What is the net gain of the number of ATP molecules produced during aerobic respiration of one molecule of glucose?
1. 30
2. 36
3. 38
4. 40
There is a possibility of exposure of the maternal blood [of Rh -ve mother] to small amounts of Rh +ve blood from the fetus during the:
1. First trimester of any pregnancy
2. Third trimester of the second pregnancy only
3. Delivery of the child
4. There is no such possibility
The useful purpose served by lactate fermentation is:
| 1. | Make lactose available for gluconeogenesis |
| 2. | Production of additional ATP in anaerobic conditions |
| 3. | Regeneration of NAD+ |
| 4. | Increased availability of oxygen for the skeletal muscle |
Which of the following Fungi is not capable of causing ringworm infection in a man?
1. Microsporum
2. Trichophyton
3. Epidermophyton
4. Trichoderma
Sexual reproduction by non-flagellated but similar in size gametes is seen in:
1. Chlamydomonas
2. Volvox
3. Spirogyra
4. Fucus
Identify the incorrectly matched pair:
| Animals | Feature present in both | |
| 1. | Balanoglossus and Pinctada | Open circulatory system |
| 2. | Branchiostoma and Ascidia | Persistent notochord |
| 3. | Aplysia and Pheretima | True coelom |
| 4. | Gorgonia and Pennatula | Cnidoblasts |
Pteropus is a/an:
| 1. | Limbless amphibian |
| 2. | Extinct reptile that evolved into mammals |
| 3. | Bony fish that migrates from sea water to fresh water for breeding |
| 4. | Mammal with flight |
In animal cells, lipid-like steroidal hormones are synthesized in the:
1. Nucleus
2. Rough endoplasmic reticulum
3. Smooth endoplasmic reticulum
4. Golgi apparatus
Two phenotypically normal individuals have an affected child. What can we conclude about the parents?
1. they both carried the diesease allele
2. they are not the parents of the child
3. they are affected
4. no conclusions can be drawn
What is the basis of pleiotropy?
1. A spontaneous mutation during the replication of DNA.
2. Interrelationship between various metabolic pathways in the body.
3. Chromosomal aberration as chromosomes are the vehicles of genes.
4. the behaviour of chromosomes during meiosis or gamete formation.
The skeletal structure shown in the given diagram shows the structure of :
| 1. | Morphine | 2. | Diacetyl morphine |
| 3. | Cannabinoid | 4. | Cocaine |
In general, which of the following is not an adaptation seen in parasites in accordance with their life style?
1. Loss of unnecessary sense organs
2. Presence of adhesive organs
3. Low reproductive capacity
4. Loss of digestive system
Which of the following taxonomic categories contains organisms least similar to one another?
1. Class
2. Genus
3. Family
4. Species
Genetic information in a DNA molecule is coded in the:
1. Sequence of nucleotides
2. Base pairings
3. Proportion of each base present
4. The turning pattern of the helix
In DNA replication, the Okazaki fragments on the lagging strand are joined together by:
1. DNA ligase
2. DNA polymerase
3. Primase
4. Helicase
The enzyme that catalyzes the peptide bonding in prokaryotes is located in the:
1. Leader region of the mRNA
2. Central part of tRNA
3. Smaller subunit of the ribosome
4. Larger subunit of the ribosome
A woman receives her X chromosomes from:
1. Her mother only
2. Her father only
3. Both her mother and father
4. Mitochondria of mother only
Cells in some filamentous cyanobacteria that are specialized for nitrogen fixation are called:
1. Phycobilisomes
2. Chromatophores
3. Grana
4. Heterocysts
Atrial natriuretic hormone (ANH) will promote a(n) ________ in the excretion of sodium and water; thus ________ blood volume and blood pressure.
1. decrease, decreasing
2. decrease; increasing
3. increase, decreasing
4. increase, increasing
Which of the following would not be a feature seen in a patient with the following karyotype ?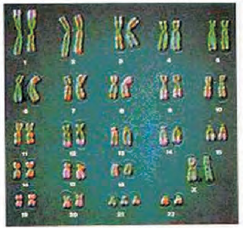
1. Many loops on finger tips
2. Congenital heart disease
3. Big and wrinkled tongue
4. Mucus clogging of airways
Given below is the initial amino acids of the beta chain of a hemoglobin molecule. What will be true about this hemoglobin molecule ?
| 1. | The RBCs carrying this molecule will undergo sickling at high oxygen tension |
| 2. | The RBCs carrying this molecules will undergo sickling at low oxygen tension |
| 3. | There will be a quantitative decrease in the synthesis of this molecule |
| 4. | This is normal beta chain of the hemoglobin molecule |
Consider the following statements:
| I. | Myasthenia gravis is an autoimmune disorder affecting the neuromuscular junction |
| II. | Muscular dystrophy is a progressive degeneration of skeletal muscles mostly due to a genetic disorder |
| III. | Tetany is rapid spasms in muscles due to high Ca++ in body fluid. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
Consider the following statements:
I. Somatic neural system relays impulses from CNS to skeletal muscles.
II. Autonomic neural system transmits impulses from CNS to involuntary organs and smooth muscles.
III. Unmyelinated nerve fibres are commonly found in spinal and cranial nerves.
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II, and III
Consider the following statements:
| I: | The resting axonal membrane is nearly impermeable to sodium ions. |
| II: | Depolarization of the axonal membrane is due to the influx of sodium ions. |
| III: | The size of the action potential, if produced, does not depend on the strength of the stimulus. |
Which of the above statements are true?
