What would be the electron dot structure of carbon dioxide which has molecular formula ?
What would be the electron dot structure of a molecule of sulphur which is made up of 8 atoms of sulphur?
How many structural isomers can you draw for pentane?
What are the two properties of carbon which lead to the huge number of carbon compounds, we see around us?
What will be the formula and electron dot structure of cyclopentane?
Draw the structures of the following compounds
(i) Ethanoic acid (ii) Bromopentane
(iii) Butanone (iv) Hexanol
Are structural isomers possible for bromopentane?
How would you name the following compounds?
(i) 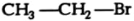 (ii)
(ii) 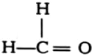 (iii)
(iii) 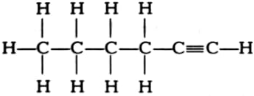
Why is conversion of ethanol to ethanoic acid an oxidation reaction?
A mixture of oxygen and ethyne is used for welding. Can you tell why a mixture of ethyne and air is not used?
How would you distinguish experimentally between and alcohol and a carboxylic acid?
What oxidising agents?
Would you be able to check if water is hard by using a detergent?
People use a variety of methods to wash clothes. Usually, after adding soap, they 'beat' the clothes on a stone or beat it with a paddle scrub with a brush or agitate the mixture in a washing machine. Why is this agitation necessary to get clean clothes?
Ethane with molecular formula has
1. 6 covalent bonds
2. 7 covalent bonds
3. 8 covalent bonds
4. 9 covalent bonds
Butanone is a four carbon compound with functional group
1. carboxylic acid
2. aldehyde
3. ketone
4. alcohol
While cooking, if the bottom of the vessel is getting blackened on the outside, it means that
1. food is not cooked completely
2. the fuel is not burning completely
3. fuel is wet
4. fuel is burning completely
Explain the nature of the covalent bond using the bond formation in
Draw the electron dot structures for
(a) Ethanoic acid (b)
(c) Propanone (c)
What is a homologous series? Explain with an example.
How can ethanol and ethanoic acid can be differentiated on the basis of their physical and chemical properties?
Why does micelle formation take place when soap is added to water? Will a micelle be formed in other solvents such as ethanol also?
Why are carbon and its compounds used as fuel for most applications?
Explain the formation of scum when hard water is treated with soap.
What change will you observe if you test soap with litmus (red or blue)?
What is hydrogenations? What is its industrial application?
Which of the following hydrocarbons undergo addition reaction?
Give a test that can be used to differentiate between butter and cooking oil.
Explain the mechanism of the cleansing action of soaps.
What type of bond is present in F2, molecule?
Draw the electron dot structure for an alkane having molecular formula
"Carbon tetrachloride is not a good conductor of electricity". Give reason to justify this statement.
Diamond is a poor conductor of electricity while graphite is a good conductor. Assign reason.
Why is fullerene so called?
Catenation is the ability of an atom to form bonds with other atoms of the same element.
Compare the catenation property of carbon and silicon.
Which of the following formula represents a saturated hydrocarbon?
Select saturated hydrocarbons from the following:
Which functional group(s) always occurs at the terminal position of a carbon chain?
State the valency of each carbon atom in
(i) an alkane and
(ii) an alkyne.
Draw the structure of butanone molecule.
Write the name and formula of the second member of the carbon compounds having a functional group— OH.
Write the molecular formula of alcohol which can be derived from butane.
Write the name and structure of an alcohol with three carbon atoms in its molecule.
Why do alkanes burn with blue or clean flame?
On burning a hydrocarbon in air, a student obtains sooty flame. What does it indicate?
Under what conditions, an oxidation reaction becomes a combustion reaction?
Name the catalyst commonly used in hydrogenation of oils to form fats.
What is the composition of natural gas used for cooking?
What happens when a small piece of sodium is dropped into ethanol?
How ethanoic acid got its name as glacial acetic acid?
State the part of soap molecule that attaches itself to dirt when soap is dissolved in water.
Name the ions whose presence makes the water hard.
How is scum formed?
What do you mean by the term soapless soaps?
What would be the disadvantage of detergents over soaps?
Apart from the organic compounds, where else do we find carbon?
Mention the form in which it is available there and also its percentage.
How does a carbon atom achieve noble gas configuration?
OR
Explain why carbon generally forms compounds by covalent bond?
In the electron dot structure, the valence shell represented by crosses or dots.
(i) The atomic number of chlorine is 17. Write configuration.
