In the given conformation C2 is rotated about C2-C3 bond anticlockwise by an angle of 120° then the conformation obtained is

1. Fully eclipsed conformation
2. Partially eclipsed conformation
3. Gauche conformation
4. Staggered conformation

What is the decreasing order of stability of the carbocation ions?
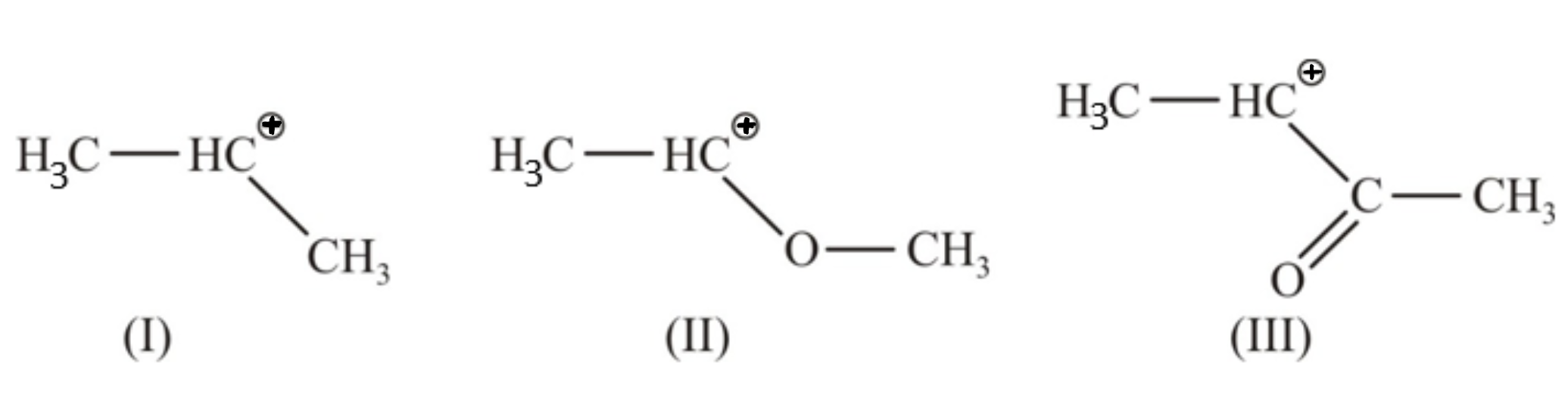
1. I>II>III
2. II>III>I
3. III>I>II
4. II>I>III
The most acidic compound among the following is:
| 1. | o-Cresol | 2. | p-Nitrophenol |
| 3. | Phenol | 4. | m-Cresol |
The correct order of basicities of the following compounds is:
(1)
(2)
(3)
(4)
1. 2>1>3>4
2. 1>3>2>4
3. 3>1>2>4
4. 1>2>3>4
Consider the acidity of the following carboxylic acids
(i) PhCOOH
(ii) o-NO2C6H4COOH
(iii) p-NO2C6H4COOH
(iv)m-NO2C6H4COOH
1. i>ii>iii>iv
2. ii>iv>iii>i
3. ii>iv>i>iii
4. ii>iii>iv>i
The IUPAC name of
1. 2,3-diethyl butane
2. 3,4-dimethyl hexane
3. 3-methyl pentane
4. 2-ethyl 3-methyl pentane
Hybridisation of N in pyrrole 
1. sp
2. sp2
3. sp3
4. sp3d
The most reactive of these compounds towards sulphonation is
1. Toluene 2. Chlorobenzene
3. Nitrobenzene 4. m-Xylene
In the above compound, arrange the nitrogen according to their decreasing basic strength.
1. 1>2>3>4
2. 4>3>1>2
3. 2>4>1>3
4. 3>4>1>2
The correct stability order for the following species is:
1. II>IV>I>III
2. II>I>III>IV
3. II>I>IV>III
4. I>III>II>IV