1. I and II only
2. I and III only
3. II and III only
4. I, II and III
The rate of enzyme action decreases at higher temperature [after a certain value] because the increased heat
| 1. | changes the pH of the system |
| 2. | alters the active site of the enzyme |
| 3. | neutralizes the acids and bases in the system |
| 4. | increases the concentration of the enzyme |
The animals belonging to all the following groups are exclusively marine except:
1. Ctenophora
2. Porifera
3. Protochordata
4. Echinodermata
Non-membranous nucleoplasmic structures in the nucleus, are the sites for active synthesis of:
| 1. | protein | 2. | mRNA |
| 3. | rRNA | 4. | tRNA |
Match the following enzymes with their functions:
|
Column-I |
Column-II |
||
|
(a) |
Restriction endonuclease |
(i) |
joins the DNA fragments |
|
(b) |
Exonuclease |
(ii) |
extends primers on genomic DNA template |
|
(c) |
DNA ligase |
(iii) |
cuts DNA at a specific position |
|
(d) |
Tag polymerase |
(iv) |
removes nucleotides from the ends of DNA |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (iii) | (i) | (iv) | (ii) |
| 2. | (iii) | (iv) | (i) | (ii) |
| 3. | (iv) | (iii) | (i) | (ii) |
| 4. | (ii) | (iv) | (i) | (iii) |
The two antibiotic resistance genes on vector pBR 322 are for:
1. Ampicillin and Tetracycline
2. Ampicillin and Chloramphenicol
3. Chloramphenicol and Tetracycline
4. Tetracycline and Kanamycin
The exploitation of bioresources of a nation by multinational companies without authorization from the concerned country is referred to as:
| 1. | Bioweapon | 2. | Biopiracy |
| 3. | Bioethics | 4. | Biowar |
Carnivorous animals - lions and leopards, occupy the same niche but lions predate mostly larger animals and leopards take smaller ones. This mechanism of competition is referred to as:
| 1. | Character displacement | 2. | Altruism |
| 3. | Resource partitioning | 4. | Competitive exclusion |
Artificial light, extended work-time and reduced sleep-time disrupt the activity of:
| 1. | Thymus gland | 2. | Pineal gland |
| 3. | Adrenal gland | 4. | Posterior pituitary gland |
Which of the following conditions will stimulate parathyroid gland to release parathyroid hormone?
1. Fall in active Vitamin D levels
2. Fall in blood Ca+2 levels
3. Fall in bone Ca+2 levels
4. Rise in blood Ca+2 levels
Which of the following animals are true coelomates with bilateral symmetry?
| 1. | Adult Echinoderms | 2. | Aschelminthes |
| 3. | Platyhelminthes | 4. | Annelids |
The maximum volume of air a person can breathe in after a forced expiration is known as:
1. Expiratory Capacity
2. Vital Capacity
3. Inspiratory Capacity
4. Total Lung Capacity
Match the following parts of a nephron with their function:
| (a) | Descending limb of Henle's loop | (i) | Reabsorption of salts only |
| (b) | Proximal convoluted tubule | (ii) | Reabsorption of water only |
| (c) | Ascending limb of Henle's loop | (iii) | Conditional reabsorption of sodium ions |
| (d) | Distal convoluted tubule | (iv) | Reabsorption of ions, water, and organic nutrients |
Select the correct option from the following:
| Options: | (a) | (b) | (c) | (d) |
| 1. | i | iii | ii | iv |
| 2. | ii | iv | i | iii |
| 3. | i | iv | ii | iii |
| 4. | iv | i | iii | ii |
Which of the following statements is correct?
1. Lichens do not grow in polluted areas.
2. Algal component of lichens is called mycobiont.
3. Fungal component of lichens is called phycobiont.
4. Lichens are not good pollution indicators.
From the following, identify the correct combination of salient features of the Genetic Code:
| 1. | Universal, Non-ambiguous, Overlapping |
| 2. | Degenerate, Overlapping, Commaless |
| 3. | Universal, Ambiguous, Degenerate |
| 4. | Degenerate, Non-overlapping, Non-ambiguous |
In the process of transcription in Eukaryotes, the RNA polymerase I transcribe -
1. mRNA with additional processing, capping and tailing
2. tRNA, 5 S rRNA and snRNAs
3. rRNAs - 28 S, 18 S and 5.8 S
4. Precursor of mRNA, hnRNA
What initiation and termination factors are involved in transcription in prokaryotes?
1. , respectively
2. , respectively
3. , respectively
4. , respectively
Prosthetic groups differ from co-enzymes, in which:
| 1. | they require metal ions for their activity. |
| 2. | they (prosthetic groups) are tightly bound to apoenzymes. |
| 3. | their association with apoenzymes is transient. |
| 4. | they can serve as co-factors in a number of enzyme-catalyzed reactions. |
"Ramachandran plot" is used to confirm the structure of:
1. RNA
2. Proteins
3. Triacylglycerides
4. DNA
Where is the respiratory electron transport system (ETS) located in plants?
