earth is best represented by:
(\(R\) = Earth's radius)
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| Assertion (A): | For a simple microscope, the angular size of the object equals the angular size of the image. |
| Reason (R): | Magnification is achieved as the small object can be kept much closer to the eye than \(25~\text{cm}\) and hence it subtends a large angle. |
| 1. | (A) is True but (R) is False. |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 4. | (A) is False but (R) is True. |
River of width \(500~\text{m}\) is flowing at a speed of \(10~\text{m/s}\). A swimmer can swim at a speed of \(10~\text{m/s}\) in still water. If he starts swimming at an angle of \(120^\circ\) with the flow direction, then the distance he travels along the river while crossing the river is:
1. \(250~\text{m}\)
2. \(500\sqrt{3}~\text{m}\)
3. \(\dfrac{500}{\sqrt{3}}~\text{m}\)
4. \(500~\text{m}\)

| 1. | \( {i}_{0}T\) | 2. | \( \dfrac{{i}_{0}T}{2}\) |
| 3. | \( \dfrac{{i}_{0}T}{3}\) | 4. | \( \dfrac{{i}_{0}T}{\sqrt{2}}\) |
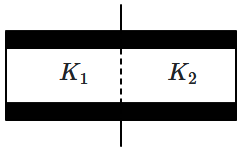
| 1. | \( \left({{K}_{1}{+}{K}_{2}}\right){C}_{0}\) | 2. | \(\frac{1}{2}\left({{K}_{1}{+}{K}_{2}}\right){C}_{0}\) |
| 3. | \( {2}\left({{K}_{1}{+}{K}_{2}}\right){C}_{0}\) | 4. | \( \frac{1}{2}\left({{K}_{1}{+}{K}_{2}{-}{2}}\right){C}_{0}\) |
| 1. | \(-4\) N | 2. | \(4\) N |
| 3. | \(8\) N | 4. | \(-8\) N |
When a slab of ice melts at \(273~\text{K}\) under atmospheric pressure, which of the following statements is correct?
| 1. | The ice-water system does positive work on the atmosphere. |
| 2. | The atmosphere does positive work on the ice-water system. |
| 3. | The internal energy of the ice-water system decreases. |
| 4. | None of the above. |
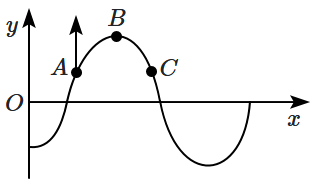
| 1. | The wave is travelling to the right. |
| 2. | The wave is travelling to the left. |
| 3. | At this instant of time, the velocity of \(C\) is also directed along the positive \(y\text-\)axis. |
| 4. | The phase difference between \(A\) and \(C\) is \(\mathit{\pi}.\) |

| 1. | \(5~\text{cm}\) |
| 2. | less than \(5~\text{cm}\) |
| 3. | greater than \(5~\text{cm}\) |
| 4. | cannot be found with this information |
| 1. | either input terminals is at \(0\) state. |
| 2. | either input terminals is at \(1\) state. |
| 3. | both input terminals are at \(0\) state. |
| 4. | both input terminals are at \(1\) state. |
| 1. | 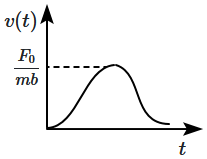 |
2. |  |
| 3. | 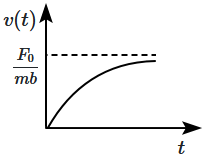 |
4. | 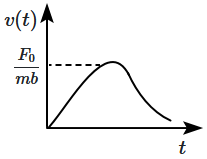 |

| 1. | \(2\) | 2. | \(12\) |
| 3. | \(6\) | 4. | \(3\) |
| 1. | \(\dfrac{{\pi}}{6}\) | 2. | \(\dfrac{{\pi}}{3}\) |
| 3. | \(\dfrac{{2}{\pi}}{3}\) | 4. | \(\dfrac{{5}{\pi}}{6}\) |
| 1. | greater than at \(Q\) |
| 2. | same as at \(Q\) |
| 3. | less than at \(Q\) |
| 4. | greater or less than at \(Q\) depending upon the strength of current |