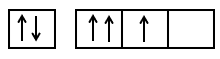
The incorrect statement among the following is:
1.
Total orbital angular momentum of an electron in 's' orbital is equal to zero.
2.
An orbital is designated by three quantum numbers, while an electron in an atom is designated by four quantum numbers.
3.
The electronic configuration of N atom is 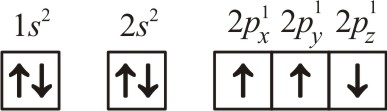
4.
The value of m for dz2 is zero.

The number of d-electrons in Fe2+ (Z = 26) is not equal to the number of:
1. s-electrons in Mg (Z = 12)
2. p-electrons in Cl (Z = 17)
3. d-electrons in Fe (Z = 26)
4. p-electrons in Ne (Z = 10)
The angular momentum of electrons in d orbital is equal to:
1.
2.
3.
4. 0
The correct set of four quantum numbers for the valence electron of a rubidium atom (Z =37) is:
| 1. | \(5,1,1,+ {1 \over2}\) | 2. | \(6,0,0,+ {1 \over2}\) |
| 3. | \(5,0,0,+ {1 \over2}\) | 4. | \(5,1,0,+ {1 \over2}\) |
If n = 6, the correct sequence for the filling of electrons will be:
1.
2.
3.
4.
The rule used to determine the maximum number of electrons in a subshell of an atom is:
| 1. | 4l+2 | 2. | 2l+1 |
| 3. | 4l-2 | 4. | 2n2 |
The incorrect set of quantum numbers among the following is:
1. n= 4, l= 0, m= 0, s= -1/2
2. n= 5, l= 3, m= 0, s= +1/2
3. n= 3, l= 2, m= -3, s= -1/2
4. n= 3, l= 2, m= 2, s= -1/2
The orbital diagram in which both Pauli's exclusion principle and Hund's rule are violated is:
1.
2.
3.
4.
The correct order of the total number of nodes of atomic orbitals is:
1. 4f > 6s > 5d
2. 6s > 5d > 4f
3. 4f > 5d > 6s
4. 5d > 4f > 6s
Correct statement among the following is:
1. Number of angular nodes = n - l - 1
2. Number of radial nodes = l
3. Total number of nodes = n -1
4. All of the above