1. Mitochondrial matrix
2. Outer mitochondrial membrane
3. Inner mitochondrial membrane
4. Intermembrane space
Removal of shoot tips is a very useful technique to boost the production of tea leaves. This is because:
| 1. | Gibberellins prevent bolting and are inactivated. |
| 2. | Auxins prevent leaf drop at early stages. |
| 3. | Effect of auxins is removed and the growth of lateral buds is enhanced. |
| 4. | Gibberellins delay the senescence of leaves. |
Match the placental types Column-I with their examples Column-II.
| Column-I | Column-II | ||
| (a) | Basal | (i) | Mustard |
| (b) | Axile | (ii) | China rose |
| (c) | Parietal | (iii) | Dianthus |
| (d) | Free central | (iv) | Sunflower |
Choose the correct answer from the following options:
| Options: | (a) | (b) | (c) | (d) |
| 1. | (ii) | (iii) | (iv) | (i) |
| 2. | (i) | (ii) | (iii) | (iv) |
| 3. | (iv) | (ii) | (i) | (iii) |
| 4. | (iii) | (iv) | (i) | (ii) |
A selectable marker is used to:
| 1. | help in eliminating the non-transformants so that the transformants can be regenerated. |
| 2. | identify the gene for the desired trait in an alien organism. |
| 3. | select a suitable vector for transformation in a specific crop. |
| 4. | mark a gene on a chromosome for isolation using a restriction enzyme. |
Western Ghats have a large number of plant and animal species that are not found anywhere else. Which of the following terms will you use to notify such species?
| 1. | Endemic | 2. | Vulnerable |
| 3. | Threatened | 4. | Keystone |
Between which of the following, the relationship is not an example of commensalism?
| 1. | Orchid and the tree on which it grows |
| 2. | Cattle Egret and grazing cattle |
| 3. | Sea Anemone and Clownfish |
| 4. | Female wasp and fig species |
An enzyme catalysing the removal of nucleotides from ends of DNA is:
| 1. | DNA ligase | 2. | Endonuclease |
| 3. | Exonuclease | 4. | Protease |
In RNAi, the genes are silenced using:
| 1. | dsRNA | 2. | ssDNA |
| 3. | ssRNA | 4. | dsDNA |
Oxygen binding to haemoglobin in blood is:
| 1. | directly proportional to the concentration of in the medium. |
| 2. | inversely proportional to the concentration of in the medium. |
| 3. | directly proportional to the concentration of CO in the medium. |
| 4. | independent of the concentration of CO in the medium. |
Which one of the following four glands is correctly matched with the accompanying description?
1. Thyroid - hyperactivity in young children causes cretinism
2. Thymus - starts undergoing atrophy after puberty
3. Parathyroid - secrete parathormone which promotes movement of calcium ions from blood into bones during calcification
4. Pancreas - Delta cells of the Islets of Langerhans secrete a hormone which stimulates glycolysis
Which one of the following statements is correct?
| 1. | Neurons regulate endocrine activity, but not vice versa. |
| 2. | Endocrine glands regulate neural activity, and nervous system regulates endocrine glands. |
| 3. | Neither hormones control neural activity nor do the neurons control endocrine activity. |
| 4. | Endocrine glands regulate neural activity, but not vice versa |
Which one of the following statements about human sperm is correct?
| 1. | Acrosome has a conical pointed structure used for piercing and penetrating the egg, resulting in fertilization |
| 2. | The sperm lysins in the acrosome dissolve the egg envelope facilitating fertilization |
| 3. | Acrosome serves as a sensory structure leading the sperm towards the ovum |
| 4. | Acrosome serves no particular function |
The second maturation division of the mammalian ovum occurs
1. shortly after ovulation before the ovum makes entry into the Fallopian tube
2. after the ovum has been penetrated by a sperm
3. until the nucleus of the sperm has fused with that of the ovum
4. in the Graafian follicle following the first maturation division
Which one of the following is the correct matching of the events occurring during menstrual cycle?
|
|
Column I |
Column II |
|
|
1. |
Ovulation |
LH and FSH attain peak level and sharp fall in the secretion of progesterone |
|
|
2. |
Proliferative phase |
Rapid regeneration of myometrium and maturation of Graafian follicle |
|
|
3. |
Development of corpus luteum |
Secretory phase and increased secretion of progesterone |
|
|
4. |
Menstruation |
|
What is the correct sequence of sperm formation?
| 1. | Spermatid, Spermatocyte, Spermatogonia, Spermatozoa |
| 2. | Spermatogonia, Spermatocyte, Spermatozoa, Spermatid |
| 3. | Spermatogonia, Spermatozoa, Spermatocyte, Spermatid |
| 4. | Spermatogonia, Spermatocyte, Spermatid, Spermatozoa |
Contraceptive oral pills help in birth control by
| 1. | killing the sperms in uterus |
| 2. | preventing implantation |
| 3. | preventing ovulation |
| 4. | both 2. and 3. |
The technique called gamete intrafallopian transfer (GIFT) is recommended for those females
1. Who cannot provide a suitable environment for fertilisation.
2. Who cannot produce an ovum.
3. Who cannot retain the fetus inside the uterus.
4. Whose cervical canal is too narrow to allow passage for the sperms
What is true for individuals of same species?
1. Live in same niche
2. Live in same habitat
3. Interbreeding
4. Live in different habitat
Areolar connective tissue joins
1. integument to the muscles
2. bones to the muscles
3. bones to the bones
4. fat body to the muscles
Maximum nutritional diversity is found in the group.
