Did Döbereine's triads also exist in the columns of Newlands' octaves?
Compare and find out.
What were the limitations of Döbereiner's classification?
What were the limitations of Newlands' law of octaves?
Use Mendeleev's periodic table to predict the formulae for the oxides of the following elements. K, C, Al, Si and Ba.
Besides gallium, which other elements have since been discovered that were left by Mendeleev in his periodic table (any two)?
What were the criteria used by Mendeleev in creating his periodic table?
Why do you think noble gases are placed in a separate group?
How could the modern periodic table remove various anomalies of Mendeleev's periodic table?
Name two elements you would expect to show chemical reactions similar to magnesium. What is the basis for your choice?
Name
(i) three elements that have a single electron in their outermost shells.
(ii) two elements that have two electrons in their outermost shells.
(iii) three elements with filled outermost shells.
(ii) Lithium, sodium and potassium are all metals that react with water to liberate hydrogen gas. Is there any similarity in the atoms of
these elements?
(ii) Helium is an unreactive gas and, neon is a gas of extremely low reactivity. What, if anything does their atoms have in common?
In the modern periodic table, which are the metals among the first ten elements?
By considering their position in the periodic table, which one of the following elements would you expect to have maximum metallic characteristic?
Ga, Ge, As, Se and Br
Which of the following statements is not a correct statement about the trends when going from left to right across the periods of periodic table?
1. The elements become less metallic in nature
2. The number of valence electrons increases
3. The atoms lose their electrons more easily
4. The oxides become more acidic
Element X forms a chloride with formula, XCl2, which is a solid with a high melting point. X would most likely to be in the same group of the periodic table as
1. Na
2. Mg
3. Al
4. Si
Which elements has
(a) two shells, both of which ere completely filled with electrons?
(b) the electronic configuration 2, 8, 2?
(c) a total of three shells, with four electrons in its valence shell?
(d) a total of two shells, with three electrons in its valence shell?
(e) twice as many electrons in its second shell as in its first shell?
(a) What property do all elements in the same column of the periodic table as boron have in common?
(b) What property do all elements in the same column of the periodic table as fluorine have in common?
An atom has electronic configuration 2, 8, 7.
(a) What is the atomic number of this element?
(b) To which of the following elements would it be chemically similar? (Atomic numbers are given in parentheses).
N (7), F (9), P (15) and Ar (18)
The position of three elements A, B and C in the periodic table are shown below:
|
Group 16 |
Group 17 |
|
A |
|
|
B |
C |
(a) State whether A is a metal or non-metal.
(b) State whether C is more reactive or less reactive than A.
(c) Will C be larger or smaller in size than B?
(d) Which type of ion-cation or anion, will be formed by A?
Nitrogen (atomic number = 7) and phosphorus (atomic number = 15) belong to group 15 of the periodic table. Write the electronic configuration of these two elements. Which will be more electronegative and why?
How does the electronic configuration of an atom relate to its position in the modem periodic table?
In the modern periodic table, calcium (atomic number = 20) is surrounded by elements with atomic numbers 12, 19, 21 and 38. Which of these have physical and chemical properties resembling calcium?
Compare and contrast the arrangement of elements in Mendeleev's periodic table and the modern periodic table.
Chlorine, bromine and iodine form a Döbereiner's triad. The atomic masses of chlorine and iodine are 35.5 and 126.9, respectively. Predict the atomic mass of bromine.
Up to which element, the law of octaves was found to be applicable?
A and B are the two elements having similar properties which obey Newlands' law of octaves. How many elements are there in between A and B?
Why are noble gases missing from Newland's octaves?
What is meant by periodicity in properties of elements with reference to periodic table?
Atomic number is considered to be a more appropriate parameter than atomic mass for classification of elements in a periodic table. Why?
Write the atomic numbers of two elements X and Y having electronic configuration 2, 8, 2 and 2, 8, 6 respectively.
Write the valency and usual number of valence electrons of group 18 of the periodic table.
Three elements B, Si and Ge are known by which names?
Where would you locate the element with electronic configuration 2, 8 in the modern periodic table?
Out of Na, Al, Si, P which element exhibits maximum number of valence electrons?