(ii) Draw the electron dot structure of chlorine molecule.
List two characteristics of covalent compounds.
Out of sodium chloride (NaCl) or methyl chloride (CH3Cl), which has higher melting and boiling points? Why?
Allotropy is the virtue of which an element exists in more than one form and each form has different physical but identical chemical properties. Give two examples of crystalline forms of carbon.
Diamond and graphite show different physical properties although they are made up of carbon. Name this relationship between diamond and graphite. Give the basis of this relationship also.
Name an element, other than carbon, which exhibits the property of catenation upto seven or eight atoms. Are these compounds stable?
Draw the electron dot structure of ethyne, also draw its structural formula.
Mention four differences between saturated and unsaturated hydrocarbons.
Out of two more compounds, ethane and ethene which is more reaction? Give reason.
Define the functional groups in a carbon compound. Mention names of any four functional groups with their formulae.
Give the name of the following:
(i) An aldehyde derived from ethane.
(ii) Ketone derived from butane.
Name the following groups present in the following compounds.
(i)
(ii)
(iii)
(iv)
Justify the nature of physical and chemical properties of compounds of homologous series.
How do the melting and boiling points of the hydrocarbons change with increase in molecular mass?
Hydrocarbons X and Y have melting points -190°C and - 90°C, respectively. Which one of these has minimum carbon in the molecule and why?
Draw the possible isomers of the compound with molecular formula and also give their electron dot structures.
Name the following hydrocarbon,
Also mention its reactive site.
"Saturated hydrocarbons burn with a blue flame while unsaturated hydrocarbons burn with a sooty flame". Why?
A compound X with molecular formula burns with a sooty flame. It decolourises bromine water. Identify X. Will it dissolve in water or not? Will it conduct electricity in aqueous solution? Will it have high melting point or low melting point?
A cyclic compound X has molecular formula It is an unsaturated compound and burns with sooty flame. Identify X and write its structural formula.
What is flame? Under what conditions it is called luminous and non-luminous?
(i) When is flame produced?
(ii) Why does combustion of fossil fuels form oxides of sulphur and nitrogen?
What happens when a molecule of hydrogen is added to ethyne? Explain.
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
Unsaturated hydrocarbons contain multiple bonds between the two C-atoms and show addition reaction. Give the test to distinguish ethane from ethene.
A gas is evolved when ethanol reacts with sodium. Name the gas evolved and also write the balanced chemical equation of the reaction involved.
How would you bring about the following conversions? Name the process and write the reaction involved.
(i) Ethanol to ethene
(ii) Propanol to propanoic acid
Write the reactions.
Ethene is formed when ethanol at 443 K is heated with excess of concentrated sulphuric acid. What is the role of sulphuric acid in this reaction? Write the balanced chemical equation of this reaction.
Write the name and molecular formula of an organic compound having its name suffixed with '-ol' and having two C-atoms in the molecule. With the help of a balanced chemical equation indicate what happens when it is heated with excess of conc. H2SO4 ?
Can we use sodium metal for drying alcohol, why?
An organic compound X is an essential constituent of wine and beer, which is responsible for intoxication caused by these drinks. Oxidation of X yields an organic acid Y which is present in vinegar. Name the compounds X and Y and write their structural formulae.
What is esterification?
Or
What happens when ethyl alcohol acetic acid react with each other in the presence of conc. ?
(i) Write the formula and draw the electron dot structure of carbon tetrachloride.
(ii) What is saponification? Write the reaction involved in this process.
Why are soaps not considered as effective cleansing agents in hard water?
Or
Discuss the drawback of using soaps over detergents for cleansing process.
Write the difference between soaps and detergents.
What is covalent bond? What type of bond exists in
(i) CCl4 (ii) CaCl2 and
(iii) CH4? (iii) NH3
Choose the kind of chemical bonding from the ionic bond. covalent bond, both ionic and covalent bonds present in the following compounds.
Potassium chloride, magnesium oxide, sulphuric acid, ammonium hydroxide. zinc sulphide, phosphorus trichloride .
A compound has the formula (Y=non-metal). State the following:
(i) The outer electronic configuration of Y.
(ii) The valency of Y.
(iii) The bonding present in
Give two examples of covalent compounds which you have studied. State any four properties in which covalent compounds differ from ionic compounds.
Give answers to the following statements.
(i) An allotrope of carbon which has a two dimensional layered structure consisting of fused benzene rings.
