Lewis symbols for the atoms and ions: S and S2–; Al and Al3+; H and H– respectively, would be:
1.




2.




3.




4.




















The favourable factors for the formation of an ionic bond would be:
| 1. | Low ionization enthalpy, High negative electron gain enthalpy, High lattice energy. |
| 2. | High ionization enthalpy, High electron gain enthalpy, High lattice energy. |
| 3. | Low ionization enthalpy, Low electron gain enthalpy, Low lattice energy. |
| 4. | High ionization enthalpy, Low electron gain enthalpy, High lattice energy. |
The correct relationship between bond strength and bond order is :
| 1. | Bond strength is directly proportional to the bond order. |
| 2. | Bond strength is inversely proportional to the bond order. |
| 3. | There is no relationship between these two terms. |
| 4. | None of these. |
The bond length can be defined as -
| 1. | The equilibrium distance between the nuclei of two bonded atoms in a molecule. |
| 2. | The farthest distance between the nuclei of two bonded atoms in a molecule. |
| 3. | The shortest distance between the nuclei of two bonded atoms in a molecule. |
| 4. | None of the above. |
can be represented by structures 1 and 2 shown below.
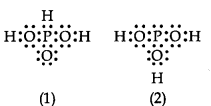
These two structures cannot be taken as the canonical forms of the resonance hybrid, because :
1. The positions of the atoms have changed.
2. The positions of the atoms are constant.
3. H3PO3 does not show resonance.
4. Two hydrogen atoms are missing.
The shape of molecule is bent while that of is linear. The correct explanation is :
| 1. | The dipole moment of carbon dioxide is 3.84 D and water is zero |
| 2. | The dipole moment of water is zero and carbon dioxide is 1.84 D |
| 3. | The dipole moment of carbon dioxide is zero and water is 1.84 D |
| 4. | The dipole moment of carbon dioxide is 3.84 D and water is 1.84 D |
The application of dipole moment is/are :
| 1. | It is used to differentiate polar and non-polar bonds. |
| 2. | It is helpful in calculating the percentage ionic character of a molecule. |
| 3. | Both '1' and '2' |
| 4. | None of the above. |
The difference between electronegativity and electron gain enthalpy is :
| 1. | An element has a constant value of the electron gain enthalpy but not of electronegativity. |
| 2. | There is no difference between these two terms. |
| 3. | An element has a constant value of electronegativity but not of electron gain enthalpy. |
| 4. | None of the above. |
The correct order of increasing ionic character in the molecules: LiF, K2O, and ClF3 is -
1.
2.
3.
4.
CH4 does not exhibit square planar geometry because:
| 1. | For square planar geometry, 5 bonds are required. |
| 2. | Carbon does not have d-orbitals to undergo dsp2 hybridization. |
| 3. | Due to steric hindrance CH4 does not exhibit square planar geometry. |
| 4. | Carbon does not have d-orbitals to undergo d2sp3 hybridization. |