1. Fungi
2. Animalia
3. Monera
4. Plantae
At what stage of the cell cycle are histone proteins synthesized in a eukaryotic cell
1. During telophase
2. During S-phase
3. During G2 stage of prophase
4. During entire prophase
Nucellar polyembryony is reported in species
1. Citrus
2. Gossypium
3. Triticum
4. Brassica
The gynoecium consists of many free pistils in flowers of
1. Aloe
2. Tomato
3. Papaver
4. Michelia
The hilum is a scar on the:
1. Fruit, where it was attached to the pedicel
2. Fruit, where style was present
3. Seed, where micropyle was present
4. Seed, where funicle was attached
Which one of the following may require pollinators, but is genetically similar to autogamy?
1. Xenogamy
2. Apogamy
3. Cleistogamy
4. Geitonogamy
A dioecious flowering plant prevents both:
1. Autogamy and xenogamy
2. Autogamy and geitonogamy
3. Geitonogamy and xenogamy
4. Cleistogamy and xenogamy
A common biocontrol agent for the control of plant diseases is
1. Trichoderma
2. Baculovirus
3. Bacillus thuringiensis
4. Glomus
An organism used as biofertilizer for raising soyabean crop is
1. Nostoc
2. Azotobacter
3. Azospirillum
4. Rhizobium
The figure below shows three steps (A, B, C) of Polymerase Chain Reaction (PCR). Select the option giving correct ‘identification together with what it represents:
| 1. | B - Denaturation at a temperature of about 98°C separating the two DNA strands |
| 2. | A - Denaturation at a temperature of about 50°C |
| 3. | C - Extension in the presence of heat-stable DNA polymerase |
| 4. | A - Annealing with two sets of primers |
Some of the characteristics of BT cotton are
1. Medium yield, long fibre and resistance to beetle pests
2. High yield and production of toxic protein crystals which kill dipterans pests
3. High yield and resistance to bollworms
4. Long fibre and resistance to aphids
In fungi, asexual reproduction takes place by
1. Fission, conidia and ascospores
2. Conidia, hypnospores and zoospores
3. Conidia, sporangiospores and zoospores
4. Sporangiospores, conidia and basidiospores
Photosynthetic organ originates from _______ meristem and arranged in ______ order.
| 1. | Lateral, basipetal |
| 2. | Root apical, acropetal |
| 3. | Shoot apical, acropetal |
| 4. | Intercalary, acropetal |
In the STPs, biological process of microbial degradation of organic matter involves
1 Utilisation of activated sludge as inoculant produced in the physical process
2 Mainly involves anaerobic breakdown of organic matter
3 Masses of unicellular bacteria entangled in filamentous bacterial forms represent flocs
4 BOD is greatly reduced by the microbial activity
Intercalated discs are types of
1. Adhering junction
2. Anchoring junction
3. Communication junction
4. Tight junction
Citrus differs from Opuntia in
1. Showing adventive embryony
2. Having zygotic embryos
3. Having modified axillary buds as thorns
4. Both 1 and 3
Sporophyte in bryophytes is
1. Parasitic on gametophyte
2. Formed by germination of meiospore
3. Specialized to produce gametes
4. Haploid phase
Choose the incorrectly matched pair:
1. Ulothrix – Zygote is single celled diploid generation
2. Spirogyra – Zygote is a resistant structure
3. Cycas – Coralloid roots
4. Fucus – Isogametes
Choose the incorrect match with respect to blood components
1. Leucocytes – 6000 - 8000 mm–3
2. Globulins – Involved in defence mechanisms
3. O– blood group – Antibodies A and B absent
4. Thrombokinase – Converts prothrombin into thrombin
Which of the following is least likely to be caused by reduced blood/oxygen supply to heart muscles?
1. Atherosclerosis
2. Myocardial ischaemia
3. Angina pectoris
4. Heart attack
Complete the analogy with respect to reabsorption
PCT : Glucose and amino acids :: DCT : _____
1. K+ and H+
2. H+ and
3. NaCl and
4. K+ and H2O
Cells of immune system that do not provide innate immunity are
1. T-lymphocytes
2. Neutrophils
3. NK cells
4. Macrophages
Which of the following function is not related to the organelles that are considered to be a part of endomembrane system?
| 1. | Detoxification of drugs |
| 2. | Providing precursors of enzyme for the formation of lysosomes |
| 3. | Formation of plasma membrane during cytokinesis |
| 4. | Synthesizing non-secretory proteins |
Match the following columns and select the correct option.
| Column-A | Column-B | ||
| (a) | Metaphase | (i) | Duplication of centrioles |
| (b) | G2 phase | (ii) | Synthesis of tubulin protein |
| (c) | S phase | (iii) | Congression of chromosomes |
| (d) | Anaphase | (iv) | Shortening of chromosomal fibres |
| 1. | (a) – iv, (b) – ii, (c) – i, (d) – iii |
| 2. | (a) – iv, (b) – i, (c) – ii, (d) – iii |
| 3. | (a) – iii, (b) – ii, (c) – i, (d) – iv |
| 4. | (a) – iii, (b) – i, (c) – ii, (d) – iv |
Metaphase I is different from mitotic metaphase as in the later case
| 1. | Two metaphasic plates are formed |
| 2. | Tetrads are arranged at the equator |
| 3. | Single metaphasic plate is formed |
| 4. | Homologous chromosomes get separated from each other |
The product(s) of photophosphorylation that occurs mostly in the stroma lamellae membrane of green plants is/are
1. ATP and NADPH
2. ATP and
3. NADPH and
4. ATP only
Select the incorrect match from the following
| 1. | One Calvin cycle – Requires 3 ATP and 2 NADPH |
| 2. | C4 plant – Shows slower process of carbon fixation |
| 3. | C3 plants – 20-25°C is optimum for photosynthesis |
| 4. | Hatch and Slack – Primary CO2 acceptor pathway is phosphoenolpyruvate |
Match the following columns and select the correct option
| Column-I | Column-II | ||
| (a) | Abscisic acid | (i) | Sex expression |
| (b) | Ethylene | (ii) | Closure of stomata |
| (c) | Cytokinin | (iii) | Stem elongation in rosette plants |
| (d) | Gibberellin | (iv) | Counteracts the apical dominance |
| 1. | a→(ii), b→(iii), c→(iv), d→(i) |
| 2. | a→(iv), b→(ii), c→(i), d→(iii) |
| 3. | a→(ii), b→(iv), c→(iii), d→(i) |
| 4. | a→(ii), b→(i), c→(iv), d→(iii) |
Trunbull's blue is :
1. ferricyanide
2. ferrous ferricyanide
3. ferrous cyanide
4. ferri ferrocyanide
How many structural isomers of primary alcohols can be formed with the given molecular formula \(\text{C}_5\text{H}_{11}\text{OH}\)?
