Endosperm is formed during double fertilization by
| 1. | two polar nuclei and one male gamete |
| 2. | one polar nuclei and one male gamete |
| 3. | ovum and male gametes |
| 4. | two polar nuclei and two male gametes |
How many pairs of contrasting traits in pea plants were studied by Mendel in his experiments?
1. Five
2. Six
3. Eight
4. Seven
Which one of the following pairs of nitrogenous bases of nucleic acids is wrongly matched with the category mentioned against it?
1. Thymine, Uracil - Pyrimidines
2. Uracil, Cytosine - Pyrimidines
3. Guanine, Adenine - Purines
4. Adenine, Thymine - Purines
"The activated sludge is pumped into large tanks. Here, other kinds of bacteria, which grow anaerobically, digest the bacteria and the fungi in the sludge". In which tank, this process of sewage treatment is performed?
1. Secondary settling tank
2. Aeration tank
3. Primark settling tank
4. Anaerobic sludge digesters
Similar biological structures or sequences in different taxa are homologous if they:
| 1. | always perform the same function |
| 2. | result from convergent evolution |
| 3. | do not serve any known function |
| 4. | are derived from a common ancestor |
If a bamboo plant is growing in a far forest then what will be the trophic level of it?
1. First trophic level (T1)
2. Second trophic level (T2)
3. Third trophic level (T3)
4. Fourth trophic level (T4)
The floral formula has which of the following symbols to represent zygomorphic symmetry?
1. #
2. %
3. K
4. C
Mode of nutrition in euglenoids is:
1. Photosynthetic
2. Heterotrophic
3. Chemosynthetic
4. Both 1 & 2
Which one is/are correct?
| 1. | In prokaryotes, a single type of RNA polymerase can transcribe mRNA, tRNA, and rRNA. |
| 2. | In eukaryotes, RNA polymerase I transcribe rRNAs (28S, 18S, and 5.8S), whereas RNA polymerase III is responsible for the transcription of tRNA, 5srRNA, and snRNAs. |
| 3. | RNA pol II transcribes hnRNA in eukaryotes. |
| 4. | Ribosomal large subunit has p and A-sites. |
| 1. | A, C |
| 2. | A, B, C, D |
| 3. | B, C, D |
| 4. | A, B, D |
Frameshift mutation arises due to:
| 1. | Addition or deletion of a nucleotide base in DNA |
| 2. | Loss and gain of DNA segments respectively |
| 3. | Shifting of a part of chromosome |
| 4. | Rotating a section of the chromosome by 180° |
What is the fate of the male gametes discharged in the synergid?
| 1. | One fuses with the egg and the other fuses with central cell nuclei |
| 2. | One fuses with the egg and other(s) degenerate(s) in the synergid |
| 3. | All fuse with the egg |
| 4. | One fuses with the egg and the other(s) fuse(s) with the synergid nucleus |
Identify X:
1. Stem
2. Runner
3. Offset
4. Bud
| Column I | Column II | ||
| I. | Pusa Swarnim | 1. | Cauliflower |
| II. | Pusa Shubhra | 2. | Brassica |
| III. | Pusa Komal | 3. | Okra |
| IV. | Pusa Sawani | 4. | Cowpea |
| I | II | III | IV | |
| 1. | 1 | 2 | 3 | 4 |
| 2. | 2 | 1 | 4 | 3 |
| 3. | 2 | 1 | 3 | 4 |
| 4. | 1 | 2 | 4 | 3 |
Study the cycle shown below and select the option which gives the correct words for all the four blanks a, b, c, and d:
| (a) | (b) | (c) | (d) | |
| 1. | Denitrification | Ammonification | Plant biomass |
Animal biomass |
| 2. | Nitrification | Denitrification | Animal biomass |
Plant biomass |
| 3. | Denitrification | Nitrification | Plant biomass |
Animal biomass |
| 4. | Nitrification | Ammonification | Animal biomass |
Plant biomass |
The enzyme decarboxylase catalyses which of the following step:
| 1. | conversion of citric acid to cis aconitic acid |
| 2. | fumaric acid to malic acid |
| 3. | oxalosuccinic acid to α -ketoglutaric acid |
| 4. | malic acid to oxaloacetic acid |
How does pruning help in making the hedge dense?
| 1. | It induces the differentiation of new shoots from the rootstock |
| 2. | It frees axillary buds from apical dominance |
| 3. | The apical shoot grows faster after pruning |
| 4. | It releases wound hormones |
Sacred groves are especially useful in:
| 1. | Generating environmental awareness |
| 2. | Preventing soil erosion |
| 3. | Year-round flow of water in rivers |
| 4. | Conserving rare and threatened species |
Depletion of which gas in the atmosphere can lead to an increased incidence of skin cancers?
1. Methane
2. Nitrous oxide
3. Ozone
4. Ammonia
Pentamerous actinomorphic flowers, bicarpellary ovary with an oblique septum, and fruit of capsule or berry belong to which of the following family?
1. Liliaceae
2. Asteraceae
3. Brassicaceae
4. Solanaceae
The primitive prokaryotes responsible for the production of biogas from the dung of ruminant animals include:
1. Eubacteria
2. Halophiles
3. Thermoacidophiles
4. Methanogens
Identify the correct match:
1. Felis: Canidae
2. Datura: Solanaceae
3. Petunia: Leguminoceae
4. Dogs: Felidae
Which of the following nucleic acids is present in an organism having 70 S ribosomes only?
| 1. | Single stranded DNA with a protein coat |
| 2. | Double stranded circular naked DNA |
| 3. | Double stranded DNA enclosed in a nuclear membrane |
| 4. | Double stranded circular DNA with histone proteins |
One scientist cultured Cladophora in a suspension of Azotobacter and illuminated the culture by splitting light through a prism. He observed that bacteria accumulated mainly in the region of :
1. Violet and green light
2. Indigo and green light
3. Orange and yellow light
4. Blue and red light
What type of pollination takes place in Vallisneria?
| 1. | Pollination occurs in submerged conditions by water. |
| 2. | Flowers emerge above the surface of the water and pollination occurs by insects. |
| 3. | Flowers emerge above the water's surface and pollen is carried by the wind. |
| 4. | Male flowers are carried by water currents to female flowers at the surface of the water. |
Grass leaves curl inwards during very dry weather. Select the most appropriate reason from the following :
| 1. | Tyloses in vessels |
| 2. | closure of stomata |
| 3. | Flaccidity of bulliform cells |
| 4. | Shrinkage of air spaces in spongy mesophyll |
Match the following columns and select the correct option.