Out of the two elements X and Y which has bigger atomic radius?
(i) X has atomic number = 18 and atomic mass = 40.
(ii) Y has atomic number = 20 and atomic mass =40.
Write the correct increasing order of the atomic radii of O, F and N.
Out of two elements potassium and sodium, which one can lose electron easily? Give reason for your answer.
Account for the following: Elements of group 17 are monovalent.
Account for the following: Elements of group 18 are called zerovalent.
Which elements will form an acidic oxide?
The element with atomic number 14 is hard and forms acidic oxide and a covalent halide. To which of the categories does the element belong?
Elements A, B, C and D have atomic numbers 1, 8, 11 and 19 respectively. Choose the odd element and give reason for your answer.
Mention the type of compounds formed between group 1 and group 17 elements.
An element X is in the second period of group 16 of the periodic table. Is it metal or non-metal? Give reason for your answer.
An element X forms a chloride with formula XCl3. The element X would most likely be in the same group of the periodic table as is Na, Mg, Al or Ca.
The three elements A, B and C with similar properties have atomic masses X, Y and Z respectively. The mass of Y is approximately equal to the average mass of X and Z. What is such an arrangement of elements called as? Give one example of such a set of elements.
Can the following groups of elements be classified as Döbereiner's triads?
(i) Na, Si and Cl
(ii) Be, Mg and Ca
[Atomic mass of Be=9, Na=23, Mg=24, Si=28, Cl=35 and Ca=40]
Explain by giving reason.
In Mendeleev's periodic table, why does tellurium with atomic mass = 127.6 appear before iodine having atomic mass = 126.9?
In Mendeleev's periodic table, the elements were arranged in the increasing order of their atomic masses. However, cobalt with atomic mass of 58.93 amu was placed before nickel having an atomic mass of 58.71 amu. Give reason for the same.
Which group of elements could be placed in Mendeleev's periodic table without disturbing the original order? Give reason.
Atomic number of an element is 16. Write its electronic configuration. Find the number of valence electrons and its valency.
How were the positions of nickel and cobalt resolved in the modern periodic table?
State the reason for the following:
(i) The elements of the same group have similar chemical properties.
(ii) The elements of the same period have different properties.
Arrange the following elements in increasing order of their atomic radii.
(i) Li, Be, F and N (ii) Cl, At, Br and I
Compare the radii of two species X and Y. Give reasons for your answer.
(i) X has 12 protons and 12 electrons
(ii) Y has 12 protons and 10 electrons
The atomic radii of three elements A, B and C of the periodic table are 186 pm, 104 pm and 143 pm respectively. Giving a reason, arrange these elements in the increasing order of atomic numbers in the period.
State any two changes in the properties of group 1 elements of modern periodic table, if we move downwards.
Arrange the following elements in the increasing order of their metallic character Mg, Ca, K, Ge, Ga.
Identify and name the metals out of the following elements whose electronic configurations are given below.
(i) 2, 8, 2
(ii) 2, 8, 1
(iii) 2, 8, 7
(iv) 2, 1
Three elements X, Y, and Z belong to 17th group but to 2nd, 3rd and 4th period respectively. Number of valence electrons in Y is 7. Find the number of valence electrons in X and Z.
Fluorine, chlorine and bromine were placed in a group on the basis of their similar properties.
(i) What is the name of this group or family?
(ii) State two of their similar properties.
Neon and argon are unreactive gases.
(i) What do their atoms have in common?
(ii) Why are they non-reacting gases?
A metal M forms an oxide having the formula MO. It belongs to 2nd period in the modem periodic table. Write its atomic number and valency.
Two elements X and Y have atomic number 12 and 16 respectively. Write the electronic configuration of these elements. State the period of these elements. What type of bond will be formed if these two elements combine together?
If an element X is placed in group 14, what will be the formula and the nature of bonding of its chloride?
Write the formulae of chlorides of Eka-silicon and Eka-aluminium, the elements predicted by Mendeleev.
Name
(i) three elements having three electrons in their outermost shells.
(ii) three elements with completely filled outermost shells, having atomic number more than 25.
Write the electric configuration of the following ions.