| 1. | 5 | 2. | 4 |
| 3. | 2 | 4. | 3 |
Phenyl ethyl ether with concentrated hydrobromic acid on boiling yields:-
1. Phenol and ethyl bromide
2. Bromobenzene and ethanol
3. Phenol and ethane
4. Bromobenzene and ethane
Phenol on treatment with dil. HNO3 at room temperature gives-
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | All of the above |
Common oxidizing agents used in organic chemistry are;
1. Fenton's reagent
2. Osmium tetroxide
3. Acidified KMnO4
4. All are correct
The rate constant for a reaction of zero-order in A is 0.0030 mol L-1 s-1. How long will it take for the initial concentration of A to fall from 0.10 M to 0.075 M?
1. 8.3 sec
2. 0.83 sec
3. 83 sec
4. 10.3 sec
A compound forms hcp structure. The total number of voids in 0.5 mol will be-
1. 9.0331023
2. 6.0111023
3. 5.0231023
4. 7.0331023
In piperidine "N" atom has hybridization:
1. sp
2.
3.
4.
In a reaction, the rate = k[A]1[B]-2/3 the order of the reaction is-
1. 1/3
2. 2
3. -1/3
4. Zero
Given cell:
Pt(s)|H2(g)(P = 1 atm)| CH3COOH(0.1 M), HCl(0.1 M) || KCl(aq)|Hg2Cl2(s)|Hg
EMF of the cell is found to be 0.045 V at 298 K and the temperature coefficient is 3.4×10-4 V K-1. The cell entropy change of the following cell is :
[Given Ka CH3COOH = 10-5 M]
1. 70.8 J K−1 mol−1
2. 65.2 J K−1 mol−1
3. 79.2 J K−1 mol−1
4. 83.5 J K−1 mol−1
Para- hydrogen at room temp is:
1. less stable than ortho- hydrogen
2. more stable than ortho- hydrogen
3. as stable as ortho- hydrogen
4. none of these
At 298 K, assuming ideal behaviour, the average kinetic energy of a deuterium molecule is -
1. Two times that of a hydrogen molecule
2. Four times that of a hydrogen molecule
3. Half of that of a hydrogen molecule
4. Same as that of a hydrogen molecule
At room temperature, the average speed of Helium is higher than that of Oxygen by a factor of -
1.
2.
3. 8
4. 6
The final product Y in the below mentioned sequence is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The number of d-electrons in is not equal to
that of the
1. p-electrons in Ne
2. s-electrons in Mg
3. d-electrons in Co+3
4. p-electrons in
The charge on the electron and proton are reduced to of their original values.
Let the present value of the Rydberg constant of H-atom be R, then the new value of the Rydberg constant will be
A compound is made by mixing cobalt (III) nitrite and potassium nitrite solution in a ratio of 1 : 3. The aqueous solution of the compound showed 4 particles per molecule whereas molar conductivity reveals the presence of six electrical charges. The formula of the compound is:
1. Co(NO2)3 · 2KNO2
2. Co(NO2)3 · 3 KNO2
3. K3[Co(NO2)6]
4. K[Co(NO2)4]
The total number of 3rd period elements with more than one electron in a 3d orbital is:
| 1. | 9 | 2. | 11 |
| 3. | 0 | 4. | 8 |
The group of metals having the positive value of electron gain enthalpy is:
| 1. | Cr, Fe | 2. | Mn, Zn |
| 3. | Fe, Zn | 4. | Cr, Mn |
Percentage ionic character, if electronegativity value of X=2.1 & Y=3.0, is :-
| 1. | 20 | 2. | 30 |
| 3. | 17 | 4. | 23 |
The correctly matched option is:-
1. : Order of atomic radius
2. O > C > B > N : Order of ionisation energy
3. : Increasing order of Zeff
4. O < N < F < Ne : Order of electron affinity
For a reaction A → B, the Arrhenius equation is given as \(log_{e}k \ = \ 4 \ - \ \frac{1000}{T}\) the activation energy in J/mol for the given reaction will be:
1. 8314
2. 2000
3. 2814
4. 3412
ln a closed vessel, 50 ml of completely reacts with 200 ml of C2 according to the following equation:
What will be the composition of the gaseous mixture in the system?
1. 100 ml
2. 25 ml
3. 75 ml
4. 10 ml
The concentration of ion in a solution containing 0.1 M HCN and 0.2 M NaCN will be:
( for HCN = )
1. 3.1
2.
