The figure shows a plot of photo current versus anode potential for a photo sensitive surface for three difference radiations. Which one of the following is a correct statement?
1. Curves a and b represent incident radiations of different frequencies and different intensities
2. Curves a and b represent incident radiations of same frequency but of different intensities
3. Curves b and c represent incident radiations of different frequencies and different intensities
4. Curves b and c represent incident radiations of same frequency having same intensity
The number of photoelectrons emitted for light of a frequency v (higher than the threshold frequency ) is proportional to
1.
2. threshold frequency
3. intensity of light
4. frequency of light (v)
| 1. | \(\frac{1}{8}\) | 2. | \(\frac{3}{8}\) |
| 3. | \(\frac{5}{8}\) | 4. | \(\frac{7}{8}\) |
The kinetic energy of electron and proton is J. Then the relation between their de-Broglie wavelengths is
(1)
(2)
(3)
(4)
The de-Broglie wavelength of a particle accelerated with 150 volt potential is m. If it is accelerated by 600 volts p.d., its wavelength will be
(1) 0.25 Å
(2) 0.5 Å
(3) 1.5 Å
(4) 2 Å
The de-Broglie wavelength associated with a hydrogen molecule moving with a thermal velocity of 3 km/s will be
(1) 1 Å
(2) 0.66 Å
(3) 6.6 Å
(4) 66 Å
When the momentum of a proton is changed by an amount , the corresponding change in the de-Broglie wavelength is found to be 0.25%. Then, the original momentum of the proton was
(a) (b) 100
(c) 400 (d) 4
The de-Broglie wavelength of a neutron at 27 is . What will be its wavelength at 927
(a) / 2 (b) / 3
(c) / 4 (d) / 9
An electron and proton have the same de-Broglie wavelength. Then the kinetic energy of the electron is
(1) Zero
(2) Infinity
(3) Equal to the kinetic energy of the proton
(4) Greater than the kinetic energy of the proton
For the moving ball of cricket, the correct statement about de-Broglie wavelength is:
1. It is not applicable for such big particle
2. \(\frac{h}{\sqrt{2mE}}\)
3. \(\sqrt{\frac{h}{2mE}}\)
4. \(\frac{h}{2mE}\)
The kinetic energy of an electron with de-Broglie wavelength of 0.3 nanometer is
(1) 0.168 eV
(2) 16.8 eV
(3) 1.68 eV
(4) 2.5 eV
The wavelength of de-Broglie wave is 2m, then its momentum is (h = J-s)
(a) kg-m/s (b) kg-m/s
(c) kg-m/s (d) kg-m/s
If the kinetic energy of a free electron doubles, its de-Broglie wavelength changes by the factor
(1)
(2)
(3)
(4) 2
The energy that should be added to an electron to reduce its de Broglie wavelength from one nm to 0.5 nm is
(1) Four times the initial energy
(2) Equal to the initial energy
(3) Twice the initial energy
(4) Thrice the initial energy
The wavelength of the matter wave is independent of
(1) Mass
(2) Velocity
(3) Momentum
(4) Charge
Kinetic energy with which the electrons are emitted from the metal surface due to photoelectric effect is
(1) Independent of the intensity of illumination
(2) Independent of the frequency of light
(3) Inversely proportional to the intensity of illumination
(4) Directly proportional to the intensity of illumination
The number of photo-electrons emitted per second from a metal surface increases when:
| 1. | The energy of incident photons increases. | 2. | The frequency of incident light increases. |
| 3. | The wavelength of the incident light increases. | 4. | The intensity of the incident light increases. |
The work function for tungsten and sodium are 4.5 eV and 2.3 eV respectively. If the threshold wavelength for sodium is 5460 Å, the value of for tungsten is
(1) 5893 Å
(2) 10683 Å
(3) 2791 Å
(4) 528 Å
Light of wavelength \(4000~\mathrm{\mathring{A}}\) is incident on a sodium surface for which the threshold wavelength of photoelectrons is \(5420~\mathrm{\mathring{A}}\). The work function of sodium is:
1. \(4.58\) eV
2. \(2.29\) eV
3. \(1.14\) eV
4. \(0.57\) eV
The stopping potential for photoelectrons:
| 1. | does not depend on the frequency of the incident light. |
| 2. | does not depend upon the nature of the cathode material. |
| 3. | depends on both the frequency of the incident light and the nature of the cathode material. |
| 4. | depends upon the intensity of the incident light. |