| Column I | Column II | ||
| (a) | Bt cotton | (i) | Gene therapy |
| (b) | Adenosine deaminase deficiency | (ii) | Cellular defence |
| (c) | RNAi | (iii) | Detection of HIV infection |
| (d) | PCR | (iv) | Bacillus thuringiensis |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (ii) | (i) | (iv) |
| 2. | (ii) | (iii) | (iv) | (i) |
| 3. | (i) | (ii) | (iii) | (iv) |
| 4. | (iv) | (i) | (ii) | (iii) |
Match the following techniques or instruments with their usage:
| Column I | Column II | ||
| (a) | Bioreactor | (i) | Separation of DNA fragments |
| (b) | Electrophoresis | (ii) | Production of large quantities of products |
| (c) | PCR | (iii) | Detection of a pathogen, based on antigen-antibody reaction |
| (d) | ELISA | (iv) | Amplification of nucleic acids |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (ii) | (iv) | (i) |
| 2. | (ii) | (i) | (iv) | (iii) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (ii) | (i) | (iii) | (iv) |
Which of the following statements is incorrect?
| 1. | Biomass decreases from the first to fourth trophic level |
| 2. | Energy content gradually increases from the first to fourth trophic level |
| 3. | Number of individuals decreases from the first trophic level to the fourth trophic level |
| 4. | Energy content gradually decreases from the first to the fourth trophic level |
Select the incorrect statement:
| 1. | The transport of molecules in phloem can be bidirectional. |
| 2. | The movement of minerals in the xylem is unidirectional. |
| 3. | Unloading of sucrose at the sink does not involve the utilization of ATP. |
| 4. | Elements most easily mobilized in plants from one region to another are phosphorus, sulfur, nitrogen, and potassium. |
Match the following columns and select the correct option:
| Column - I | Column -II | ||
| a. | Dragonflies | (i) | Biocontrol agents of several plant pathogens |
| b. | Bacillus thuringiensis | (ii) | Get rid of mosquitoes |
| c. | Glomus | (iii) | Narrow-spectrum insecticidal applications |
| d. | Baculoviruses | (iv) | Biocontrol agents of lepidopteran plant pests |
| (v) | Absorb phosphorus from soil | ||
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (v) | (iv) | (i) |
| 2. | (ii) | (i) | (iii) | (iv) |
| 3. | (ii) | (iii) | (iv) | (v) |
| 4. | (ii) | (iv) | (v) | (iii) |
Genotypic ratio of 1:2:1 is obtained in a cross between:
1. AA X AA
2. Aa X Aa
3. Aa X aa
4. aa X aa
Which of the following algae produces Carrageen?
1. Red algae
2. Blue-green algae
3. Green algae
4. Brown algae
In the exponential growth equation Nt =N0ert, e represents:
1. The base of natural logarithms
2. The base of geometric logarithms
3. The base of number logarithms
4. The base of exponential logarithms
Which is the "only enzyme" that has the "capability to catalyze initiation, elongation, and termination" in the process of transcription in prokaryotes?
1. DNA Ligase
2. DNase
3. DNA dependent DNA polymerase
4. DNA dependent RNA polymerase
Identify the labels A, B, C, and D in the figure given below:
| A | B | C | D | |
| 1. | endosperm | scutellum | plumule | seed coat |
| 2. | aleurone | endosperm | radicle | coleorhiza |
| 3. | epithelium | aleurone | plumule | seed coat |
| 4. | endosperm | aleurone | plumule | seed coat and fruit wall |
Correct statement in relation to vacuoles is:
| 1. | It is a triple membrane-bound space found in cytoplasm containing sap. |
| 2. | It can occupy 90% of cell volume in plants. |
| 3. | Its membrane allows the transport of materials along the concentration gradient only. |
| 4. | Concentration of ions is significantly lesser in vacuole than in cytoplasm. |
Find the odd one with respect to the fungi imperfecti:
1. Alternaria
2. Colletotrichum
3. Trichoderma
4. Penicillium
The edible part of the mango is:-
1. Receptacle
2. Epicarp
3. Mesocarp
4. Endocarp
Select the odd one with respect to developmental plasticity:
1. Buttercup
2. Cotton
3. Larkspur
4. Coriander
What is/are done by catalytic converters?
1. Unburnt hydrocarbons are converted to CO2 and H2O
2. CO is converted to CO2
3. Nitric oxide is changed to N2 gas
4. All of these
A male insect mistakes an orchid flower to be female due to its shape and performs the act of copulation and induces pollination. This is an example of:
1. Mimicry
2. Pseudocopulation
3. Pseudo pollination
4. None
Maximum DDT is present in birds feeding on:
1. Fishes
2. Meat
3. Insects
4. Seeds
A mature corn plant absorbs…….. of water in a day while a mustard plant absorbs water equal to its own weight in about……..
1. 5L, 3hours
2. 3L, 5 hours
3. 5L, 5 hours
4. 3L, 3 hours
Select the correct option based on the statements below:
| Assertion (A): | The amount of secondary xylem produced is more than the secondary phloem in the dicot stem. |
| Reason (R): | Cambium is generally more active on the inner side than on the outer. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Match the following:
| Algae | Stored food | Flagellation |
| (a) Brown algae | (p) Floridean starch | (i) 2, lateral |
| (b) Red algae | (q) Mannitol | (ii) 2-4, apical |
| (c) Green algae | (r) Starch | (iii) absent |
Options:
| 1. | (a-r-ii) | (b-p-iii) | (c-q-i) |
| 2. | (a-q-i) | (b-p-iii) | (c-r-ii) |
| 3. | (a-q-i) | (b-r-ii) | (c-p-iii) |
| 4. | (a-q-i) | (b-p-ii) | (c-r-iii) |
With reference to factors affecting the rate of photosynthesis, which of the following statements is not correct?
