Convert the temperature of C to the kelvin scale.
Convert the temperature of 450 K to the Celsius scale.
Which of the following are matter?
Chair, air, love, smell, hate, almonds, thought, cold-drink, the smell of perfume.
Give reasons for the following observation. The smell of hot sizzling food reaches you several meters away. but to get the smell of cold food, you have to go close.
A diver is able to cut through water in a swimming pool. Which property of matter does this observation show?
What are the characteristics of the particles of matter?
The mass per unit volume of a substance is called density (Density = Mass/Volume).
Arrange the following in the order of increasing density.
Air, exhaust from chimneys, honey, water,
chalk, cotton, and iron.
Comment upon the following.
Rigidity, compressibility, fluidity, filling a gas container, shape, kinetic energy, and density.
Give reasons.
(i) A gas fills completely the vessel in which it is kept.
(ii) A gas exerts pressure on the walls of the container.
(iii) A wooden table should be called a solid.
(iv) We can easily move our hands in the air but to do the same through a solid block of wood, we need a karate expert.
Liquids generally have lower density as compared to solids. But you must have observed that ice floats on water. Find out why?
Convert the following temperatures to the Celsius scale.
(i) 300K
(ii) 573K
What is the physical state of water at
For any substance, why does the temperature remain constant during the change of state?
Suggest a method to liquefy atmospheric gases.
Why does a desert cooler cool better on a dry day?
How does the water kept in an earthen pot (matka) become cool during summer?
Why does our palm feel cold when we some acetone or petrol or perfume on it?
Why are we able to sip hot tea or milk from a saucer rather than a cup?
What type of clothes should we wear in summer?
Convert the following temperatures to the Celsius scale.
(i) 293 K
(ii) 470 K
Converting the following temperatures to the Kelvin scale.
Give a reason for the following observations:
(i) Naphthalene balls disappear with time without leaving any solid.
(ii) We can get the smell of perfume sitting several metres away.
Arrange the following substances in increasing order of forces of attraction between the particles - water, sugar, oxygen.
what is the physical state of water at
Give two reasons to justify.
(i) Water at room temperature is a liquid.
(ii) An iron almirah solid at room temperature.
Why is ice at 273 K more effective in cooling than water at the same temperature?
What produces more severe burns, boiling water or steam?
Name, A, B, C, D, E, and F in the following diagram showing a change in its state.

A diver is able to cut through water in a swimming pool. The property shown by the matter is
1. the particles are of very small size
2. the matter have space between them
3. the particles are in solid-state
4. the particles are running here and there, have no space between them
Which of the following is found in the solid state at room temperature?
1. Stone
2. Sand
3. Mercury
4. Both (a) and (b)
The similarity between a liquid and a gas is
1. both do not show the definite shape
2. both have a definite volume
3. both have same boiling point
4. both have the same nature
The property to flow is unique to fluids. Which one of the following statements is correct?
1. Only gases behave like fluids
2. Gases and solids behave like fluids
3. Gases and liquids behave like fluids
4. Only liquids are fluids
Choose the correct statement Of the following.
1. Conversion of solid into vapours without passing through the liquid state is called vaporisation.
2. Conversion Of vapours into solid without passing through the liquid state is called sublimation.
3. Conversion of vapours into solid without passing through the liquid state is called freezing.
4. Conversion of solid into liquid is called sublimation.
The boiling points of diethyl ether, acetone, and n-butyl alcohol are , and respectively. Which one of the following correctly represents their boiling points in the kelvin scale?
1. 306 K, 329 K, 391 K
2. 308 K, 329 K, 292 K
3. 308 K, 329 K, 391 K
4. 329 K, 392 K, 308 K
Temperature C equals nearly to
1. 300 K
2. 373 K
3. 473 K
4. -73 K
Which condition out of the following will increase the evaporation of water?
1. Increase in temperature of water
2. Decrease in temperature of water
3. The less exposed surface area of water
4. Adding common salt to water
During summer, water kept in an earthen pot becomes cool because of the phenomenon of
1. diffusion
2. transpiration
3. osmosis
4. evaporation
In the following questions, a statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as
1. If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
2. If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
3. If Assertion is true but Reason is false.
4. If Assertion is false, Reason is true.
5. If both Assertion and Reason are false.
Assertion Smell of burning incense stick spreads all around due to the diffusion of its fumes into the air.
