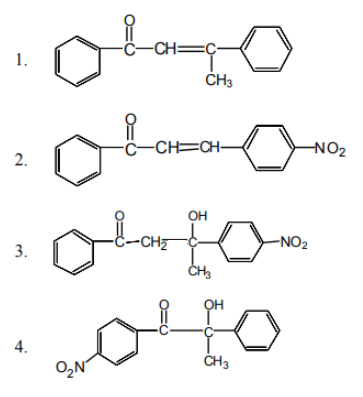
Arrange the following compounds in the order of
leaving group ability
1. I >II>III>IV
2. IV>III>I>II
3. III>II>I>IV
4. II>III>IV>I
On warming with I2 and aqueous NaOH, iodoform
and sodium succinate are formed. The formula of
the compound should be
1. CH3COCH2CH2CH3
2. CH3COC6H5
3. CH3COCH2CH2COCH3
4. none of these
An organic compound (A) (MF C4H10O) upon
dehydrogenation gives a compound (B) which forms
phenyl hydrazone with phenylhydrazine and both the
compounds (A) and (B) respond to iodoform test.
Hence, the compound (A) is
Identify ‘Y’ in
In a Cannizzaro’s reaction, the intermediate that will
be the best hydride donor is
The appropriate reagent for the transformation is
1. Zn (Hg)/HCl
2. NH2–NH2/OH–
3. Both 1. and 2.
4. None of the above
In the reaction
the oxidizing agent can be
1. alkaline KMnO4
2. acidified K2Cr2O7
3. benedict’s solution
4. All of the above
When propanoic acid is treated with aqueous sodium
bicarbonate, CO2 is liberated. The ‘C’ of CO2
comes from
1. carboxylic acid group
2. methylene group
3. bicarbonate
4. methyl group
When glycerol is reacted with oxalic acid at 260oC,
then the final product formed is
1. Formic acid
2. Allyl alcohol
3. Acrolein
4. Acetic Acid
Which carboxylic acid is least reactive towards
decarboxylation on heating?
The end product of the following reaction would be
I. R1 = H II R1 = CH3
R2 = CH3 R2 = -CH(CH3)2
III. R1 = CH3 IV R1 = CH3
R2 = CH3 R2 = C2H5
The rate of addition follows the order
1. I > II > III > IV
2. I > III > IV > II
3. I > III > II > IV
4. I > II > IV > III
The correct order of increasing reactivity towards
acid hydrolysis is
1. 1 < 2 < 3
2. 3 < 1 < 2
3. 1 < 3 < 2
4. 2 < 1 < 3
The product ‘A’ is
The major product formed would be