| 1. | Light saturation for CO2-fixation occurs at 10% of full sunlight. |
| 2. | Increasing atmospheric CO2 concentration by, up to, 0.05% can enhance the CO2-fixation rate |
| 3. | C3 plants respond to higher temperatures with enhanced photosynthesis, while C4 plants have a much lower temperature optimum. |
| 4. | Tomato is a greenhouse crop, which can be grown in a CO2-enriched atmosphere for a higher yield. |
The first step of decarboxylation in cellular respiration is:
1. Conversion of PGAL to DHAP
2. Conversion of PGAL to PGA
3. Conversion of Pyruvate to Acetyl CoA
4. Conversion of BPGA to PGA
Crossing over takes place between which chromatids and in which stage of the cell cycle?
| 1. | Non-sister chromatids of non-homologous chromosomes at Zygotene stage of prophase I. |
| 2. | Non-sister chromatids of homologous chromosomes at Pachytene stage of prophase I. |
| 3. | Non-sister chromatids of homologous chromosomes at Zygotene stage of prophase I. |
| 4. | Non-sister chromatids of non-homologous chromosomes at Pachytene stage of prophase I. |
| (a) | Inhibitor of catalytic activity | (i) | Ricin |
| (b) | Possess peptide bonds | (ii) | Malonate |
| (c) | Cell wall material in fungi | (iii) | Chitin |
| (d) | Secondary metabolite | (iv) | Collagen |
| (a) | (b) | (c) | (d) | |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iii) | (iv) | (i) | (ii) |
| 3. | (ii) | (iii) | (i) | (iv) |
| 4. | (ii) | (iv) | (iii) | (i) |
What term is associated with the sensory structure of the semicircular canal of the internal ear?
1. Macula
2. Ampulla
3. Crista
4. Cupula
Match the following columns and select the correct option:
| Column-I | Column-II | ||
| (a) | Smooth endoplasmic reticulum |
(i) | Protein synthesis |
| (b) | Rough endoplasmic reticulum |
(ii) | Lipid synthesis |
| (c) | Golgi complex | (iii) | Glycosylation |
| (d) | Centriole | (iv) | Spindle formation |
Options:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iii) | (iv) |
| 2. | (iii) | (i) | (ii) | (iv) |
| 3. | (iv) | (ii) | (i) | (iii) |
| 4. | (i) | (ii) | (iii) | (iv) |
Sphincter of Oddi is present at :
1. Gastro-oesophageal junction
2. Junction of jejunum and duodenum
3. Ileo-caecal junction
4. Junction of hepato-pancreatic duct and duodenum
DNA fragments generated by the restriction endonucleases in a chemical reaction can be separated by:
1. Polymerase chain reaction
2. Electrophoresis
3. Restriction mapping
4. Centrifugation
Large scale death of fishes occur in:
| 1. | Saline lake | 2. | Oligotrophic lake |
| 3. | Eutrophic lake | 4. | Shallow lake |
GFR is regulated by:
| 1. | JGA | 2. | ADH |
| 3. | OT | 4. | HGP |
Which of the following helps in the regulation of the respiratory rhythm?
1. Respiratory rhythm centre
2. Pneumotaxic centre
3. Aortic arch and carotid artery receptors
4. All of the above
A canal called as the cerebral aqueduct passes through the:
| 1. | Diencephalon | 2. | Mid brain |
| 3. | 3rd ventricle | 4. | 4th ventricle |
Which of the following is an incorrect statement?
| 1. | Hormones bind to specific proteins located in the target tissues only. |
| 2. | Receptors for hormones can only be present on the cell membrane. |
| 3. | Each receptor is specific to one hormone only. |
| 4. | Nuclear receptors are intracellular receptors. |
Irregular thickening of arterial walls and narrowing of their lumen due to deposition of plaque is:
1. Atherosclerosis
2. Arteriosclerosis
3. Varicose vein
4. Rete Mirabile
Which of the following structure of protein is absolutely necessary for the many biological activities of proteins?
1. Primary
2. Secondary
3. Tertiary
4. Quaternary
| 1. | Cells and tissues are directly bathed in blood |
| 2. | Capillaries are present |
| 3. | Blood is circulated only through a series of vessels of varying diameter |
| 4. | Present in earthworm |
Homozygous purelines in cattle can be obtained by:
1. mating of related individuals of the same breed.
2. mating of unrelated individuals of the same breed.
3. mating of individuals of different breed.
4. mating of individuals of different species.
A foreign DNA and plasmid cut by the same restriction endonuclease can be joined to form a recombinant plasmid using:
| 1. | Ligase | 2. | Eco RI |
| 3. | Taq polymerase | 4. | polymerase II |
Mark out the correct statement regarding the characteristics of variants in a mixed population:
| 1. | Industrialisation results in an increase in the types of variants. |
| 2. | In a mixed population, those that can better adapt, survive and increase in population size. |
| 3. | Less adapted variants will be completely wiped out. |
| 4. | All of these |
Which of the following is an example of Mendelian disorder?
1. Klinefelter's syndrome
2. Down's syndrome
3. Turner's syndrome
4. Thalassemia
Extrusion of the second polar body from the egg nucleus occurs:
1. simultaneously with the first cleavage
2. after entry of sperm but before fertilization
3. after fertilization
4. before entry of sperm into the ovum
What will happen if the secretion of parietal cells of gastric glands is blocked with an inhibitor?
| 1. | Gastric juice will be deficient in chymosin |
| 2. | Gastric juice will be deficient in pepsinogen |
| 3. | In the absence of HCl secretion, inactive pepsinogen is not converted into the active enzyme pepsin |
| 4. | Enterokinase will not be released from the duodenal mucosa and so trypsinogen is not converted to trypsin |
Three of the following statements about enzymes are correct and one is wrong. Which one is wrong?
| 1. | Most enzymes are proteins but some are lipids. |
| 2. | Enzymes require optimum pH for maximal activity. |
| 3. | Enzymes are denatured at high temperatures but in certain exceptional organisms they are effective even at temperatures 80°- 90°C. |
| 4. | Enzymes are highly specific. |
Which one of the following pairs of animals are similar to each other pertaining to the feature stated against them?