The maximum wavelength of radiation that can produce photoelectric effect in a certain metal is 200 nm. The maximum kinetic energy acquired by electron due to radiation of wavelength 100 nm will be
(1) 12.4 eV
(2) 6.2 eV
(3) 100 eV
(4) 200 eV
When the light source is kept 20 cm away from a photo cell, stopping potential 0.6 V is obtained. When source is kept 40 cm away, the stopping potential will be
1. 0.3 V 2. 0.6 V
3. 1.2 V 4. 2.4 V
| 1. | The stopping potential will decrease. |
| 2. | The stopping potential will increase. |
| 3. | The kinetic energy of emitted electrons will decrease. |
| 4. | The value of the work function will decrease. |
Which of the following is dependent on the intensity of incident radiation in a photoelectric experiment
(1) Work function of the surface
(2) Amount of photoelectric current
(3) Stopping potential
(4) Maximum kinetic energy of photoelectrons
Which of the following statements is correct
(1) The current in a photocell increases with increasing frequency of light
(2) The photocurrent is proportional to applied voltage
(3) The photocurrent increases with increasing intensity of light
(4) The stopping potential increases with increasing intensity of incident light
4 eV is the energy of the incident photon and the work function in 2eV. What is the stopping potential ?
(1) 2V
(2) 4V
(3) 6V
(4)
The magnitude of saturation photoelectric current depends upon
(1) Frequency
(2) Intensity
(3) Work function
(4) Stopping potential
A particle of mass M at rest decays into two particles of masses and , having non-zero velocities. The ratio of the de-Broglie wavelengths of the particles, is
(a) / (b) /
(c) 1.0 (d)
A photon and an electron have equal energy E, then is proportional to
(1)
(2)
(3) 1/E
(4) Does not depend upon E
A photon of Joules is absorbed by a material under special circumstances. The correct statement is:
1. Electrons of the atom of absorbed material will go the higher energy states
2. Electron and positron pair will be created
3. Only positron will be produced
4. Photoelectric effect will occur and electron will be produced
In a photoemissive cell with executing wavelength , the fastest electron has speed v. If the exciting wavelength is changed to 3/4, the speed of the fastest emitted electron will be
(a) (b)
(c) Less than (d) Greater than
The ratio of de-Broglie wavelengths of molecules of hydrogen and helium which are at temperature 27 and 127 respectively is
(1)
(2)
(3)
(4) 1
The ratio of de-Broglie wavelength of a -particle to that of a proton being subjected to the same magnetic field so that the radii of their path are equal to each other assuming the field induction vector is perpendicular to the velocity vectors of the -particle and the proton is
1. 1
2.
3.
4. 2
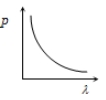
Which of the following figure represents the variation of particle momentum and the associated de-Broglie wavelength?
1. 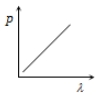
2. 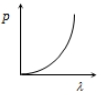
3. 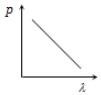
4. 
The stopping potential as a function of the frequency of the incident radiation is plotted for two different photoelectric surfaces \(A\) and \(B\). The graphs demonstrate that \(A\)'s work function is:

| 1. | Greater than that of \(B\). | 2. | Smaller than that of \(B\). |
| 3. | Equal to that of \(B\). | 4. | No inference can be drawn about their work functions from the given graphs. |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The value of stopping potential in the following diagram is given by:

| 1. | \(-4\) V | 2. | \(-3\) V |
| 3. | \(-2\) V | 4. | \(-1\) V |
In the following diagram if , then,
(1)
(2)
(3)
(4)
The curves (a), (b) (c) and (d) show the variation between the applied potential difference (V) and the photoelectric current (i), at two different intensities of light ( ). In which figure is the correct variation shown ?
The hybridization of carbon atoms in C-C single bond of HC≡C-CH=CH2 is-
1.
2.
3.
4.
Assertion : The energy (E) and momentum (p) of a photon are related by p=E/c .
Reason : The photon behaves like a particle.
Assertion : The de-Broglie wavelength of a molecule varies inversely as the square root of temperature.
Reason : The root mean square velocity of the molecule depends on the temperature.