Two flasks A and B of equal volume containing and HCl gases, are connected by a narrow tube of negligible volume. The two gases were prevented from mixing by stopper fitted in connecting tube. For further detail of experiment refer to the given figure. What will be final pressure in each flask when passage connecting two tubes are opened. Assume ideal gas behaviour of and gas and the reaction.
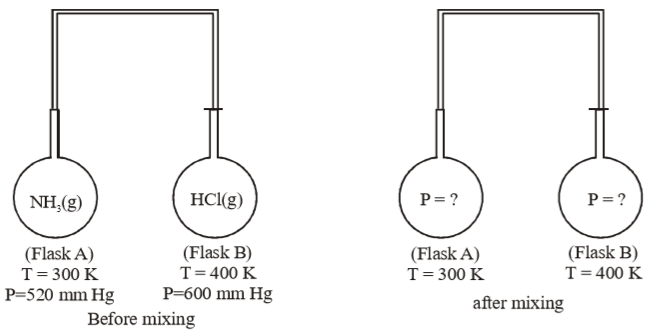
(1) 40 mm Hg
(2) 60 mm Hg
(3) 20 mm Hg
(4) 10 mm Hg

The ratio of average molecular kinetic energy of to that of , both at 300 K is
(1) 1 : 1
(2) 7 : 2
(3) 176 : 1
(4) 2 : 7
A mono atomic gas diatomic gas and triatomic gas are mixed, taking one mole of each for the mixture is
(1) 1.40
(2) 1.428
(3) 1.67
(4) 1.33
According to kinetic theory of gases, for a diatomic molecule
(1) The pressure exerted by the gas is proportional to the mean velocity of the molecule.
(2) The pressure exerted by the gas is proportional to the root mean square velocity of the molecule.
(3) The root mean square velocity of the molecule is inversely proportional to the temperature.
(4) The mean translational kinetic energy of the molecule is proportional to the absolute temperature.
The values of vander waals constant ‘a’ for the gases , , and are 1.36, 1.39, 4.17 and 2.253 atom mol-2 respectively. The gas which can most easily be liquefied is
(A)
(B)
(C)
(D)
At low pressure, the vander waals equation is written as :
(1)
(2)
(3)
(4)
X ml of gas effuses through a hole in a container in 5 seconds. The time taken for the effusion of the same volume of the gas specified below under ideal condition is
(1) 10 seconds :
(2) 20 seconds :
(3) 25 seconds :
(4) 55 seconds :
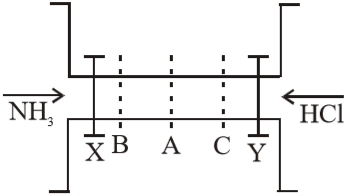
The valves X and Y are opened simultaneously. The white fumes of will first form at :
(1) A
(2) B
(3) C
(4) A, B and C simultaneously
A gas mixture consists of 2 moles of oxygen and 4 moles of a argon at temperature T. Neglecting all vibrational modes, the total internal energy of the system is –
(1) 4RT
(2) 5RT
(3) 15RT
(4) 11RT
Given reaction : . Calculate the volume at STP from 48 gm of carbon and excess
(A) 179.2 lit.
(B) 89.6 lit.
(C) 44.8 lit.
(D) 22.4 lit.