| 1. | Pteropus and Ornithorhyncus | Viviparity |
| 2. | Garden lizard and Crocodile | Three chambered heart |
| 3. | Ascaris and Ancylostoma | Metameric segmentation |
| 4. | Sea horse and Flying fish | Cold-blooded (poikilothermal) |
Out of ‘X’ pairs of ribs in humans, only ‘Y’ pairs are true ribs. Select the option that correctly represents values of X and Y and provides their explanation:
| X | Y | |
| 1. | 12 | 15 true ribs are attached dorsally to the vertebral column and sternum on the two ends |
| 2. | 24 | 7 true ribs are dorsally attached to the vertebral column but are free on the ventral side |
| 3. | 24 | 12 true ribs are dorsally attached to the vertebral column but are free on the ventral side |
| 4. | 12 | 7 true ribs are attached dorsally to the vertebral column and ventrally to the sternum |
| Column I | Column II | ||
| A. | IUI | a. | injection of washed sperm into the uterus with a catheter. |
| B. | ZIFT | b. | an egg fertilized in vitro (outside the body) is placed into a woman's fallopian tube. |
| C. | GIFT | c. | eggs are removed from a woman's ovaries, and placed in one of the Fallopian tubes, along with the man's sperm. |
| D. | ICSI | d. | injection of a single sperm directly into a mature egg. |
Codes:
| A | B | C | D | |
| 1. | a | b | c | d |
| 2. | d | b | c | a |
| 3. | a | c | b | d |
| 4. | d | c | b | a |
Vasa recta in cortical nephrons:
| 1. | Arises from afferent arteriole rather than efferent arteriole |
| 2. | Does not get involved in counter-current exchange with the loop of Henle |
| 3. | Carries deoxygenated blood rich in urea |
| 4. | Is absent or highly reduced |
The impulses of sound waves are analysed and recognised in which area of brain?
1. Auditory medulla
2. Auditory cortex
3. Auditory fissure
4. Auditory ridges
The group of neurosecretory cells found in hypothalamus is known as:
| 1. | ganglia | 2. | nuclei |
| 3. | Tract | 4. | nerve |
The correct match is:
|
1. |
 Heroin/smack |
2. |  Morphine |
|
3. |
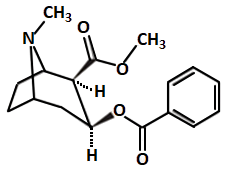 Cannabis sativa Chemical structure of cannabinoid molecule |
4. |  Papaver somniferum Chemical structure of morphine |
Select the correct option based on the statements below:
| Assertion (A): | Spermatogenesis starts at the age of puberty. |
| Reason (R): | Primary spermatocytes undergo second maturation division to become spermatids. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Which of the following statements about ozone is correct?
1. Tropospheric ozone protects us from UV radiation.
2. Stratospheric ozone is 'bad'.
3. Tropospheric ozone is 'good'
4. Stratospheric ozone protects us from UV radiation.
Which of the following statements is not correct?
| 1. | Lysosomes are formed by the process of packaging in the endoplasmic reticulum |
| 2. | Lysosomes have numerous hydrolytic enzymes |
| 3. | The hydrolytic enzyme of lysosomes are active under acidic pH |
| 4. | Lysosomes are membrane bound structure |
What would be the heart rate of a person if the cardiac output is 5L, blood volume in the ventricles at the end of diastole is 100 mL and at the end, ventricular systole is 50 mL?
1. 125 beats per minute
2. 50 beats per minute
3. 75 beats per minute
4. 100 beats per minute
Identify the wrong statement with reference to the transport of oxygen.
| 1. | Partial pressure of CO2 can interfere with O2 binding with haemoglobin. |
| 2. | Higher H+ conc. in alveoli favours with the formation of oxyhaemoglobin. |
| 3. | Low pCO2 in alveoli favours the formation of oxyhaemoglobin. |
| 4. | Binding of oxygen with haemoglobin is mainly related to the partial pressure of O2. |
Match List-I with List-II:
| List-I | List-II |
| (a) Adaptive radiation | (i) Selection of resistant varieties due to excessive use of herbicides and pesticides |
| (b) Convergent evolution | (ii) Bones of forelimbs in Man and Whale |
| (c) Divergent evolution | (iii) Wings of Butterfly and Bird |
| (d) Evolution by anthropogenic action | (iv) Darwin Finches |
Choose the correct answer from the options given below:
| (a) | (b) | (c) | (d) | |
| 1. | (ii) | (i) | (iv) | (iii) |
| 2. | (i) | (iv) | (iii) | (ii) |
| 3. | (iv) | (iii) | (ii) | (i) |
| 4. | (iii) | (ii) | (i) | (iv) |
Which one of the following characteristic is incorrect with respect to cockroach?
| 1. | In females, 7th - 9th sterna together form a genital pouch. |
| 2. | 10th abdominal segment in both sexes, bears a pair of anal cerci. |
| 3. | A ring of gastric caeca is present at the junction of midgut and hind gut. |
| 4. | Hypopharynx lies within the cavity enclosed by the mouth parts. |
A specific recognition sequence identified by endonucleases to make cuts at specific positions within the DNA is:
1. Palindromic Nucleotide sequences
2. Poly(A) tail sequences
3. Degenerate primer sequence
4. Okazaki sequences
| 1. | Rosie cow | - | 1997 |
| 2. | Patent on Basmati rice by an American company | - | 1997 |
| 3. | Human insulin production by Eli Lilly, an American company | - | 1987 |
| 4. | First clinical gene therapy | - | 1990 |
The volume of air expired forcefully after forceful inspiration is:
| 1. | TV+ IRV | 2. | TV+ERV+IRV |
| 3. | TV+ERV+IRV+RV | 4. | None of the above |
The metal present in hemoglobin is:
| 1. | Magnesium | 2. | Sodium |
| 3. | Iron | 4. | Copper |
The primary oocyte completes the first meiotic division at the stage of:
| 1. | Primordial follicle | 2. | Primary follicle |
| 3. | Secondary follicle | 4. | Tertiary follicle |
Given below are four statements A - D each with one or two blanks. Select the option which correctly fills up the blanks in two statements.
| A. | Members of kingdom Animalia are (i) , all of them do not exhibit the same pattern of organisation of cells. Sponges exhibit (ii) level of organisation. |
| B. | For sessile animals, (i) symmetry is advantageous, as it allows the food to be gathered from all sides. |
| C. | Bilateral symmetry arose when animals on the ocean floor became mobile. A crawling animal is most likely to encounter food with the end that goes ahead. Head, enclosing the brain became associated with mouth end. This is called (i) . |
| D. | Notochord is a (i) derived rod-like structure formed on the (ii) side during embryonic development in some animals. |
| 1. | A | B |
| (i) Unicellular or multicellular (ii) Cellular |
(i) Radial | |
| 2. | B | C |
| (i) Bilateral | (i) Cephalisation | |
| 3. | A | D |
| (i) Multicellular (ii) Cellular |
(i) Mesodermally (ii) Dorsal |
|
| 4. | B | D |
| (i) Bilateral | (i) Ectodermally (ii) Dorsal |
In human evolution, the development of the brain and language is an example of:-
1. Convergent evolution
2. Divergent evolution
3. Parallel evolution
4. Adaptive radiation
How many times can human urine be concentrated in a counter-current mechanism?