Reason Increased temperature results increased kinetic energy of the molecules.
In the following questions, a statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as
1. If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
2. If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
3. If Assertion is true but Reason is false.
4. If Assertion is false, Reason is true.
5. If both Assertion and Reason are false.
Assertion The smell of hot cooked food reaches us in seconds.
Reason Rate of diffusion of solids is greater than that of gases.
In the following questions, a statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as
1. If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
2. If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
3. If Assertion is true but Reason is false.
4. If Assertion is false, Reason is true.
5. If both Assertion and Reason are false.
Assertion Naphthalene does not leave a residue when kept open for some time.
Reason The conversion of gas directly into solid is called condensation.
In the following questions, a statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as
1. If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
2. If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
3. If Assertion is true but Reason is false.
4. If Assertion is false, Reason is true.
5. If both Assertion and Reason are false.
Assertion There is no change in the temperature of a substance when it undergoes a change of state though it is still being heated.
Reason The heat supplied is absorbed either as latent heat of fusion or as latent heat of vaporization.
In the following questions, a statement of Assertion (A) is followed by a statement of Reason (R). Mark the correct choice as
1. If both Assertion and Reason are true and Reason is the correct explanation of Assertion.
2. If both Assertion and Reason are true but Reason is not the correct explanation of Assertion.
3. If Assertion is true but Reason is false.
4. If Assertion is false, Reason is true.
5. If both Assertion and Reason are false.
Assertion During evaporation of liquids the temperature remains unaffected.
Reason Kinetic energy of the molecules directly proportional to temperature.
A substance has no mass. Can we consider it a matter?
A given substance X has a definite volume, but no definite shape and can diffuse easily. What is the physical state of a substance X?
Rubber band changes its shape. Is it solid?
Why do liquids take up the shape of the container in which they are kept?
What would be the effect of
(i) Temperature and
(ii) The density of liquids on the rate of diffusion of liquids?
Why do gases exert more pressure on the walls of the container than the solids?
Which characteristic of gas is used in supplying oxygen cylinders to hospitals?
Mention two ways to liquefy atmospheric gases.
Why is boiling called a bulk phenomenon?
Why the temperature remains constant during sublimation?
What is meant by the latent heat of vapourization?
Benzene is a liquid at 800C, liquid benzene is in equilibrium with its vapours. It is found that particles of benzene vapours are more energetic than particles of liquid benzene. Explain the observation.
What happens to the melting point of solids with the decrease in pressure?
Why should wet clothes be spread while drying?
What is the effect of the sprinkling of water on the roof or open ground on a hot sunny day?
(i) A sponge can be compressed, yet it is solid? Explain.
(ii) Name the state of matter that has minimum space between particles.
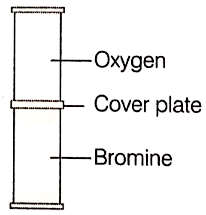
The cover plate is removed from the gas jars shown in the diagram, After several days, the color of the gas is the same in both jars. Why does this happen? Explain
Classify the following into osmosis/ diffusion.
(i) Swelling up of a raisin on keeping in water.
(ii) Spreading of the virus on sneezing.
(iii)Earthworm dying on coming in contact with common salt.
(iv) Shrinking of grapes kept in thick sugar syrup.
(v) Preserving pickles in salt.
(vi) Spreading of the smell of cake being baked throughout the house.
(vii) Aquatic animals use oxygen dissolved in water during respiration.
(i) Explain the interconversion of three states of matter in terms of the force of attraction and kinetic energy of the molecules.
(ii) Arrange the three states Of matter in the increasing order of rate of diffusion a particle motion.
How do you differentiate between solids, liquids and gases on the basis of their melting and boiling points?
(i) Dry ice is compressed under high pressure. What happens to it when the pressure is released?
(ii) Define
(a) Melting point
(b) Fusion.
What is dry ice? How is it formed?
Draw a well-labeled diagram showing sublimation of ammonium chloride.
From the graph below:
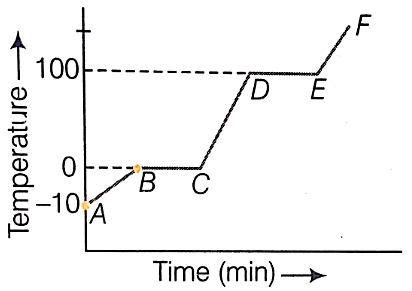
(i) Which region contains only solid?