(ii) An allotrope of carbon which looks like a soccer ball.
(iii) An allotrope of carbon which contains both single and double bonds.
Give the molecular formula and electron dot structure of ethyne and ethene.
Define homologous series. Examine it with an example. Will there be any change in their physical properties? Give reason for your answer.
Write the next higher order homologues of
Name the following compounds.
(i) ![]()
(ii) 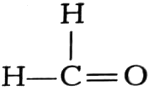
(iii) 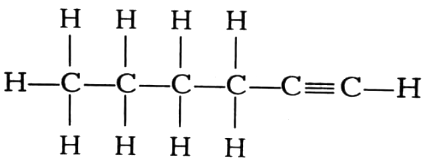
Name the following compounds.
(i) 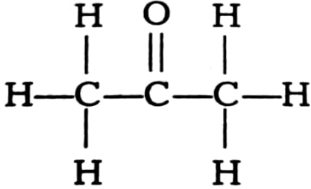
(ii) 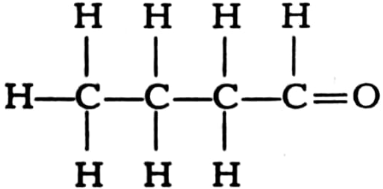
(iii) 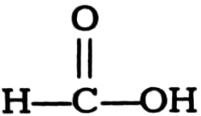
Name the following compounds.
(i) 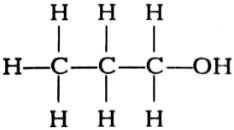
(ii) 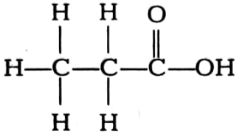
(iii) 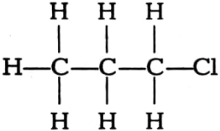
(iv) 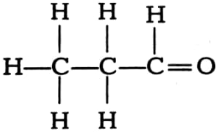
(v) 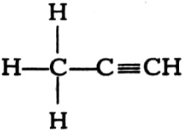
(vi) 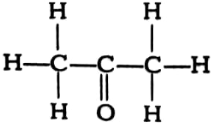
Write the structural formula and IUPAC name of the following:
(i) A carboxylic acid with four C-atoms.
(ii) An alkyne with three C-atoms.
(iii) An alcohol having one C-atom.
Name the type of carbon compounds that can be hydrogenated. With the help of suitable example explain the process of hydrogenation.
Complete the following reactions and name the main product formed in each cash:
(i)
(ii)
(iii) 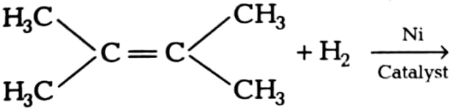
Write the chemical equation for the following conversions, stating the essential conditions.
(i) Ethanol to ethene.
(ii) Propanol to propanoic acid
Name the compound formed when ethanol is heated in excess of concentrated sulphuric acid at 443 K. Also write the chemical equation of the reaction stating the role of concentrated sulphuric acid in it. What would happen, if hydrogen is added to the product of this reaction in the presence of catalyst such as palladium or nickel?
Answer the following questions.
(i) State the functional group present in alcohols.
(ii) Give the general formula of alcohol.
(iii) What is meant by denatured alcohol?
A student reports the police about the illegal vending of alcohol near his school. He also knew about denatured alcohol.
(i) What is denatured alcohol?
(ii) What would happen if somebody consumes denatured alcohol?
An organic compound A is a constituent of many medicines and used as an antifreeze and has the molecular formula . Upon reaction with alk. , the compound A is oxidised to another compound B with formula . Identify the compounds A and B. Write the chemical equation for the reaction which leads to the formation of B.
A compound X is used in cough soluble and many tonics. It is also soluble in water in all proportions.
(i) Name the compound X. Write its chemical formula.
(ii) Which gas is evolved when the compound X reacts with sodium? How will you test the presence of this gas? Write the chemical
equation involved in the reaction of X with sodium.
(iii) Complete the following equation for X and identify Y.
On dropping a small piece of sodium in a test tube containing carbon compound X with molecular formula , a brisk effervescence is observed and a gas Y is produced. On bringing a burning splinter at the mouth of the test tube the gas evolved burns with a pop sound. Identify X and Y.
Also write the chemical equation for the reaction. Write the name and structure of the product formed, when you heat X with excess concentrated sulphuric acid.