1. Four times
2. Five times
3. Three times
4. Two times
Five events in the transmission of nerve impulses across the synapse are mentioned below:
| A. | Opening of specific ion channels allows the entry of ions, a new action potential is generated in the post-synaptic neuron |
| B. | Neurotransmitter binds to the receptor on the post-synaptic membrane |
| C. | Synaptic vesicle fuses with pre-synaptic membrane, neurotransmitter releases into the synaptic cleft |
| D. | Depolarization of pre-synaptic membrane |
| E. | Arrival of an action potential at the axon terminal |
In which sequence do these events occur?
| 1. | E → D → C → B → A |
| 2. | A → B → C → D → E |
| 3. | A → B → D → C → E |
| 4. | E → D → C → A → B |
Find out the incorrect statement with respect to the metabolism of calcium in the human body:
| 1. | High level of in the blood stimulates the thyroid gland's parafollicular cells to release calcitonin |
| 2. | Calcitonin promotes deposition of into the bone matrix to reduce blood level |
| 3. | Hypersecretion of PTH causes a deficiency of in the blood which increases the excitability of nerves and muscles and causes sustained muscular contractions (Tetany) |
| 4. | Calcitriol hormone released from kidneys stimulates the absorption of in the gastrointestinal tract |
The anaerobic contraction of skeletal muscle causes the deposition or accumulation of:
1. Gluconic acid
2. Lactic acid
3. Pyruvic acid
4. Hydrochloric acid
The bioreactor shown in the diagram is:
1. Sparged stirred tank type
2. Airlift type
3. Simple stirred tank type
4. Fluidized bed type
Maximum number of existing transgenic animals is of:
1. Fish
2. Mice
3. Cow
4. Pig
The phase of the menstrual cycle in humans that last for 7-8 days is:
1. follicular phase
2. ovulatory phase
3. luteal phase
4. menstruation
Natural methods of birth control include:
1. Abstinence
2. Coitus interruptus
3. Lactational amenorrhoea
4. All of these
Gambusia is a …….. that feeds on ..….
| 1. | Mosquito, larvae |
| 2. | Fish, mosquito-larvae |
| 3. | Frog species, mosquito of stagnant water |
| 4. | Snail, Protozoan |
Which organism has more base pairs?
1. E.coli
2. Bacteriophage
3. human
4. Yeast
Which reagent is used to convert benzoic acid into benzoyl chloride?
1. Cl2, hv
2. SO2
3. SOCl2
4. Cl2, H2O
Considering that , the magnetic moment (in BM) of would be:
1. 0
2. 4.9
3. 6.9
4. 3.5
Consider the following chemical reaction,
\(\mathrm{CH} \equiv \mathrm{CH}\xrightarrow[(2)~~ \mathrm{CO}, ~\mathrm{HCl}, ~\mathrm{AlCl}_3]{\text { (1) } \text { Red hot / Fe tube, } 873 \mathrm{~K}}~~~\text{Product}\)
The number of sp2 hybridized carbon atom(s) present in the product is:
1. 6
2. 7
3. 8
4. 5
The products obtained when chlorine gas reacts with cold and dilute aqueous NaOH are:
1. Cl– and ClO–
2. Cl– and ClO2–
3. ClO– and ClO3–
4. ClO3– and ClO3–
For the reaction:
\(FeO_{(s)} + CO_{(g)} \rightleftharpoons Fe_{(s)} + {CO_2}_{(g)}, K_p = 0.265 \) at 1050 K. If the initial partial pressures are: \(P_{CO}\)= 1.4 atm and \(P_{CO_2}\)= 0.80 atm, the partial pressure of CO2 at equilibrium at 1050 K would be:
| 1. | 4.61 atm | 2. | 1.74 atm |
| 3. | 0.46 atm | 4. | 0.17 atm |
The polyamide among the following is-
1. Bakelite
2. Terylene
3. Nylon-6,6
4. Teflon
The term anomers of glucose refer to:
| 1. | Isomers of glucose that differ in configurations at carbons one and four (C-1 and C-4) |
| 2. | A mixture of (D)-glucose and (L)-glucose |
| 3. | Enantiomers of glucose |
| 4. | Isomers of glucose that differ in configuration at carbon one (C-1) |
Compound A given below is :
1. Antiseptic
2. Antibiotic
3. Analgesic
4. Pesticide
In a mixture of A and B, components show negative deviations when:
| 1. | A-B interaction is stronger than A-A and B-B interaction |
| 2. | A-B interaction is weaker than A-A and B-B interaction |
| 3. | \(\Delta V_{\text {mix }}>0, \quad \Delta S_{\text {mix }}>0\) |
| 4. | \(\Delta V_{\text {mix }}=0, \quad \Delta S_{\text {mix }}>0\) |
The number of atoms in 560 g of Fe (atomic mass 56 g mol–1) is:
| 1. | Twice that of 70 g N |
| 2. | Half that of 20 g H |
| 3. | Both 1 and 2 |
| 4. | None of the above |
Consider the following reactions at 1100oC
\((I)\,\, 2 \mathrm{C}+\mathrm{O}_{2} \longrightarrow 2 \mathrm{CO}, \Delta G^{\circ}=-460 \mathrm{~kJ} \mathrm{~mol}^{-1}\\ (II)\,\, 2 \mathrm{Zn}+\mathrm{O}_{2} \longrightarrow 2 \mathrm{ZnO}, \Delta G^{\circ}=-360 \mathrm{~kJ} \mathrm{~mol}^{-1}\)
Based on these, select correct alternate :
