Pressure versus temperature graph of an ideal gas at constant volume V of an ideal gas is shown by the straight line A. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the line

1. A
2. B
3. C
4. None of these

Two different isotherms representing the relationship between pressure P and volume V at a given temperature of the same ideal gas are shown for masses and of the gas respectively in the figure given, then
1.
2.
3.
4. None of these
Two different masses m and 3m of an ideal gas are heated separately in a vessel of constant volume, the pressure P and absolute temperature T, graphs for these two cases are shown in the figure as A and B. The ratio of slopes of curves B to A is
1. 3 : 1
2. 1 : 3
3. 9 : 1
4. 1 : 9
Under constant temperature, graph between and is
1. Parabola
2. Hyperbola
3. Straight line
4. Circle
Which one, of the following, graphs represents the behaviour of an ideal gas at constant temperature?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
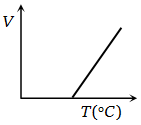
Volume-temperature graph at atmospheric pressure for a monoatomic gas is
1. 
2.
3. 
4.
From the following P-T graph what interference can be drawn
1.
2.
3.
4. None of the above
The figure shows the volume V versus temperature T graphs for a certain mass of a perfect gas at two constant pressures of and . What interference can you draw from the graphs
1.
2.
3.
4. No interference can be drawn due to insufficient information
A volume V and pressure P diagram was obtained from state 1 to state 2 when a given mass of a gas is subjected to temperature changes. During this process the gas is
1. Heated continuously
2. Cooled continuously
3. Heated in the beginning and cooled towards the end
4. Cooled in the beginning and heated towards the end
A pressure P - absolute temperature T diagram was obtained when a given mass of gas was heated. During the heating process from the state 1 to state 2 the volume
1. Remained constant
2. Decreased
3. Increased
4. Changed erratically