(ii) Which region contains all liquids?
(iii) Which region shows the latent heat of vaporization?
Comment on the following statements:
(i) Evaporation produces cooling
(ii) Rate of evaporation of an aqueous solution decreases with increases in humidity
(iii) Sponge though compressible is solid.
(i) How will you show that the process of evaporation depends on the nature of the liquid?
(ii) Why a drop of Dettol is evenly distributed in a bucket of water without the need of stirring?
Look at the following figures and suggest in which of the vessels or D, the rate of evaporation will be the highest? Explain.

How will you change water from gaseous state to liquid state? Suggest a simple activity.
How does the change in temperature and humidity affect the rate of evaporation?
Which factors determine the state of a substance?
Convert 300C into Kelvin.
Water droplets are observed on the outer surface of a glass tumbler containing ice-cold water. Give reason.
It is a hot summer day. Priyanshi and Ali wearing cotton and nylon cloth respectively. Who do you think would be more comfortable and why?
What is the difference between solid, liquid, and gaseous states?
(i) Explain the term density. Arrange different states of matter in increasing order of density.
(ii) Explain how ice floats on water?
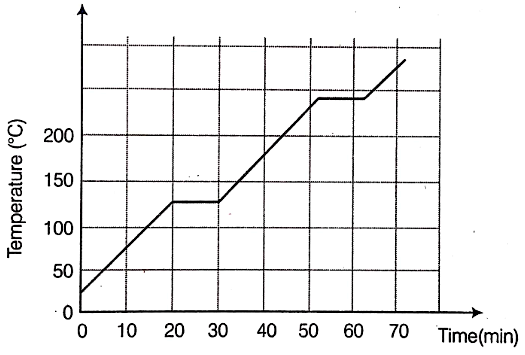
Benzoic acid is used as a food preservative. The given graph shows the heating curve for benzoic acid. Study the graph and answer the following questions:
(i) At what time does benzoic acid begin to
(a) melt?
(b) boil?
(ii) What is the melting point of benzoic acid?
(iii) What happens to the temperature while benzoic acid melts?
(iv) What is the physical state of benzoic acid during a time interval of 35—45 mins?
(i) What is the matter? Write two properties of solids and two properties of liquids.
(ii) Give reasons for the following:
(a) Ice at OOC appears colder in the mouth than water at OOC.
(b) Doctors advise putting the strips of wet cloth on the forehead of a person having a high temperature.
The temperature-time graph given below shows the heating for pure wax.
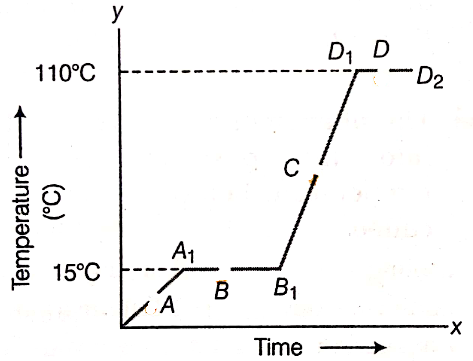
From the graph, answer the following:
(i) What is the physical state of the substance at the points A, B, C, and D?
(ii) What is the melting point of the wax?
(iii) What is its boiling point?
(iv) Which portions of the graph indicate that change of state is taking place?
(v) Name the terms used for heat absorbed during the change of states involved in the above processes.
Fill in the blanks.:-
(i) Evaporation of a liquid at room temperature leads to a _______________ effect.
(ii) At room temperature, the forces of attraction between the particles of solid substances are _______________ than those which exist in the gaseous state.
(iii) The arrangement of particles is less ordered in the _______________ state. However, there is no order in the _______________ state.
(iv) _______________ is the change of gaseous state directly to a solid state without going through the _______________ state.
(v) The phenomenon of change of a liquid into the gaseous state at any temperature below its boiling point is called ________________ .
(i) What temperature in the Kelvin scale is equal to
(ii) Describe an activity to show that the rate of evaporation increases with the surface area.
(iii) Describe the method of formation of plasma and Bose-Einstein condensation.