1. Zinc can be oxidised by CO.
2. Zinc oxide can be reduced by carbon.
3. Both 1 and 2.
4. None is correct.
According to the periodic law of elements, the variation in properties of elements is related to their:
1. Atomic masses
2. Nuclear mass
3. Atomic numbers
4. Nuclear neutron-proton number ratio
Among the following, the incorrect characteristics for physical adsorption is-
| 1. | Adsorption on solids is reversible |
| 2. | Adsorption increase with the increase in temperature |
| 3. | Adsorption is spontaneous |
| 4. | Both enthalpy and entropy of adsorption are negative |
Bottles containing C6H5I and C6H5CH2I lost their original labels. They were labeled as A and B respectively for testing. A and B were separately taken in a test tube and boiled with NaOH solution. The end solution in each test tube was made acidic with dilute HNO3 and then some AgNO3 was added. A substance present in test tube B gave a yellow precipitate. The true statement among the following regarding the experiment is:
1. A was C6H5I
2. A was C6H5CH2I
3. B was C6H5I
4. Addition of HNO3 was unnecessary
The elementary step of the reaction, \(2 \mathrm{Na}+\mathrm{Cl}_2\rightarrow2 \mathrm{NaCl}\) is found to follow 3rd order kinetics. Its molecularity is:
| 1. | One(1) | 2. | Two(2) |
| 3. | Three(3) | 4. | Four(4) |
Identify the compound B in the reaction sequence shown below:
| 1. | 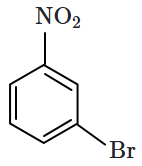 |
2. | 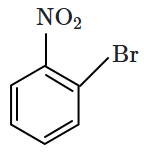 |
| 3. | 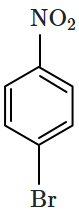 |
4. | 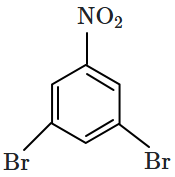 |

| 1. |  |
2. |  |
| 3. |  |
4. | None of these |
| I: | [Fe(H2O)6]3+ is strongly paramagnetic whereas [Fe(CN)6]3− is weakly paramagnetic. |
| II: | [Fe(H2O)6]3+ has 4 unpaired electrons while [Fe(CN)6]3− has 5 unpaired electrons. |
1. Both I and II are true.
2. I is true and II is false.
3. Both I and II are false.
4. I is false but II is true.
A compound among the following that is not a monomer for a high molecular mass silicone polymer is-
1.
2. Me3SiCl
3. PhSiCl3
4. MeSiCl3
The correct sequence of increasing order of viscosity among the following is -
1. Hexane < Water = Glycerine
2. Hexane > Water < Glycerine
3. Hexane < Water > Glycerine
4. Hexane < Water < Glycerine
The ionization constant of some weak bases at a particular temperature is given below:
| Base | Dimethylamine | Urea | Pyridine | Ammonia |
| Kb | 5.4 × 10-4 | 1.3 × 10-14 | 1.77 × 10-9 | 1.77 × 10-5 |
The decreasing order of the bases on the extent of their ionization at equilibrium is:
| 1. | Urea > Ammonia > Dimethylamine > Pyridine |
| 2. | Ammonia > Dimethylamine > Pyridine > Urea |
| 3. | Pyridine > Urea > Dimethylamine > Ammonia |
| 4. | Dimethylamine > Ammonia > Pyridine > Urea |
| Column I(Compound) | Column II(Oxidation state) |
| a. Cl2O7 | i. +4 |
| b. NaClO3 | ii. +1 |
| c. Cl2O | iii. +5 |
| d. ClO2 | iv. +7 |
| a | b | c | d | |
| 1. | iii | iv | ii | i |
| 2. | ii | iii | iv | i |
| 3. | iv | iii | ii | i |
| 4. | iv | i | iii | ii |
| 1. | Down the group hydration energy decreases |
| 2. | Down the group lattice energy increases |
| 3. | Down the group both hydration and lattice energy increases |
| 4. | None of the above |
 1. 2,4- Dinitrophenol
1. 2,4- Dinitrophenol | 1. |  |
2. |  |
| 3. |  |
4. |  |
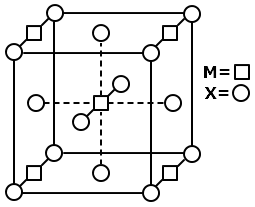
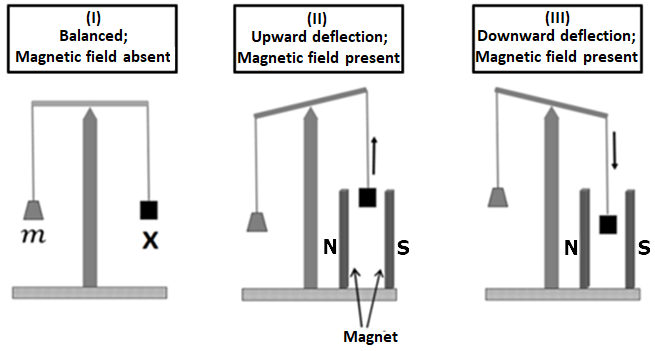
| a. | If X is \(H_2O(l)\), deflection of the pan is upwards |
| b. | If X is \(K_4[Fe(CN)_6](s)\), deflection of the pan is upwards |
| c. | If X is \(O_2 (g)\), deflection of the pan is downwards |
| d. | If X is \(C_6H_6 (l)\) deflection of the pan is downwards |
| 1. | \(2,\ 0,\ 0,\ +\frac12 \) | 2. | \(2,\ 1,\ -1,\ +\frac12\) |
| 3. | \(3,\ 1,\ +1,\ +\frac12 \) | 4. | \(3,\ 0,\ +2,\ \frac12\) |
| 1. | \(\mathrm{C}(s)+\mathrm{HCl}(g)+\mathrm{H}_{2}(g) \longrightarrow \mathrm{CH}_{3} \mathrm{Cl}(g)\) |
| 2. | \(\mathrm{C}(\mathrm{s})+3 \mathrm{H}(\mathrm{g})+\mathrm{Cl}(g) \longrightarrow \mathrm{CH}_{3} \mathrm{Cl}(g)\) |
| 3. | \(\mathrm{C}(\mathrm{s})+3 / 2 \mathrm{H}_{2}(g)+1 / 2 \mathrm{Cl}_{2}(g) \longrightarrow \mathrm{CH}_{3} \mathrm{Cl}(g) \) |
| 4. | \(\mathrm{CH}_{4}(g)+\mathrm{Cl}_{2}(g) \longrightarrow \mathrm{CH}_{3} \mathrm{Cl}(g)+\mathrm{HCl}(g)\) |
| 1. | Formation of chlorine nitrate(ClONO2) |
| 2. | Formation of HCl |
| 3. | Formation of HOCl and Cl2, which is converted back into reactive Cl atoms |
| 4. | None of the above |
Number of isomers of complex are:
| 1. | 2 | 2. | 3 |
| 3. | 4 | 4. | 5 |
General behavior of is:
1. To give electrons
2. To Give
3. Reaction with
4. To accept electrons
The major products of the following reaction are:
| 1. |  |
2. |  |
| 3. |  |
4. | 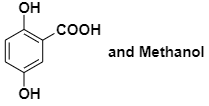 |
The molecule in which hybrid MOs involve only one d-orbital of the central atom is:
| 1. | \(X e F_{4}\) | 2. | \(\left[ N i (CN)_{4} \right]^{2 -}\) |
| 3. | \(B r F_{5}\) | 4. | \(\left[ C r F_{6} \right]^{3 -}\) |
Phenol on treatment with CO2 in the presence of NaOH followed by acidification produces compound X as the major product. X on treatment with (CH3CO)2O in the presence of catalytic amount of H2SO4 produces:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
C6H6 freezes at 5.5°C. The temperature at which a solution of 10 g of C4H10 in 200 g of C6H6 will freeze is:
(The molal freezing point depression constant of C6H6 is 5.12°C/m.)