An organic compound A on heating with conc. H2SO4 forms a compound B which on addition of one mole of hydrogen in presence of Ni forms a compound C. One mole of compound C, on combustion forms two moles of CO2 and three moles of H2O. Identify the compounds A, B and C, write the chemical equation of the reactions involved.
Name the gases evolved when ethanoic acid is added to sodium carbonate. How would you prove the presence of this gas?
Some esters are added to food items for special smells. An ester can be made from ethanol and ethanoic acid.
(i) Name the ester which is obtained due to the chemical reaction between ethanol and ethanoic acid in the presence of concentrated
sulphuric acid and write the chemical equation.
(ii) Name the process.
Name the product formed when an organic acid and alcohol react in the presence of acid catalyst. Write the equation and give two uses of the product formed.
Name the group of derivatives of hydrocarbons that have pleasant fruity smell. Explain with the help of chemical equation how these can be prepared? Name of the process involved.
Ethyl ethanoate smells like pears and is used for flavouring sweets.
(i) Write the chemical formula of ethyl ethanoate.
(ii) Write the chemical reaction between ethanoic acid and ethanol in the presence of concentrated sulphuric acid.
(iii) Suggest the function of concentrated sulphuric acid in the reaction.
When ethanol reacts with ethanoic acid in the presense of conc. H2SO4 , a substance with fruity smell is produced.
Answer the following.
(i) State the class of compounds to which the fruity smelling compounds belong. Write the chemical equation for the reaction and write
the chemical name of the product formed.
(ii) State the role of conc. H2SO4, in this reaction.
(i) The formula of an ester is Write the structural formulae of the corresponding alcohol and the acid.
(ii) (a) Mention the experimental conditions involved in obtaining ethene from ethanol.
(b) Write the chemical equation for the above equation.
An organic acid X is a liquid which often freezes during winter time in cold countries. It has molecular formula . On warming with ethanol in the presence of a few drops of conc. , a compound Y with sweet smell is formed.
(i) Identify X and Y.
(ii) Write the chemical equation for the reaction involved.
(i) Name the property of ethanol which makes it useful in medicines.
(ii) Name the organic compound which is used in pickles. Mention its composition.
(iii) Mention any two uses of alcohol in medicines.
Name the oxidising agent used for the conversion of ethanol to ethanoic acid. Distinguish between ethanol and ethanol acid on the basis of
(i) litmus test,
(ii) reaction with sodium carbonate.
A compound X is formed by the reaction of a carboxylic acid C2H4O2 and an alcohol in the presence of a few drops of H2SO4. The alcohol on oxidation with alk. KMnO4, followed by acidification gives the same carboxylic add as used in this reaction. Give the names and structures of (i) carboxylic acid, (ii) alcohol and (iii) the compound X. Also write the reaction.
What are soaps chemically?
How do they differ from synthetic detergents? Also, mention their uses?
An aldehyde as well as a ketone can be represented by the same molecular formula, say C3H6O. Write their structures and name them. State the relation between the two in the language of science.
State the reason why carbon can neither form C4+ cations nor C4- anions but forms covalent compounds?
Also state the reasons to explain why covalent compounds
(i) are bad conductors of electricity?
(ii) have low melting and boiling points?
(i) What are hydrocarbons? Give examples.
(ii) Give the structural differences between saturated and unsaturated hydrocarbons with two examples each.
(iii) What is a functional group? Give examples of four different functional groups.
Define structural isomer and draw the isomeric structures of butane. Compare the structure of benzene and cyclohexane by drawing them.
Give an example of each of the following.
(i) A carbon compound containing two double bonds.
(ii) A molecule in which central atom is linked to three other atoms.
(iii) A compound containing both Ionic and covalent bonds.
(iv) An organic compound which is soluble in water.
(v) A carbon compound which burns with a sooty flame.
What is the difference between combustion and oxidation? Under what condition, an oxidation reaction can be called as combustion? Illustrate your answer with one example in each case.
Describe the addition reaction of carbon compounds with its application. State the function of catalyst in this reaction. How this reaction different from a substitution reaction? Explain with an example.
(i) Give a chemical test to distinguish between saturated and unsaturated hydrocarbon.
(ii) Name the products formed when ethane burns in air. Write the balanced chemical equation for the reaction showing the types of
energies liberated.
(ii) Why is reaction between methane and chlorine in the presence of sunlight considered a substitution reaction?