(i) (ii) (iii) (iv)
(i) How does the tendency to gain electrons change as we go down a group? Give reason.
(ii) A part of the periodic table is given below. How does the valency vary, as we move vertically downward from Li to Fr? Give reason.
|
Li |
Be |
|
Na |
|
|
K |
|
|
Rb |
|
|
Cs |
|
|
Fr |
Ra |
The position of three elements X, Y and Z in the periodic table is shown below.
|
Group 1 |
Group 2 |
|
X |
|
|
Y |
Z |
(i) Which type of ion cation or anion, will be formed by element X?
(ii) Which element Y or Z, has the atom of comparatively larger size? Give reason in each case.
Give reason for the following:
(i) Lithium atom is smaller than potassium atom.
(ii) Chlorine (atomic number = 17) is more electronegative than sulphur (atomic number = 16).
Consider the given formulae of compounds KOH, NH3, HCl, Al2O3, Mg(OH)2. Choose the
(i) compound which consists a non-metal with oxidation state of —3.
(ii) compound with most electropositive element.
(iii) compound with a metal having valency 3.
Write the formula of the product formed when the element A (atomic number = 19) combines with the element B (atomic number = 17). Draw its electron dot structure. What is the nature of the bond formed?
Give an account of the process adopted by Mendelev for the classification of elements. How did he arrive at "periodic law"?
'Hydrogen occupies a unique position in modern periodic table', justify the statement.
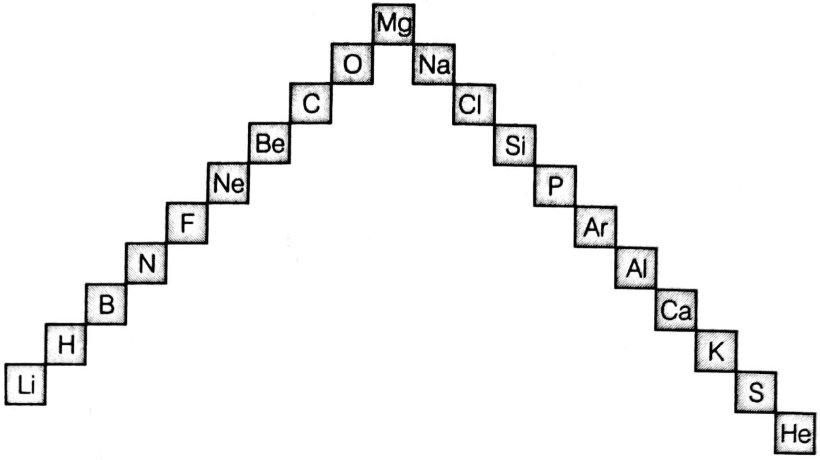
(i) In this ladder (Figure) symbols of elements are jumbled up. Rearrange these symbols of elements in the increasing order of their
atomic numbers in the periodic table.
(ii) Arrange them in the order of their group also.
(i) What term can be used for the elements separating metal from non-metals and why?
(ii) Give the names of the metalloids in the periodic table alongwith their atomic number.
(iii) In which groups of the periodic table are they located?
Taking the example of an element of number = 16, explain how the electronic configuration of the atom of an element relates to its position in the modern period, table and how valency of an element is calculated on the basis of its atomic number?
(i) Name of the element with atomic number 7.
(ii) To which group does it belong?
(iii) To which period does it belong?
(iv) Write its electronic configuration.
Atomic number of three elements X, Y and Z are given below:
Identify the group and period to which these elements belong.
Which of the following elements belong to
(i) same period? (ii) same group?
Explain giving reason.
(i) How does electronic configuration of an atom relate to its position in the modern periodic table?
(ii) Which element has a total of three shells with four electrons in its valence shell?
Explain why the size of the atom increases down the group of periodic table?
The elements Li, Na and K each having one valence electron, are in periods 2, 3 and 4 respectively of modem periodic table.
(i) In which group of the periodic table should they be?
(ii) Which one of them is least reactive?
(iii) Which one of them has the largest atomic radius?
Give reason to justify your answer in each case.
Atomic radii of the elements of the second period are given below:
(i) Arrange the elements in the decreasing order of their atomic radii.