| 1. | 2 °C | 2. | 1 °C |
| 3. | 6 °C | 4. | 5 °C |
The correct expression for the following reaction is:
Fe2N(s) + \(\frac{3}{2}\)H2(g) \(\leftrightharpoons \) 2Fe(s) + NH3(g)
| 1. | 2. | ||
| 3. | 4. |
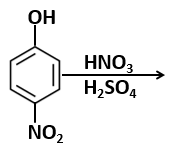
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is:
(to the nearest integer) (Atomic mass : C =12; H=1; O=16)
| 1. | 65 | 2. | 69 |
| 3. | 73 | 4. | 76 |
The rate of a reaction is decreased by 3.555 times when the temperature was changed from 40°C to 30°C. The activation energy (in kJ ) of the reaction is:
(Take R=8.314 J In 3.555=1.268)
1. 100 kJ/mol
2. 120 kJ/mol
3. 95 kJ/mol
4. 108 kJ/mol
Given
\(\begin{aligned} &\mathrm{{C}_{{(graphite) }}+{O}_{2}({~g})} → \mathrm{{CO}_{2}({~g})} \\ &\mathrm{\Delta_{r} {H}^{\circ}=-393.5 {~kJ} {~mol}^{-1}} \\ &\mathrm{H_{2}(g) + \frac{1}{2} {O}_{2}({~g})} → \mathrm{{H}_{2} {O}({l})} \\ &\mathrm{\Delta_{r} {H}^{\circ}=-285.8 {~kJ} {~mol}^{-1}} \\ &\mathrm{{CO}_{2}({~g})+2 {H}_{2} {O}({l})} → \mathrm{{CH}_{4}({~g})+2 {O}_{2}({~g})} \\ &\mathrm{\Delta_{r} {H}^{\circ}=+890.3 {~kJ} {~mol}^{-1}} \end{aligned}\)
Based on the above thermochemical equations, the value of ΔrH° at 298 K for the reaction
\(\mathrm{C_{(graphite)} + 2 H_{2} (g) → CH_{4} (g)}\) will be :
1. –74.8 kJ mol–1
2. –144.0 kJ mol–1
3. +74.8 kJ mol–1
4. +144.0 kJ mol–1
The reduction potential of hydrogen half-cell will be negative if:
1. P(H2) = 1atm and [H+] = 2.0 M
2. P(H2) = 1 atm and [H+] = 1.0 M
3. P(H2) = 2 atm and [H+] = 1.0 M
4. P(H2) = 2 atm and [H+] = 2.0 M
Which of the following pairs are metamers of ethyl propionate?
1. C4H9COOH and HCOOC4H9
2. C4H9COOH and CH3COOC3H7
3. CH3COOCH3 and CH3COOC3H7
4. CH3COOC3H7 and C3H7COOCH3
The molecular shapes of SF4, CF4, and XeF4 are:
| 1. | Different with 1, 0, and 2 lone pairs of electrons on the central atom, respectively |
| 2. | Different with 0, 1, and 2 lone pairs of electrons on the central atom, respectively |
| 3. | The same with 1, 1, and 1 lone pair of electrons on the central atoms, respectively |
| 4. | The same with 2, 0, and 1 lone pair of electrons on the central atom, respectively |
A hydrocarbon of molecular formula C6H10 reacts with sodamide and the same on ozonolysis followed by hydrogen peroxide oxidation gives two molecules of carboxylic acids, one being optically active. The hydrocarbon maybe:
| 1. | 1-Hexyne | 2. | 3-Hexyne |
| 3. | 3-Methyl-1-pentyne | 4. | 3,3-Dimethyl-1-butyne |
The ammonia evolved from the treatment of 0.30 g of an organic compound for the estimation of nitrogen was passed in 100 mL of 0.1 M sulphuric acid. The excess acid required 20 mL of 0.5 M sodium hydroxide solution for complete neutralization. The percentage of nitrogen in the organic compound is:
1. 46.6 %
2. 50.4 %
3. 42.8 %
4. 40.5 %
A stationary horizontal disc is free to rotate about its axis. When a torque is applied on it, its kinetic energy as a function of \(\theta,\) where \(\theta\) is the angle by which it has rotated, is given as \(k\theta^2\) (where \(k\) is constant). If its moment of inertia is \(I,\) then the angular acceleration of the disc is:
1. \(\frac{k}{I} \theta\)
2. \(\frac{k}{2 I} \theta\)
3. \(\frac{k}{4 I} \theta\)
4. \(\frac{2 k}{I} \theta\)
| 1. | \(0^\circ\) | 2. | \(45^\circ\) |
| 3. | \(180^\circ\) | 4. | \(360^\circ\) |
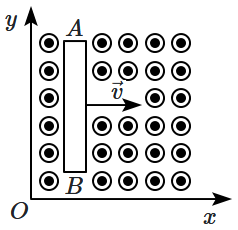
| 1. | positively charged |
| 2. | negatively charged |
| 3. | neutral |
| 4. | first positively charged and then negatively charged |
| 1. | \(\dfrac{I_1+I_2}{\sqrt2}\) | 2. | \(\dfrac{(I_1+I_2)^2}{\sqrt2}\) |
