

The product X in the above reaction is -
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The reaction of toluene with Cl2 in the presence of FeCl3 gives 'X' and the reaction of toluene in the presence of light gives 'Y'. Hence, the products 'X' and 'Y' are:
1. X= benzal chloride, Y=o-chlorotoluene
2. X=m-chlorotoluene, Y=p-chlorotoluene
3. X=o and p-chlorotoluene, Y= trichloromethyl benzene
4. X= benzyl chloride, Y=m-chlorotoluene
Benzene reacts with n-propyl chloride in the presence of anhydrous AlCl3 to give
1. 3-propyl-1-chlorobenzene
2. n-propyl benzene
3. no reaction
4. iso-propyl benzene
Propene, CH3-CH=CH2 can be converted into 1-propanol by oxidation. Indicate which set of reagents amongst the following is ideal to effect the above conversion?
1. KMnO4(alkaline)
2. Osmium tetroxide (OsO4/CH2Cl2)
3. B2H6 and alk H2O2
4. O3/Zn

The reaction of toluene with in the presence of gives 'X' and reaction in presence of light gives 'Y'. Thus, 'X' and 'Y' are
1. X = benzal chloride, Y= o-chlorotoluene
2. X = m-chlorotoluene, Y= p-chlorotoluene
3. X = o and p-chlorotoluene, Y = trichloromethyl benzene
4. X = benzal chloride, Y = m-chlorotoluene
Ethylene forms ethylene chlorohydrin by the action of:
1. dry chlorine gas
2. dry hydrogen chlorine gas
3. solution of chlorine gas in water
4. dilute hydrochloric acid
A mixture of ethane, ethene and ethyne is passed through an ammoniacal solution. The gases that remain unreacted are-
1. Ethane and Ethene
2. Ethane and Ethyne
3. Ethene and Ethyne
4. Ethane only
What product(s) will be obtained from the ozonolysis of the compound shown below?

| 1. |  |
2. |  |
| 3. |  |
4. | None of the above |
An octane number 100 is given to:
1. n-hexane
2. Iso-octane
3. neo-pentane
4. neo-octane
Which of the following molecules is aromatic in character?

1. B
2. C
3. D
4. A
Xylene, on oxidation with acidic , gives:
| 1. | Phthalic acid | 2. | Isophthalic acid |
| 3. | Terephthalic acid | 4. | All of the above |
1-butyne on reaction with hot alkaline gives:
1.
2. +
3.
4. + HCOOH
What is the end product of the following sequences of operations?
\(\mathrm{CaC}_2 \xrightarrow{\mathrm{H}_2 \mathrm{O}} \mathrm{~A} \xrightarrow[\mathrm{Hg}^{2+}]{\mathrm{Dil} \cdot \mathrm{H}_2 \mathrm{SO}_4} \mathrm{~B} \underset{\mathrm{H}_2}{\stackrel{\mathrm{Ni}}{\rightarrow}} \mathrm{C}\)
1. Methyl alcohol
2. Acetaldehyde
3.
4.
is called:
1. Clemmensen reduction
2. Fisher-Spier reduction
3. Birch reduction
4. Arndt-Eistert reduction
The cylindrical shape of an alkyne is due to
1. three sigma bonds
2. three bonds
3. two sigma and one bonds
4. one sigma and two bonds
In Friedel-Craft's alkylation, besides the other reactants are
1.
2.
3.
4.

In the above reaction, X is:
1.
2.
3.
4.
The major product of the following reaction is:

1. Butene
2. But-2-ene
3. Butane
4. But-1-yne
Alkene-1 on hydroboration followed with action of gives:
1. alkanol-2
2. alkanol-1
3. alkanal
4. alkanone
A (Major)
The product "A" is -
1. 
3. 
Which of the provided alkenes exhibits the greatest stability?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
XY
Y is:
1. 
3. 4.
Consider the given reaction:
 Products formed
Products formed
| (A) |  |
(B) |  |
| (C) |  |
(D) |  |
1. (A) and (B) only 2. (A) and (C) only
3. (B) and (C) only 4. (A), (B), (C) and (D)
Observe the following sequence of reactions.

The product R is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
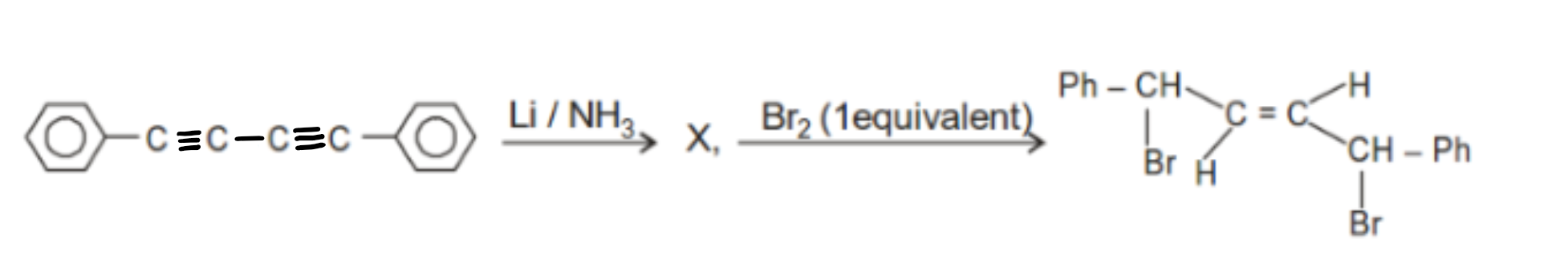
'X' is :
| (1) |  |
| (2) |  |
| (3) | Both |
| (4) | None of these |
Determine the double bond equivalent (degree of unsaturation) of the compound 'A':
(A)
Given that compound 'A' undergoes hydrogenation with 1 mole of H₂ in the presence of Pt, yielding the depicted product, what is the double bond equivalent of 'A'?
1. 1
2. 2
3. 3
4. 4
(I)
(II)
(III)
The correct order of reactivity of I,II and III towards addition reactions is:
(1) I>III>II
(2) I>II>III
(3) III>II>I
(4) III>I>II
On catalytic reduction by one mole H2/Pt ,how many alkenes will give n-butane?
(a)1
(b)2
(c)3
(d)4
Taking into account the stability of various carbocations and, as well as the rules governing mechanisms of carbocation rearrangements, which reaction is most likely to occur during the given reaction ?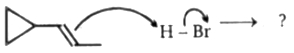
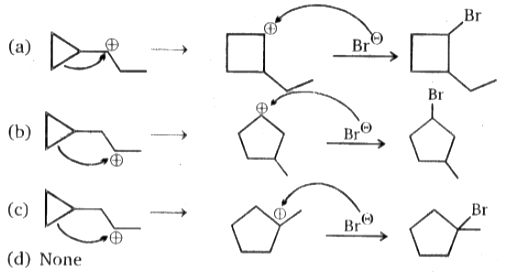
Consider the given reaction:

The product Y is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |