| Assertion (A): | Half-filled and fully-filled degenerate orbitals are more stable. |
| Reason (R): | Extra stability is due to the symmetrical distribution of electrons and high exchange energy. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Shell & Subshell |
93%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
| Assertion (A): | In the third energy level, there is no f subshell. |
| Reason (R): | For n = 3, the possible values of l are 0, 1, 2 and for f subshell l = 3. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Shell & Subshell |
78%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
Links
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
| Assertion (A): | All d orbitals are tetra-lobed structures having two nodal planes. |
| Reason (R): | Out of five, only four d-orbitals are degenerate orbitals. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Subtopic: Shell & Subshell |
50%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
Links
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
The graph that represents the variation of (probability of density of finding the electron) with distance (r) is given below:
The number of nodes as per the graph is-
1. zero
2. 1
3. 2
4. Cannot predict
Subtopic: Shell & Subshell |
62%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
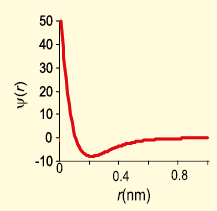
The graph represents the variation of the wave function with distance from the nucleus.
The atomic orbital represented by above-mentioned graph is -
1. 1s
2. 2s
3. 2p
4. None of the above
Subtopic: Shell & Subshell |
52%
From NCERT
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh
Hints
To view explanation, please take trial in the course.
NEET 2023 - Target Batch - Aryan Raj Singh