| 3. | \(\dfrac{{\left({{I}_{1}^{2}{+}{I}_{2}^{2}}\right)}^{1/2}}{\sqrt{2}}\) | 4. | \(\dfrac{1}{2}{\left({{I}_{1}^{2}{+}{I}_{2}^{2}}\right)}^{1/2}\) |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | \(\dfrac{\left({{W}_{1}{+}{W}_{2}}\right)}{A}\) | 2. | \(\dfrac{\left({{W}_{1}{-}{W}_{2}}\right)}{A}\) |
| 3. | \(\dfrac{\left({{W}_{1}/2}\right){+}{W}_{2}}{A}\) | 4. | \(\dfrac{\left({{W}_{2}/2}\right){+}{W}_{1}}{A}\) |

| 1. | \(8\) | 2. | \(\frac{1}{2\sqrt{2}}\) |
| 3. | \(3\) | 4. | \(4\) |
| 1. | when an electric field is changing in the circuit. |
| 2. | when an electric field is constant. |
| 3. | when an electric field is absent. |
| 4. | always exists independent of the electric field. |
| 1. | \(2:1\) | 2. | \(4:1\) |
| 3. | \(8:1\) | 4. | \(16:1\) |
| 1. | reflected beam is polarised \(100\) percent. |
| 2. | reflected and refracted beams are partially polarised. |
| 3. | the reason for (1) is that almost all the light is reflected. |
| 4. | all of the above. |
| 1. | \(11 \text{%}\) | 2. | \(4 \text{%}\) |
| 3. | \(7 \text{%}\) | 4. | \(9 \text{%}\) |
| 1. | \(\dfrac{9}{2}\) | 2. | \(\dfrac{8}{3}\) |
| 3. | \(2\) | 4. | \(1\) |
| 1. | \(\dfrac{g}{R}\) | 2. | \(\dfrac{R}{g}\) |
| 3. | \(gR\) | 4. | \(\dfrac{g}{R^{2}}\) |
| 1. | to make it attractive |
| 2. | for shining |
| 3. | to absorb all radiation from outside |
| 4. | to reflect all radiation from outside |
| 1. | \(V\) volts | 2. | \(2000V\) volts |
| 3. | \(\dfrac{V}{2000} \) volts | 4. | \(\sqrt{2000}V\) volts |
| 1. | \(1~\text{eV}\) | 2. | \(5~\text{eV}\) |
| 3. | \(1~\text{keV}\) | 4. | \(1~\text{MeV}\) |
| 1. | hit the monkey. |
| 2. | go above the monkey. |
| 3. | go below the monkey. |
| 4. | hit the monkey if the initial velocity of the bullet is more than a definite velocity. |
| 1. | \(50~\text{ms}^{-1}\) | 2. | \(9~\text{ms}^{-1}\) |
| 3. | \(40~\text{ms}^{-1}\) | 4. | \(90~\text{ms}^{-1}\) |
| 1. | Kinetic energy |
| 2. | Momentum |
| 3. | Neither momentum nor kinetic energy |
| 4. | Momentum and kinetic energy |
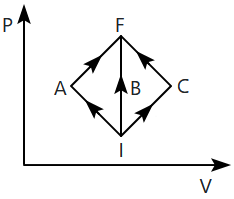
| 1. | the same in all three processes. |
| 2. | the same in (i) and (ii). |
| 3. | greater in (i) and (ii) than in (iii). |
| 4. | the same in (i) and (iii). |
| 1. | \(10~\text{cm}\) | 2. | \(75~\text{cm}\) |
| 3. | \(13.6~\text{cm}\) | 4. | \(1.36~\text{cm}\) |
The initial velocity \(v_i\) required to project a body vertically upwards from the surface of the earth to just reach a height of \(10R\), where \(R\) is the radius of the earth, described in terms of escape velocity \(v_e\) is:
| 1. | \(\sqrt{\dfrac{10}{11}}v_e\) | 2. | \(\sqrt{\dfrac{11}{10}}v_e\) |
| 3. | \(\sqrt{\dfrac{20}{11}}v_e\) | 4. | \(\sqrt{\dfrac{11}{20}}v_e\) |
A particle is thrown with a velocity of \(\mathrm{u}\) m/s. If it passes through A and B as shown in the figure at time \(t_1=1~\text{s}\) and \(t_2=3~\text{s}\), then the value of \(\mathrm{h}\) is: ( Take \(g=10~\text{m/s}^{2}\))
| 1. | \(15~\text{m}\) | 2. | \(10~\text{m}\) |
| 3. | \(30~\text{m}\) | 4. | \(20~\text{m}\) |
| 1. | \(2~\Omega\) | 2. | \(20~\Omega\) |
| 3. | \(48~\Omega\) | 4. | \(96~\Omega\) |
| 1. | voltage of the ball becomes high enough to destroy the insulating property of air. |
| 2. | capacity of the ball is too high to hold the charge. |
| 3. | nuclear forces wipe off the charge of the ball. |
| 4. | electromagnetic forces reduce the charge of the ball. |
| 1. | \(W=\pi r^{2}T\) | 2. | \(W=2\pi rT\) |
| 3. | \(W=2\pi r^{2}T \) | 4. | \(W=\frac{4}{3}(\pi r^{3}T)\) |

| 1. | \({10}^{3}\mathit{\pi}\) J | 2. | \({10}^{2}\mathit{\pi}\) J |
| 3. | \({10}^{4}\mathit{\pi}\) J | 4. | \({10}^{7}\mathit{\pi}\) J |
| 1. | hoop |
| 2. | disc |
| 3. | both the hoop and the disc |
| 4. | none of these |