(ii) Identify the element that respectively has
(a) largest radius
(b) smallest radius
(iii) Give reason for the variation in atomic radii.
From the part of a periodic table, answer the following questions:
(i) Atomic number of oxygen is 8. What would be the atomic number of fluorine?
(ii) Out of X and Q which element has larger atomic size? Give reason for your answer.
(iii) Out of Y and Z which element has smaller atomic size? Give reason for your answer.
The atomic number of nitrogen (N), oxygen (O) and fluorine (F) are 7, 8 and 9 respectively.
(i) What is the number of valence electrons in N and F?
(ii) Name the element having smallest and largest atomic radii of any of the above three elements. Give reason for your answer.
Consider the following elements,
Li, Cl, Br, Na, K and I
(i) Arrange the elements according to the groups to which they belong in the periodic table.
(ii) Which of the elements have been grouped together on the basis of their common properties?
The position of three element A, B and C in the periodic table is shown below:
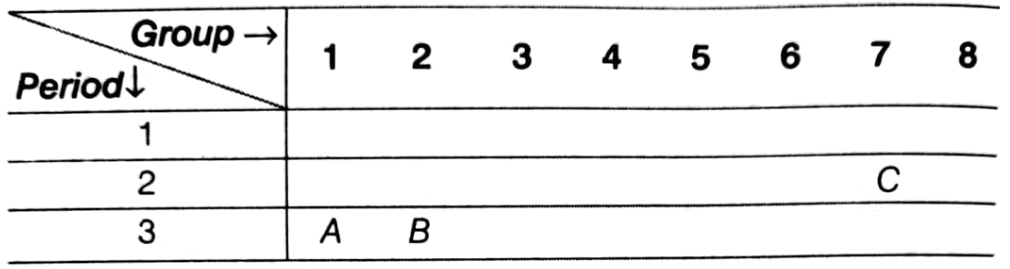
Giving reasons explain:
(i) Element A is a metal.
(ii) Element B has larger atomic size than the element C.
(iii) Element C has a valency of 1.
An element Y has a total of three shells, with six electrons in its valence shell.
(i) What will be the atomic number of this element?
(ii) In which period will you find this element?
(iii) Name another element which belongs to the same group as this element.
(i) An element X has both the shells K and L completely filled with electrons. The element has atomic number = 10. Identify
the element X.
(ii) In which group of the periodic table is this element placed?
(iii) Write its electronic configuration. What is the valency of the element?
An element X (atomic number = 17) reacts with an element Y (atomic number = 20) to form a divalent halide.
(i) What is the position of elements X and y in the periodic table?
(ii) What will be the nature of oxide of element Y? Identify the nature of bonding in the compound formed.
The following table shows the positions of four elements A, B, C and D in the periodic table.
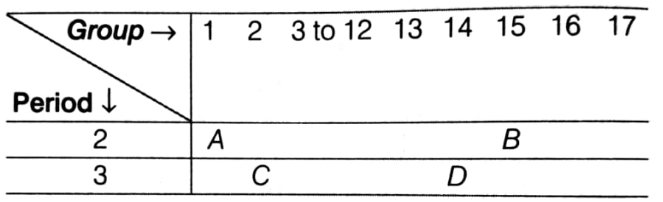
Using the above table answer the following questions.
(i) Which element will form only covalent compounds?
(ii) Which element is a metal with valency 2?
(iii) Out of C and D which one has bigger atomic radius and why?
A part of the periodic table has been shown below.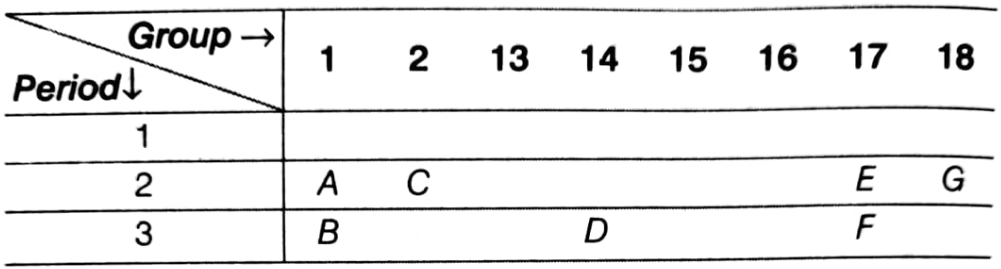
Answer the following questions on the basis of position of elements in the above table.