(i) Write the names of the functional groups in
(a) 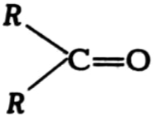 (b)
(b) 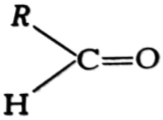
(ii) Describe a chemical test to distinguish between ethanol and ethanoic acid.
(iii) Write a chemical equation to represent what happens when hydrogen gas is passed through an unsaturated hydrocarbons in the
presence of nickel as a catalyst?
Explain the given reactions with the examples.
(i) Hydrogenation reaction
(ii) Oxidation reaction
(iii) Substitution reaction
(iv) Saponification reaction
(v) Combustion reaction
Esters are sweet-smelling substances and are used in making perfumes. Suggest some activity and the reaction involved for the preparation of an ester with well labelled diagram.
(i) How is vinegar made?
(ii) What is glacial acetic acid? What is its melting point?
(iii) Why is butanoic acid a weak acid?
(iv) Write the name and the formula of the two compounds formed when the ester, undergoes saponification.
A salt X is formed and a gas is evolved when ethanoic acid reacts with sodium hydrogen carbonate. Name the salt X and the gas evolved. Describe an activity and draw the diagram of the apparatus to prove that the evolved gas is the one which you have named. Also, write the chemical equation of the reaction involved.
(i) Complete the following reactions and name the main product formed in each case.
(a)
(b)
(c)
(ii) Write the names of the following compounds.
(a) 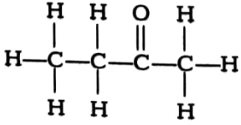
(b) 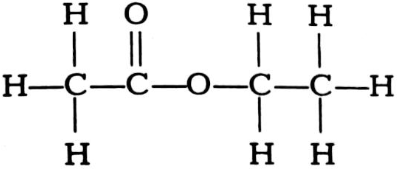
State the funtional group present in each compound.
Identify the compounds A to E in the following reaction sequence.
(i)
(ii)
(iii)
(iv)
(v)
(i) State the litmus test to distinguish between an alcohol and a carboxylic acid.
(ii) Give the equation for the reaction of a carboxylic acid with an alcohol. State the condition for the reaction and name the product
formed. What is this reaction known as?
(iii) Write a reaction which is reverse of this reaction? Mention the conditions required for the reaction. Name and write the use of this
reaction.
Look at the figure and answer the following questions.
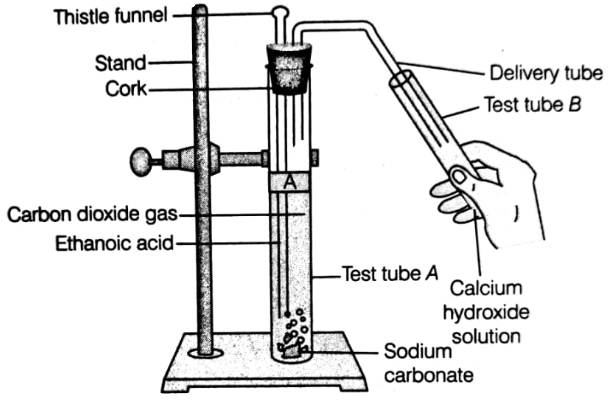
(i) What change would you observe in the calcium hydroxide solution taken in test tube B?
(ii) Write the reactions involved in test tubes A and B respectively.
(iii) If ethanol is given instead of ethanoic acid, would you expect the same change?
(iv) How can a solution of lime water be pre-pared in the laboratory?
An organic compound A is widely used as a preservative in pickles and has a molecular formula C2H2O2. This compound reacts with ethanol to form a sweet smelling compound B.
(i) Identify the compound A.
(ii) Write the chemical equation for its reaction with ethanol to form compound B.
(iii) How can we get compound A from B?
(iv) Name the process and write corresponding chemical equation.
(v) Which gas is produced when compound A reacts with washing soda? Write the chemical equation.
An organic compound A of molecular formula on reducing gives another compound B of molecular formula . B on reaction with chlorine in the presence of sunlight gives C of molecular formula .
(i) Name the compounds A, B and C.
(ii) Write chemical equation for the conversion of A to B.
A compound C (molecular formula, C2H4O2) reacts with Na metal to form a compound R and evolves a gas which burns with a pop sound. Compound C on treatment with an alcohol A in the presence of an acid forms a sweet smelling compound S (molecular formula = C3H6O2).
On addition of NaOH to C, it also gives R and water. S on treatment with NaOH solution gives back R and A.
Identify C, R, A, S and write down the reactions involved.