3.
4.
Which of the following alkenes will yield only ketones when treated with ozone?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
In alkaline medium oxidises to and it self gets reduces to . How many moles of are oxidised by 1 mol of ?
1. 1.5
2. 1
3. 2.5
4. 3.5
Consider the following reaction:-
A and B are isomeric compounds. Additionally, A is a monochloro derivative. These two can be differentiated easily by using:
1. AgN(aq)
2.
3. Conc.HN/
4. NaCl(aq)
The incorrect statement among the following is :
1. The atomic radius of Na is greater than that of Mg.
2. Density of K is more than Na.
3. Melting point & boiling point of Ca are greater than those of Mg.
4. Mg does not react with cold water.
When 4 g of iron is burnt to ferric oxide at a constant pressure, 29.28 kJ of heat is evolved.
The enthalpy of formation of ferric oxide will be-
(At. mass of Fe = 56) ?
1. 81.98 kJ
2. 819.8 kJ
3. 40.99 kJ
4. +819.8 kJ
Which one of the following pair of solution can we expect to be isotonic at the same temperature?
1. 0.1 M urea and 0.1 M NaCl
2. 0.1 M urea and 0.2 M MgCl
3. 0.1 M NaCl and 0.1 M NaSO
4. 0.1 M Ca(NO) and 0.1 M NaSO
The major product (P) in the following reaction is:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
The amount of charge required for the oxidation of 1 mol of H2O2 to O2 is:
1. 9.65×104 C
2. 93000 C
3. 1.93×105 C
3. 19.3 × 102 C
The correct order of tendency to get oxidize is
1.
2.
3.
4.
Product is:-
1.
2.
3.
4.
The three lanthanides that show a +2 oxidation state are:
1. Sm, Tb, Gd
2. Sm, Eu, Yb
3. La, Gd, Lu
4. Yb, Pm, Sm
The IUPAC name of the give compounds
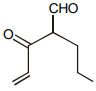
1. 3 – keto – 2 – methyl – pent – 4 – enal
2. 2 – keto – 2 – ethyl but – 2 – enal
3. 3 – keto – 2 – propyl pent – 4 – enal
4. 2 – keto – 2 – propyl pent – 4 – enal
The compound, whose stereochemical formula is shown below, exhibits A-geometrical isomers and B-optical isomers:
The values of A and B are:
1. 4 and 4
2. 4 and 2
3. 2 and 4
4. 2 and 2
The chiral compound/s among the following options is/are:
I.
II.
III.
IV.
1. II and IV
2. II and III
3. II, III and I
4. Only II
The self-ionization constant for pure formic acid, K = [\(H C O O H_{2}^{+} \left]\right. \left[\right. H C O O^{-} \left]\right.\) ] has been estimated as \(\left(10\right)^{- 4}\) at room temperature. The percentage of formic acid molecules in pure formic acid that are converted to formate ions is
\(\left(\right. Given : d_{HCOOH} = 1 . 22\ g / cc \left.\right)\)
1. 0.0185%
2. 0.0073%
3. 0.074%
4. 0.037%
The azeotropic mixture of water and ethanol boils at 78.15 oC. When this mixture is distilled, it is possible to obtain-
1. Pure H2O
2. Pure C2H5OH
3. Pure H2O as well as pure C2HOH
4. Neither H2O nor C2H5OH in their pure state
The net work done for an ideal gas is given as 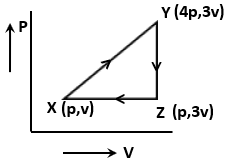
1. 3PV
2. 3PV
3. PV
4. Zero
Aluminium reacts with concentrated HCl and concentrated NaOH to liberate which of the following gases respectively-
| 1. | \(\mathrm{H}_2 ~\text{and} ~\mathrm{H}_2 \) | 2. | \(\mathrm{O}_2 ~\text{and} ~\mathrm{O}_2 \) |
| 3. | \(\mathrm{O}_2 ~\text{and} ~\mathrm{H}_2 \) | 4. | \(\mathrm{H}_2 ~\text{and} ~\mathrm{O}_2 \) |
The specie with the shortest bond length is:
| 1. | \(O^+_2\) | 2. | \(O^-_2\) |
| 3. | \(O^{2-}_2\) | 4. | \(O^{2+}_2\) |
The oxidation state of sodium in sodium amalgam is -
1. -1
2. +1
3. +2
4. 0
Lactic acid on oxidation with alkaline potassium permanganate gives:
1. Propionic acid
2. Cinnamic acid
3. Pyruvic acid
4. Tartaric acid
\(ABCA\) is a cyclic process. Its \(P\text-V\) graph would be:

| 1. |  |
2. |  |
| 3. |  |
4. |  |
A voltmeter of resistance \(660~\Omega\) reads the voltage of a very old cell to be \(1.32\) V while a potentiometer reads its voltage to be \(1.44\) V. The internal resistance of the cell is:
1. \(30~\Omega\)
2. \(60~\Omega\)
3. \(6~\Omega\)
4. \(0.6~\Omega\)
| 1. | \(\frac{GMm}{12R} \) | 2. | \(\frac{GMm}{R} \) |
| 3. | \(\frac{GMm}{8 R} \) | 4. | \(\frac{GMm}{2R}\) |
Two concentric conducting spherical shells carry charge \(Q\) each. The inner shell is earthed. The charge that flows into the earth is:

| 1. | \(Q\) | 2. | \(\frac{3Q}{2}\) |
| 3. | \(\frac{-Q}{2}\) | 4. | \(\frac{-3Q}{2}\) |
A solid sphere with a velocity (of the centre of mass) v and angular velocity is gently placed on a rough horizontal surface. The frictional force on the sphere:
1. must be forward (in direction of v)
2. must be backward (opposite to v)
3. cannot be zero
4. None of these
| 1. | \(1.0 \times 10^6 ~\text{V/m}\) | 2. | \(1.0 \times 10^5 ~\text{V/m}\) |
| 3. | \(2.0 \times 10^5 ~\text{V/m}\) | 4. | \(2.0 \times 10^6 ~\text{V/m}\) |
A body of density \(0.7\) g/cm3 floats on a lake of water. The fraction of the body that is outside water is:
1. \(30 \text{%}\)
2. \(70 \text{%}\)
3. \(25 \text{%}\)
4. \(50 \text{%}\)
A square loop of side 'a' hangs from an insulating hanger of spring balance. The magnetic field of strength B occurs only at the lower edge of loop which carries a current I. Find the change in the reading of the spring balance if the direction of current is reversed :-
1. IaB
2. 2IaB
3.