(i) Which element is a noble gas? Give reason.
(ii) Which element is most electronegative? Give reason.
(iii) Write the electronic configuration of (a) B and (b) E.
An element Y has a total of three shells with tour electrons in its valence shell. Another element X has twice as many electrons in its second shell as in its first shell.
(i) Identify the element Y and X.
(ii) Is there any similarity between the element Y and X? If so what?
An element has same number of electrons in I and IV shells and in the II and III shells.
(i) Write down the electronic configuration of the element.
(ii) Write the group number and period to which it belongs.
(iii) What is the valency of the element?
An element X has mass number = 35 and number of neutrons = 18. Write atomic number and electronic configuration of X. Also write group number, period number and valency of X.
Three elements X, Y and Z have atomic numbers 7, 8 and 9 respectively.
(i) State their positions (group number and period number both) in the modern periodic table.
(ii) Arrange these elements in the decreasing order of their atomic radii.
(iii) Write the formula of the compound formed when X combines with Z.
An element A reacts with oxygen to form A2O.
(i) State the number of electrons in the outermost orbit of A.
(ii) To which group of periodic table does A belong?
(iii) State whether A is a metal or non-metal.
Consider the part of periodic table given below and answer the following questions.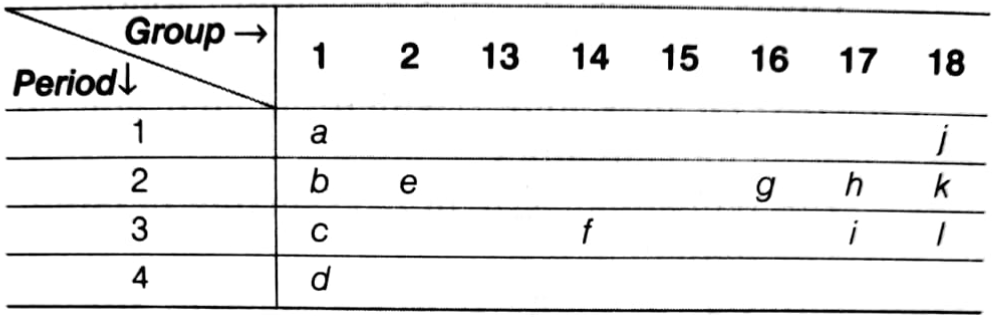
(i) The atom of which element is smaller in size e or h?
(ii) Which element is the most electropositive in nature?
(iii) Which element has only one proton in its atom?
(iv) What is the valency of g?
(v) How many valence electrons does g have?
(vi) Name the element which is a metalloid.
Atoms of seven elements A, B, C, D, E, F and G have different number of electronic shells but have the same number of electrons in their outermost shells. How will the following property vary as we move from A to G?
(i) Metallic character,
(ii) Atomic radii,
(iii) Valency.
The position of three elements X, Y and Z in the periodic table is given below.
Giving reason, answer the following questions.
(i) Out of Y and Z which element will be more metallic?
(ii) Will atomic size of Z be smaller or larger than that of X?
(iii) Out of Y and Z which element will be more electronegative?
Lithium is an alkali metal with atomic number = 3 and number of valence electron is 1. The formula of the hydride of lithium is LiH. Boron and carbon are placed in group 13 and 14 respectively with valence electrons 3 and 4. Write the formulae of the hydrides of boron and carbon giving reason for your answer.
An element X placed in 2nd group and 4th period of the periodic table burns in the presence of oxygen to form a basic oxide.
(i) Identify the element.
(ii) Write its electronic configuration.
(iii) Write a balanced equation for the reaction when this oxide is dissolved in water.
Two elements A and B belong to the 3rd period of modern periodic table and are in group 2 and 13 respectively. Compare their following characteristics in tabular form:
(i) Number of electrons in their atoms
(ii) Size of their atoms
(iii) Their tendencies to lose electrons
(iv) The formula of their oxides
(v) Their metallic character
(vi) The formula of their chlorides
An element X belongs to 3rd period and group 16 of the modern periodic table.