(i) What are fatty acids?
(ii) Which process is used for the manufacture of soaps?
(iii) What kind of solution is formed by dirt and grease with water-free solution or emulsion?
(iv) What are soap forming structures?
(v) Soaps are biodegradable or non-biodegradable?
Can you suggest a method to separate glycerine (glycerol) from the reaction mixture?
Why does test tube A (used in the experiment) has larger length of foam? When soap is added to it.
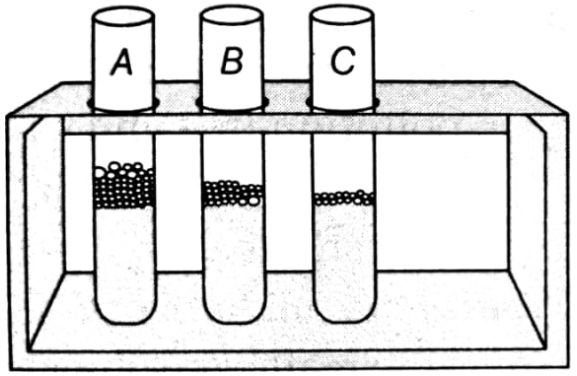
In an experiment to study the properties of acetic acid, answer the following questions.
(i) Name the substance which on addition to acetic acid produces carbon dioxide gas?
(ii) How carbon dioxide gas is tested in the laboratory?
Reena added 20% NaOH solution to 20 mL of castor oil taken in beaker. Suddenly, the beaker becomes hot. What conclusion can you draw from this reaction?
A student added too much of the baking soda in a test tube containing X substance having the smell of vinegar. She observes that reaction mixture now out of the test tube due to formation of effervescence.
(i) Identify the substance X in which the student added baking soda.
(ii) Name the gas due to which the effervescence evolved. Write one chemical test to identify this gas.
What do you observe when you drop a few drops of acetic acid to a test tube containing
(i) phenolphthaleini?
(ii) distilled water?
(iii) universal indicator?
(iii) sodium hydrogen carbonate powder?
Somya and her younger sister were playing, while their mother was washing clothes. Somya a grade 7 student saw her mother scrubbing the clothes with a brush and sometimes beating it with the paddle, she was a bit worried that her clothes will be damaged and will not look good, she tried to stop her mother, but her mother explained her that by beating clothes get cleaned properly, as all the dirt are removed. She smiled as now she understood that her mother cares for her.
(i) According to you, why usually after adding the soaps, people 'beat' the clothes with a paddle, scrub with a brush or the mixture is
agitated in a washing machine. Why is agitation necessary to get clean clothes?
(ii) What kind of values does Somya's mother possess?
Arvind was suffering from cough and cold. He went to the chemist shop and asked for 'Corex' cough syrup. But the chemist refused to give 'Corex' and told Arvind that the sale of this syrup has been banned by the government.
Read the given passage and answer the questions.
(i) What is the main constituent of 'Corex' cough syrup?
(ii) What is absolute alcohol?
(iii) According to you, why the sale of 'Corex' has been banned?
(iv) What values you have learnt from the given text?
A boy was travelling in a CNG auto with his mother. The auto-driver took the auto to the CNG filling station for filling the empty cylinder. The boy asked his mother about the gas which was filled in the auto. His mother replied, this gas is CNG. CNG is used in place of petrol or diesel because it is cheaper, pollution free and eco-friendly.
Read the given passage and answer the following questions.
(i) What is the full form of CNG?
(ii) Name the main constituent of CNG. Also draw its structure.
(iii) What values you have learnt after reading the text?
Mrs Anita observed that her cooking utensils are becoming black in colour and the flame of her gas stove is yellowish in colour. Her neighbour Mrs Anand when came to her, told the reason why her utensils becomes black and advise her to clean the stove or gas burner time to time.
Read the given passage and answer the following questions.
(i) (a) What can be the reason for this sooty flame?
(b) How is this problem harmful to our environment?
(ii) What steps should be taken to stop this process?
(iii) What is the associated value that the learners get from this passage?
Ethanol is used on a large scale. At commercial level, this is a very useful chemical. It is commonly called alcohol and is the active ingredient of alcoholic drink. But consumption of alcohol also causes drunkenness and this practice is socially condemned.
Read the given passage and answer the following questions.
(i) As a responsible student of class, what steps you would take to discourage the use of alcohol?
(ii) What values did learners have learn from the above text?