4.
A straight solenoid has \(50\) turns per cm in primary coil and \(200\) turns in the secondary coil. The area of cross-section of the solenoid is \(4\) cm2. Calculate the mutual inductance.
1. \(5.0~\text{H}\)
2. \(5.0\times 10^{-4}~\text{H}\)
3. \(2.5~\text{H}\)
4. \(2.5\times 10^{-4}~\text{H}\)
In an astronomical telescope, the focal length of the objective lens is \(100\) cm, and of eyepiece is \(2\) cm. The magnifying power of the telescope for the normal eye is:
1. \(50\)
2. \(10\)
3. \(100\)
4. \(\frac{1}{50}\)
In a transistor circuit shown here, the base current is \(35~\mu \)A . The value of the resistor\(R_{b}\) is:
(Given \(V_{BE}=4.5\) V)
1. \(128.5~\mathrm{k\Omega}\)
2. \(257~~\mathrm{k\Omega}\)
3. \(5~~\mathrm{k\Omega}\)
4. \(2.5~~\mathrm{k\Omega}\)
What is the maximum value of \(5\sin\theta-12\cos\theta\)?
1. \(12\)
2. \(17\)
3. \(7\)
4. \(13\)
In a heating arrangement, an alternating current having a peak value of 28 A is used. To produce the same heat energy if the direct current is used, its magnitude must be:-
1. about 14 A
2. about 28 A
3. about 20 A
4. cannot say
The displacement-time graph of a particle executing SHM is shown in the figure. Its displacement equation is: (Time period = \(2~\text{s}\))

1. \(x=10\sin \left(\pi t +\frac{\pi}{6}\right)\)
2. \(x=10\sin (\pi t )\)
3. \(x=10\cos (\pi t )\)
4. \(x=5\sin \left(\pi t +\frac{\pi}{6}\right)\)

| 1. | \(A\) | 2. | \(B\) |
| 3. | \(C\) | 4. | None of these |
If a particle has negative velocity and negative acceleration, its speed:
1. increases
2. decreases
3. remains the same
4. zero
Two objects are projected (with the same speed) at angles \(30^{\circ}\) and \(60^{\circ}\) respectively with respect to the horizontal direction. The range of two objects are denoted as \(R_{30^\circ}\) and \(R_{60^\circ}\) . Choose the correct relation from the following:
1. \(R_{30^\circ}=R_{60^\circ} \)
2. \(R_{30^\circ}=4R_{60^\circ} \)
3. \(R_{30^\circ}=\dfrac {R_{60^\circ}}{2} \)
4. \(R_{30^\circ}=2R_{60^\circ} \)
Two masses, \(m_1\) and \(m_2\) are experiencing the same force where \(m_1<m_2\). The ratio of their acceleration \(\frac{a_{1}}{a_{2}}\) is:
1. \(1\)
2. less than \(1\)
3. greater than \(1\)
4. all the three cases
A ball of mass \(1~\text{kg}\) and another of mass \(2~\text{kg}\) are dropped from a tall building whose height is \(80~\text m.\) After, a fall of \(40~\text{m}\) each towards Earth, their respective kinetic energies will be in the ratio of:
1. \(\sqrt{2}:1\)
2. \(1:\sqrt{2}\)
3. \(2:1\)
4. \(1:2\)
What is the minimum velocity with
which a body of mass m must enter a
vertical loop of radius R so that it can
complete the loop?
A particle is moving with a constant velocity along a line parallel to the positive x-axis. The magnitude of its angular momentum with respect to the origin is:
| 1. | zero |
| 2. | increasing with \(x\) |
| 3. | decreasing with \(x\) |
| 4. | remaining constant |
A rigid body rotates with an angular momentum of \(L.\) If its kinetic energy is halved, the angular momentum becomes:
1. \(L\)
2. \(L/2\)
3. \(2L\)
4. \(L/\)
If a wire is stretched to double its original length, then the strain in the wire is:
1. \(1\)
2. \(2\)
3. \(3\)
4. \(4\)
In a horizontal pipe of a non-uniform cross-section, water flows with a velocity of \(1~\text{ms}^{-1}\) at a point where the diameter of the pipe is \(20 ~\text{cm}.\) The velocity of water \((\text{ms}^{-1})\) at a point where the diameter of the pipe is \(5~\text{cm}\) is:
1. \(8\)
2. \(16\)
3. \(24\)
4. \(32\)
What is the graph between volume and temperature in Charle's law?