(i) Determine the number of valence electrons and the valency of X.
(ii) Molecular formula of the compound when X reacts with hydrogen and write its electron dot structure.
(iii) Name the element X and state whether it is metallic or non-metallic.
Identify, the elements with the following property and arrange them in increasing order of their reactivity.
(i) An element which is a soft and reactive metal.
(ii) The metal which is an important constituent of limestone.
(iii) The metal which exists in liquid state at room temperature.
Two elements P and Q belong to the 3rd period of the modern periodic table and are in group 1 and group 2, respectively.
Compare their following characteristics in tabular form:
(i) The number of electrons in their atoms
(ii) The sizes of their atoms
(iii) Their metallic character
(iv) Their tendencies to lose electrons
(v) The formula of their oxides
(vi) The formula of their chlorides
An element M with electronic configuration (2, 8, 2) combines separately with (NO3)-,(SO4)2- and (PO4)3- radicals.
Write the formula of the three compounds so formed. To which group and period of the modern periodic table does the elements M belong? Will M form covalent or ionic compounds? Give reason to justify your answer.
A salt when dissolved in water dissociates into cations and anions as follows:
If both the ions consist same number of electrons and the molecular weight of salt is 74.5. Then identify the position of A and B in the periodic table.
What is meant by group in the modem periodic table? How do the following changes occur on moving from top to bottom in a group?
(i) Number of valence electrons
(ii) Number of occupied shells
(iii) Size of atoms
(iv) Metallic character of element
(v) Effective nuclear charge experienced by valence electrons
The electronic configuration of three elements X, Y and Z are given below:
X = 2; Y = 2, 6; Z = 2, 8, 2
(i) Which element belongs to the second period?
(ii) Which element belongs to the eighteenth group?
(iii) Which element belongs to the second group?
(iv) What is the valency of Y?
(v) Y and Z are metal or non-metal.
Atomic number of a few elements are given below 10, 20, 7 and 14.
(i) Identify the elements.
(ii) Identity the group number of these elements In the periodic table.
(iii) Identify the periods of these elements in the periodic table.
(iv) What would be the electronic configuration for each of these elements?
(v) Determine the valency of these elements.
The element of a period of the periodic table are given below in order, from left to right with one of its element missing.
Li Be B C O F Ne
(i) To which period do these elements belong?
(ii) One element of this period is missing. Which is the missing element and where should it be placed?
(iii) Which one of these elements in the period shows the property of catenation?
(iv) Which one of the above elements belongs to the halogen series?
(v) Identify the noble gas.
Given below are few elements of the modern periodic table. Atomic number of the element is given in the parenthesis
A(4), B(9), C(14), D(19), E(20)
(i) Select the elements that has one electron in the outermost shell. Also write the electronic configuration of this element.
(ii) Which two elements amongst these, belong to the same group? Give reason for your answer.
(iii) Which two elements amongst these belong to the same period? Which one of the two has bigger atomic radius?
Mendeleev's predicted the existence of certain elements not known at that time and named two of them as Eka-silicon and Eka-aluminium.
(i) Name the elements which have taken the place of these elements.
(ii) Mention the group and the period of these elements in the modern periodic table.
(iii) Classify these elements as metals, non-metals or metalloids.
(iv) How many valence electrons are present in each one of them?
(i) Electropositive nature of the element(s) increases down the group and decreases across the period.
(ii) Electronegativity of the element decreases down the group and increases across the period.
(iii) Atomic size increases down the group and decreases across a period (left to right).
(iv) Metallic character increases down the group and decreases across a period.
On the basis of the above trends of the periodic table, answer the following about the element with atomic numbers 3 to 9.
(a) Name the most electropositive element among them.
(b) Name the most electronegative element.
(c) Name of the element with smallest atomic size.
(d) Name of the element which is a metalloid.
(e) Name of the element which shows maximum valency.
Consider two elements A (atomic number = 17) and B (atomic number = 19)
(1) Write the positions of these elements in the modem periodic table giving justification.
(2) Write the formula of the compound formed when A combines with B.
