The species, having bond angles of 120° is :
1. PH3
2. ClF3
3. NCl3
4. BCl3
Which of the following species has a bond angle of \(120^\circ\)?
1. \({SF}_6\)
2. \(NCl_3\)
3. \(BCl_3\)
4. \(PH_3\)
In which of the following molecules, all atoms are coplanar ?
The pair of electrons in the given carbanion, CH3C≡C-, is present in which of the following orbitals?
| 1. | sp3 | 2. | sp2 |
| 3. | sp | 4. | 2p |
The hybridisations of atomic orbitals of nitrogen in NO+, NO-3 and NH3 respectively are:
1.
2.
3.
4.
Pair of ions among the following that are isoelectronic and isostructural:
1.
2.
3.
4.
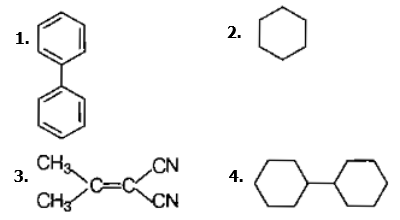
In which of the following molecules, all atoms are coplanar?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
In which orbital is the pair of electrons located in the provided carbanion, CH3C≡C–?
1. sp3
2. sp2
3. sp
4. 2p
| 1. | The H-O-H bond angle in H2O is larger than the H-C-H bond angle in CH4 |
| 2. | The H-C-H bond angle in CH4 is larger than the H-N-H bond angle in NH3 |
| 3. | The H-C-H bond angle in CH4, the H-N-H bond angle in NH3 and the H-0-H bond angle in H2O are all greater than 90o |
| 4. | The H-O-H bond angle in H2O is smaller than the H-N-H bond angle in NH3 |
The correct geometry and hybridization for XeF4 are :
1. Octahedral, sp3d2
2. Trigonal bipyramidal, sp3d3
3. Planar triangle, sp3d3
4. Square planar, sp3d2