1. An ellipse
2. A circle
3. A straight line
4. A parabola
When a uniform rod is heated, which of its following properties will increase as a result of it?
1. mass
2. weight
3. center of mass
4. moment of inertia
When food is cooked in a vessel with the lid tightly closed, steam is produced inside as the temperature increases. After some time, the pressure from the steam lifts or pushes the lid outward. Considering the steam as the thermodynamic system, which of the following statements correctly describes the signs of heat exchange \(Q\) and work done \(W\) by the system during this process?
1. \(Q>0, W>0\)
2. \(Q<0, W>O\)
3. \(Q>0, W<0\)
4. \(Q<0, W<O\)
| 1. | \(15~\text{s}\) | 2. | \(6~\text{s}\) |
| 3. | \(12~\text{s}\) | 4. | \(9~\text{s}\) |
Two identical point charges of magnitude \(–q\) are fixed as shown in the figure below. A third charge \(+q \) is placed midway between the two charges at the point \(P.\) Suppose this charge \(+q \) is displaced a small distance from the point \(P\) in the directions indicated by the arrows. In which direction(s) will \(+q \) be stable with respect to the displacement?

1. \(A_1\) and \(A_2\)
2. \(B_1\) and \(B_2\)
3. Both directions
4. None of the above
| 1. | \(10\) V | 2. | \(-20\) V |
| 3. | \(+20\) V | 4. | \(-10\) V |
A wire connected to a power supply of \(230~\text{V}\) has power dissipation \(P_1.\) Suppose the wire is cut into two equal pieces and connected in parallel to the same power supply. In this case power dissipation is \(P_2.\) The ratio \(\frac{P_2}{P_1}\) is
1. 1
2. 2
3. 3
4. 4
A particle having mass \(m\) and charge \(q\) is accelerated through a potential difference \(V. \) Find the force experienced when it is kept under perpendicular magnetic field \(\vec B.\)
| 1. | \(\sqrt{\dfrac{2q^3BV}{m}} \) | 2. | \(\sqrt{\dfrac{q^3B^2V}{2m}} \) |
| 3. | \(\sqrt{\dfrac{2q^3B^2V}{m}} \) | 4. | \(\sqrt{\dfrac{2q^3BV}{m^3}} \) |
A bar magnet of length \(l\) and magnetic moment \(p_{m}\) is bent in the form of an arc as shown in the figure below. The new magnetic dipole moment will be:

| 1. | \(p_{m}\) | 2. | \(\dfrac{3}{\pi }p_{m}\) |
| 3. | \(\dfrac{2}{\pi }p_{m}\) | 4. | \(\dfrac{1}{2 }p_{m}\) |
An electron moves on a straight-line path \(XY\) as shown in the figure. The coil \(abcd\) is adjacent to the path of the electron. What will be the direction of current, if any, induced in the coil?

| 1. | The current will reverse its direction as the electron goes past the coil |
| 2. | No current will be induced |
| 3. | \(abcd\) |
| 4. | \(adcb\) |
Consider an oscillator which has a charged particle oscillating about its mean position with a frequency of \(300\) MHz. The wavelength of electromagnetic waves produced by this oscillator would be:
1. \(1\) m
2. \(10\) m
3. \(100\) m
4. \(1000\) m
The wavelength λ of an electron and
λ of a photon of same energy E are
related by
Atomic number of H-like atom with ionization potential \(122.4~\text{V}\) for \(n=1\) is:
1. \(1\)
2. \(2\)
3. \(3\)
4. \(4\)
Two capacitors of capacitance \(2~\mu\text F\) and \(4~\mu\text F\) are connected in a circuit as shown in the figure. The potential difference \((V_B ~– V_A)\) is:

1. \(– 8~\text V\)
2. \(8~\text V\)
3. \(12~\text V\)
4. \(– 12~\text V\)
A man with hypermetropia cannot see objects closer than a distance of \(40~\text{cm}\) from the eye. The power of the lens required so that he can see objects at \(25~\text{cm}\) from the eye is:
1. \(+4.5~\text{D}\)
2. \(+4.0~\text{D}\)
3. \(+1.5~\text{D}\)
4. \(+3.0~\text{D}\)
A particle starts from rest with uniform acceleration and its velocity after \(n\) s is \(v.\) The displacement of the particle in the last \(2\) s is.
1.
2.
3.
4.
If \(x=10.0\pm0.1\) and \(y=10\pm0.1\), then \(2x-2y\) with consideration of significant figures is equal to:
1. zero
2. \(0.0\pm0.1\)
3. \(0.0\pm0.2\)
4. \(0.0\pm0.4\)
A rod is standing vertically on a smooth surface, if the rod is slightly disturbed and it falls toward the right, then:

| 1. | The center of mass moves vertically downward |
| 2. | The center of mass moves toward the right |
| 3. | The center of mass moves toward the left |
| 4. | The center of mass does not move |
One mole of an ideal gas at an initial temperature of \(T\) K does \(15R\) joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is \(\dfrac{5}{3},\) the final temperature of the gas will be:
1. \((T+2.4)\) K
2. \((T-10)\) K
3. \((T-4)\) K
4. \((T+10)\) K