(3) Draw the electron dot structure of the compound and state the nature of the bond formed between the two elements.
With reference to the first three periods of the modern periodic table, answer the questions given below.
(i) Write the formula of the sulphate of the element with atomic number 13.
(ii) Consider the following elements:
Ca, O, Ar, S, Be and He
Which of the above elements would you expect to be
(a) of in group II A of the periodic table?
(b) in group VI A of the periodic tablet
(iii) How many electrons are present in the valence shell of the element with atomic number 18?
(iv) What is the electronic configuration of the element in the third period which gain one electron to change into an anion?
(v) What type of bonding will be present in the oxide of the element with atomic number 1?
(vi) Fill in the blanks.
The atomic size ........ as we move from left to right across the periods because the ....... increases but the .......remains the same.
(i) Using the part of the periodic table given below answer the questions that follows:
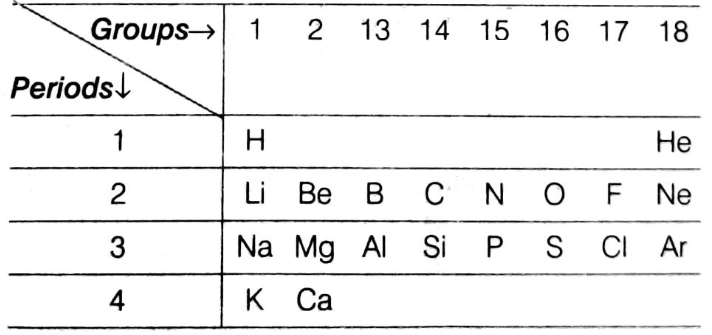
(a) Na has physical and chemical properties similar to which element(s).
(b) Write the electronic configuration of N and P. Which one of these will be more electronegative and why?
(c) State a chemical property common to fluorine and chlorine.
(ii) The neutral atom of an element E consists 12 electrons in its atoms.
(a) In which period and group placed?
(b) Name E.
(c) How many electrons it needs to lower gain to achieve noble gas configuration.
(d) What will be the nature of oxide (acidic/basic) of E? Justity your answer.
(e) Write the formulae of chloride of E.
An element is placed in 2nd group and 3rd period of the periodic table, burns in presence of oxygen to form a basic oxide.
(i) Identify the element.
(ii) Write the electronic configuration.
(iii) Write a balanced equation when it burns in the presence of air.
(iv) Write a balanced equation when this oxide is dissolved in water.
(v) Draw the electron dot structure for the formation of this oxide.
Ria and Rama are students of Class X. Ria is very much organised and maintained. The teachers love her She earns a great respect in the class whereas Rama is unorganised and always faces a lot of problems in handling life situations.
Read the given passage and answer the following questions.
(i) In your opinion how does organisation help in daily life?
(ii) How can you relate the above fact with the chapter classification of elements?
Or
How the classification of elements help as in studying them properly?
(iii) What is the associated value the learner gets from the given passage?
A mother always wants her child to drink milk. Milk is a boon for health. If one does not drink milk, he or she can face severe health problems.
Read the given passage and answer the following questions.
(i) Name one mineral (metal) present in major quantity in milk.
(ii) Write the chemical symbol, atomic number and valency of that nutrient (metal).
(iii) What value do you infer from the given text?
Class IXth and Xth students went on an education trip alongwith their respective class teachers in two separate buses.
Class teacher of class IXth asked the students to make a queue according to their height while boarding or deboarding the bus whereas class teacher of class Xth asked the students to make a queue for some purpose but according to their role numbers.
Read the given passage and answer the following questions.
(i) Name two scientists who arranged elements in periodic table with different criteria?
(ii) Which criteria of making queue was better in your view and why?
(iii) What value do you associate with the class teachers?
Jyoti, while on a bus for a trip during holidays, noticed that first few rows of the bus were marked for handicapped and old persons. She asked her father about this arrangement who explained her that it is for the ease of those people, so that they easily board and deboard the bus, being nearer to the door.
Read the given passage and answer the following questions.
(i) Name anyone more place with such arrangement.
(ii) Name the groups which consist elements which can easily accept or donate the electrons.
(ii) What value do you associate with Jyoti and her father?